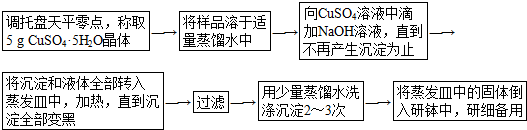
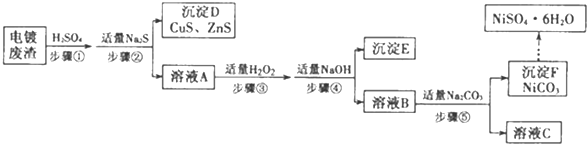
��Ŀ����
3��A��B��C��D��Ϊ��ѧ��ѧ�ij��������Ҿ�����ͬһ��Ԫ�أ�����֮���ת����ϵ��ͼ��ʾ����Ӧ���������������Ѿ���ȥ����A$\stackrel{O_{2}}{��}$B$\stackrel{O_{2}}{��}$C$\stackrel{H_{2}O}{��}$D��1����A��D��ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬B�ڳ���ʱΪ���壬��AΪ����д��ѧʽ��H2S��д��B��Cת���Ļ�ѧ��Ӧ����ʽ��2SO2+O2$\stackrel{������}{?}$2SO3��
��2����A��ˮ��Һ��ʹʪ��ĺ�ɫʯ����ֽ������D��ϡ��Һ��ʹʪ�����ɫʯ����ֽ���
��A�Ļ�ѧʽΪNH3��ʵ�����Ʊ�����A�Ļ�ѧ��Ӧ����ʽΪ2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$2NH3��+CaCl2+2H2O��A��Bת���Ļ�ѧ����ʽΪ4NH3+5O2$\frac{\underline{����}}{��}$NO+6H2O��
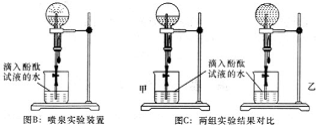
�ڼס�������ͬѧ�ø����Բ����ƿ���ռ�һƿA���壬����ͼB��Ȫʵ���װ�ý���ʵ�飬�ܹ۲쵽�����ĺ�ɫ��Ȫ���÷���ʽ������Ȫ�ʺ�ɫ��ԭ��NH3+H2O?NH3��H2O?NH4++OH-��
�ۼס�������ͬѧ�����Ȫʵ���Բ����ƿ��������Һ��ͼC��ʾ����ͨ������ȷ�ϣ�����ͬѧ������Һ�����ʵ���Ũ�ȵ��ڣ�����ڡ�����С�ڡ����ڡ�������ͬѧ������Һ�����ʵ���Ũ�ȣ�
��3����AΪ���ý���Ԫ�صĵ��ʣ�DΪǿ���ɫ��Ӧ�Ի�ɫ����B�ǣ��ѧʽ��Na2O��C����Ϊ������ߵ�������C�������̼������Ӧ�Ļ�ѧ����ʽΪ2Na2O2+2CO2=2Na2CO3+O2����C��ˮ������Ӧ�����ӷ���ʽΪ2Na2O2+2H2O=4Na++4OH-+O2����
���� ��1����A��D��ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬˵����Һ�����ԣ�B�ڳ���ʱΪ���壬��AΪH2S��BΪSO2��CΪSO3��DΪH2SO4��
��2����A��ˮ��Һ��ʹʪ��ĺ�ɫʯ����ֽ��������A��NH3��D��ϡ��Һ��ʹʪ�����ɫʯ����ֽ��죬Ϊ�ᣬ��������������NO��NO��������������NO2������B��NO��C��NO2��D��HNO3��
��3����AΪ���ý���Ԫ�صĵ��ʣ�DΪǿ���ɫ��Ӧ�Ի�ɫ����AΪNa��BΪNa2O��CΪNa2O2����DΪNaOH��
��� �⣺��1����A��D��ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬˵����Һ�����ԣ�B�ڳ���ʱΪ���壬��AΪH2S��BΪSO2��CΪSO3��DΪH2SO4��B��Cת���Ļ�ѧ��Ӧ����ʽ��2SO2+O2$\stackrel{������}{?}$2SO3��
�ʴ�Ϊ��H2S��2SO2+O2$\stackrel{������}{?}$2SO3��
��2����A��ˮ��Һ��ʹʪ��ĺ�ɫʯ����ֽ��������A��NH3��D��ϡ��Һ��ʹʪ�����ɫʯ����ֽ��죬Ϊ�ᣬ��������������NO��NO��������������NO2������B��NO��C��NO2��D��HNO3��
��A�Ļ�ѧʽΪ��NH3��ʵ�����Ʊ�����A�Ļ�ѧ��Ӧ����ʽΪ��2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$2NH3��+CaCl2+2H2O��A��Bת���Ļ�ѧ����ʽΪ��4NH3+5O2$\frac{\underline{����}}{��}$NO+6H2O��
�ʴ�Ϊ��NH3��2NH4Cl+Ca��OH��2$\frac{\underline{\;\;��\;\;}}{\;}$2NH3��+CaCl2+2H2O��4NH3+5O2$\frac{\underline{����}}{��}$NO+6H2O��
�ڸ���ͼB��Ȫʵ���װ�ý���ʵ�飬�ܹ۲쵽�����ĺ�ɫ��Ȫ���÷���ʽ������Ȫ�ʺ�ɫ��ԭ��NH3+H2O?NH3��H2O?NH4++OH-��
�ʴ�Ϊ��NH3+H2O?NH3��H2O?NH4++OH-��
����Һ������ڰ��������������ͬѧ������Һ�����ʵ���Ũ�ȵ�������ͬѧ������Һ�����ʵ���Ũ�ȣ�
�ʴ�Ϊ�����ڣ�
��3����AΪ���ý���Ԫ�صĵ��ʣ�DΪǿ���ɫ��Ӧ�Ի�ɫ����AΪNa��BΪNa2O��CΪNa2O2����DΪNaOH�����������������̼������Ӧ�Ļ�ѧ����ʽΪ��2Na2O2+2CO2=2Na2CO3+O2������������ˮ������Ӧ�����ӷ���ʽΪ��2Na2O2+2H2O=4Na++4OH-+O2����
�ʴ�Ϊ��Na2O��2Na2O2+2CO2=2Na2CO3+O2����2Na2O2+2H2O=4Na++4OH-+O2����
���� ���⿼�������ƶϣ��������ʵ���ɫ�����ʵ����ʽ����ƶϣ�����������ѧ����������Ӧ���Ѷ��еȣ�

��1��������Ӧ��������N2����֪�÷�Ӧÿת��1mole-���ų�132.6kJ���������÷���ʽ�ġ�H=-1591.2kJ/mol��
��2�����жϸ÷�Ӧ�������������ܱ������н��У��Ѿ��ﵽƽ��״̬����ACD
A����̿���������ٱ仯 B��N2 ��CO����֮��Ϊ1��3
C������6molCOͬʱ����1mol Si3N4 D�����������ܶȲ��ٱ仯
��3�����д�ʩ�п��Դٽ�ƽ�����Ƶ���BD
A�������¶� B������ѹǿ C����������SiO2 D������N2
��4���÷�Ӧ���¶ȿ�����1300-1700��C��ԭ�����¶�̫�ͣ���Ӧ����̫�ͣ��÷�Ӧ�Ƿ��ȷ�Ӧ���¶�̫�ߣ������ڷ�Ӧ���ҽ��У�
��5��ij�¶��£���ø÷�Ӧ��N2��CO����ʱ�̵�Ũ�����£���0-20min��N2��ƽ����Ӧ����3.70��102mol/��L•min�������¶��£���Ӧ��ƽ�ⳣ��K=81.0��
| ʱ��/min | 0 | 5 | 10 | 15 | 20 | 25 | 30 | 35 | 40 | 45 |
| N2Ũ��mol•L-1 | 4.00 | 3.70 | 3.50 | 3.36 | 3.26 | 3.18 | 3.10 | 3.00 | 3.00 | 3.00 |
| COŨ��/mol•L-1 | 0.00 | 0.90 | 1.50 | 1.92 | 2.22 | 2.46 | 2.70 | - | - | - |
| A�� | �٢ڢۢ� | B�� | �ڢ� | C�� | �٢ڢ� | D�� | �ڢ� |
| A�� | SiO2��CO2���ƣ���һ����ԭ�Ӻ�������ԭ�ӹ���һ��SiO2���� | |
| B�� | ������һ�㲻����ˮ������ˮ�ࡢ�մɡ���������Ҫ�ɷ� | |
| C�� | SiO2������������ܺͼӦ�����ܺ��ᷴӦ | |
| D�� | ��Ԫ���ǹ��ɿ�����ʯ����Ҫ�ɷ�֮һ���仯��̬����ȫ��������������� |
| A�� | �μ�����ʱ���Ȳ����������ܽ� | |
| B�� | ��������Ĥ | |
| C�� | ���ȡ����ɡ����գ����ն���Fe2O3���� | |
| D�� | ���ж����ЧӦ |
| A�� | v��NH3����=2v��CO2���� | B�� | �ܱ���������ѹǿ���� | ||
| C�� | �ܱ������а���������������� | D�� | �ܱ������л��������ܶȲ��� |
| ѡ�� | ʵ����� | ���� | ���� |
| A | �ֱ����Na2CO3��NaHCO3���� | �Թ��ڱڶ���ˮ�� | �������ʾ����ȷֽ� |
| B | ����ʢ��NH4Cl������Թ� | �Թܵײ�������ʧ���Թܿ��о������ᣮ | NH4Cl����������� |
| C | ��I-����ɫ��Һ�еμ�����������ˮ���ٵμӵ�����Һ | ������ۺ���Һ�����ɫ | �����ԣ�Cl2��I2 |
| D | ��FeSO4��Һ���ȵ���KSCN��Һ�ٵμ�H2O2��Һ | ����H2O2����Һ���Ѫ��ɫ | Fe2+�������������л�ԭ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |