��Ŀ����
������Դ�������þ��й�����ǰ����ij����ˮ����Ҫ���ӵĺ������±���
��1���ú���ˮ��Ca2+�����ʵ���Ũ��Ϊ mol/L��ij��ѧ�Ļ�ѧʵ������ ����д�������Լ����ƣ���ú�ˮ��pH=8.2���ú�ˮ�������Ե�ԭ���ǣ������ӷ���ʽ��ʾ�� ��
��2���ö��Ե缫���ú���ˮһ��ʱ��������������ˮ������ɷ�ΪCaCO3��Mg��OH��2����д������CaCO3�����ӷ���ʽ ��
��3�������dz����ĺ���Ʒ��ijС��ͬѧ��������ʵ��̽�������е�Ԫ�صĴ��ڣ�

�ٲ�����Ϊ���գ�������ʱ�� ʢװ������
�ڲ������Ǹ�С�����ҺA�е�Ԫ�صĴ�����ʽ���е�̽��ʵ�飮
���Ʋ⡿��������ʽ����IO3-�� ����I-��ʽ����
���������ϡ���IO3-���н�ǿ�������ԣ��ܽ�Fe2+����������ԭ����ΪI-��
��������ҺAϡ�����Ƴ�200mL��Һ�����������ʵ��̽������ѡ�Լ���3% H2O2��Һ��KSCN��Һ��FeCl2��Һ��������Һ��ϡ����
| �ɷ� | Na+ | K+ | Ca2+ | Mg2+ | Cl- | SO42- | HCO3+ |
| ����/mg?L-1 | 9360 | 83 | 200 | 1100 | 16000 | 1200 | 118 |
��2���ö��Ե缫���ú���ˮһ��ʱ��������������ˮ������ɷ�ΪCaCO3��Mg��OH��2����д������CaCO3�����ӷ���ʽ
��3�������dz����ĺ���Ʒ��ijС��ͬѧ��������ʵ��̽�������е�Ԫ�صĴ��ڣ�

�ٲ�����Ϊ���գ�������ʱ��
�ڲ������Ǹ�С�����ҺA�е�Ԫ�صĴ�����ʽ���е�̽��ʵ�飮
���Ʋ⡿��������ʽ����IO3-�� ����I-��ʽ����
���������ϡ���IO3-���н�ǿ�������ԣ��ܽ�Fe2+����������ԭ����ΪI-��
��������ҺAϡ�����Ƴ�200mL��Һ�����������ʵ��̽������ѡ�Լ���3% H2O2��Һ��KSCN��Һ��FeCl2��Һ��������Һ��ϡ����
| ��� | ʵ�鷽�� | ʵ������ | ���� |
| �� | ȡ����ϡ�ͺ����ҺA���������Һ�����������ữ����װ���Թܢ� | ������ | |
| �� | ���Թܢ��м��� | ������ | ���պ��Ԫ�ز�����IO3-��ʽ���� |
| �� | ���Թܢ��м��� | ���պ��Ԫ����I-��ʽ���� |
���㣺��ˮ��Դ�����ۺ�����
ר�⣺Ԫ�ؼ��仯����
��������1������n=
����lLCa2+�����ʵ�����������PHֵ�ľ�ȷ��ȷ����������������ˮ����������
��2�������������ӵõ����ӷ�����ԭ��Ӧ��������������������Ũ������ˮ�к���̼��������ӡ������ӣ����������Ӻ�̼��������ӷ�Ӧ����̼�����������������ɰ�ɫ����̼��ƣ�
��3������������ͼ��������������������������У�
�������������Ӿ��л�ԭ�ԣ������������������Һ�о��������ԣ������Ƿ��������ӣ�
�������Լ�����������������Կ�������������Ϊ�ⵥ�ʣ��������۱�����
| m |
| M |
��2�������������ӵõ����ӷ�����ԭ��Ӧ��������������������Ũ������ˮ�к���̼��������ӡ������ӣ����������Ӻ�̼��������ӷ�Ӧ����̼�����������������ɰ�ɫ����̼��ƣ�
��3������������ͼ��������������������������У�
�������������Ӿ��л�ԭ�ԣ������������������Һ�о��������ԣ������Ƿ��������ӣ�
�������Լ�����������������Կ�������������Ϊ�ⵥ�ʣ��������۱�����
���
�⣺��1��Ca2+�����ʵ���Ũ��ΪC��Ca2+��=
=5��10-3mol/L������pH��ֽ��pH�ƿ��Բ�ú�ˮ��pH=8.2��̼���������ˮ�⣬��Һ�ʼ��ԣ�HCO3-+H2O?H2CO3+OH-��
�ʴ�Ϊ��5��10-3������pH��ֽ��pH�ƣ�HCO3-+H2O?H2CO3+OH-��
��2�������������ӵõ����ӷ�����ԭ��Ӧ��������������������Ũ������ˮ�к���̼��������ӡ������ӣ����������Ӻ�̼��������ӷ�Ӧ����̼�����������������ɰ�ɫ����̼��ƣ�����̼��Ƴ��������ӷ���ʽΪ��Ca2++OH-+HCO3-=CaCO3��+H2O��
�ʴ�Ϊ��Ca2++OH-+HCO3-=CaCO3��+H2O��
��3��������ͼ��������������������������У�
�ʴ�Ϊ��������
�������������Ӿ��л�ԭ�ԣ������������������Һ�о��������ԣ������Ƿ��������ӣ����Թ�I�м����Ȼ�������Һ�������2��KSCN��Һ������Ѫ��ɫ����֤��������IO3-��ʽ���ڣ�
�ʴ�Ϊ��FeCl2��Һ�������2��KSCN��Һ����
�����Թ�II�м���3%����������������Կ�������������Ϊ�ⵥ�ʣ��������۱�����
�ʴ�Ϊ��3%H2O2��Һ������Һ���ֱ�����
| ||
| 1L |
�ʴ�Ϊ��5��10-3������pH��ֽ��pH�ƣ�HCO3-+H2O?H2CO3+OH-��
��2�������������ӵõ����ӷ�����ԭ��Ӧ��������������������Ũ������ˮ�к���̼��������ӡ������ӣ����������Ӻ�̼��������ӷ�Ӧ����̼�����������������ɰ�ɫ����̼��ƣ�����̼��Ƴ��������ӷ���ʽΪ��Ca2++OH-+HCO3-=CaCO3��+H2O��
�ʴ�Ϊ��Ca2++OH-+HCO3-=CaCO3��+H2O��
��3��������ͼ��������������������������У�
�ʴ�Ϊ��������
�������������Ӿ��л�ԭ�ԣ������������������Һ�о��������ԣ������Ƿ��������ӣ����Թ�I�м����Ȼ�������Һ�������2��KSCN��Һ������Ѫ��ɫ����֤��������IO3-��ʽ���ڣ�
�ʴ�Ϊ��FeCl2��Һ�������2��KSCN��Һ����
�����Թ�II�м���3%����������������Կ�������������Ϊ�ⵥ�ʣ��������۱�����
�ʴ�Ϊ��3%H2O2��Һ������Һ���ֱ�����
���������⿼��������Ũ�ȼ��㣬���ԭ���������Ʊ������̷����жϣ�ʵ�鷽�������ԭ��Ͳ��裬��Ӧ��������������ǽ���ؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���з�Ӧ�����ӷ���ʽ��ȷ���ǣ�������
| A��Cu��Ũ���ᷴӦ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O |
| B��������þ��ϡ���ᷴӦ��H++OH-=H2O |
| C��Ca��HCO3��2��Һ�������NaOH��Һ��Ӧ��Ca2++2HCO3-+2OH-=CaCO3��+CO32-+2H2O |
| D�������ʵ���Ũ�ȡ��������NH4HCO3��Һ��NaOH��Һ���ϣ�NH4++HCO3-+2OH-=NH3?H2O+CO32-+H2O |

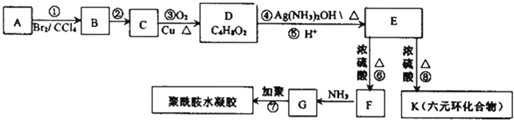
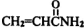 �������еȶ��ԣ�A-K�����л�����ת����ϵ��ͼ��ʾ�����������Ͳ�����ȥ������֪��AΪ��̬������״�����ܶ�Ϊ2.5g/L���Һ˴Ź���������4�����շ壻G�ȱ�ϩ������һ��CH2ԭ���ţ�
�������еȶ��ԣ�A-K�����л�����ת����ϵ��ͼ��ʾ�����������Ͳ�����ȥ������֪��AΪ��̬������״�����ܶ�Ϊ2.5g/L���Һ˴Ź���������4�����շ壻G�ȱ�ϩ������һ��CH2ԭ���ţ�