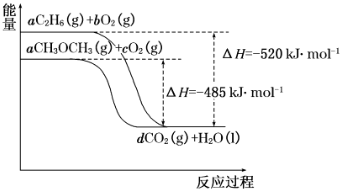
��Ŀ����
11���±���Ԫ�����ڱ���һ���֣���Ա��еĢ١�����Ԫ�أ���д���пհף�| ���� ���� | ��A | ��A | ��A | ��A | ��A | �� | V��A | 0 |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� |
��2���ڢۢ�����Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳��ΪCl��N��O����Ԫ�ط��ţ�
��3��������������ˮ�����У�������ǿ�Ļ�����Ļ�ѧʽHClO4��������ǿ�Ļ�����������������أ�
��4��Ԫ�آݵ���������Ԫ�آܵ�����������Ӧˮ���ﷴӦ�Ļ�ѧ����ʽΪ��Al2O3+2NaOH=2NaAlO2+H2O��
��5��Ԫ�آ͢��γɵ���̬�⻯����ȶ���HCl���ѧʽ�����÷���ʽ��˵��Ԫ�آߵķǽ����ԱȢķǽ���ǿ����ʵCl2+H2S=S��+2HCl��һ������������ʽ���ɣ�
��6����ʾ������γɵĻ�����ĵ���ʽ
 ��д������һ����;�������ȣ�
��д������һ����;�������ȣ�
���� ��Ԫ�������ڱ��е�λ�ÿ�֪����ΪC����ΪN����ΪO����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK��
��1��ϡ������ԭ�������Ϊ�ȶ��ṹ����ѧ��������ã�
��2��ͬ�����������ԭ�Ӱ뾶��С��һ����Ӳ�Խ��ԭ�Ӱ뾶Խ��
��3��������������ˮ�����У�������ǿ���Ǹ����ᣬ����Ԫ����K�Ľ�������ǿ�����������صļ�����ǿ��
��4��Ԫ�آݵ�������ΪAl2O3��Ԫ�آܵ�����������Ӧˮ����ΪNaOH�����߷�Ӧ����ƫ��������ˮ��
��5���ǽ�����Խǿ����Ӧ�⻯��Խ�ȶ����������õ���֮����û��Ƚ�����֤��
��6����ʾ������γɵĻ�����ΪCO2�ȣ���������������
��� ��Ԫ�������ڱ��е�λ�ÿ�֪����ΪC����ΪN����ΪO����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK��
��1��ϡ������Arԭ�������Ϊ�ȶ��ṹ����ѧ��������ã��ʴ�Ϊ��Ar��
��2��ͬ�����������ԭ�Ӱ뾶��С��һ����Ӳ�Խ��ԭ�Ӱ뾶Խ��ԭ�Ӱ뾶��Cl��N��O���ʴ�Ϊ��Cl��N��O��
��3��������������ˮ�����У�������ǿ����HClO4������Ԫ����K�Ľ�������ǿ�����������صļ�����ǿ���ʴ�Ϊ��HClO4���������أ�
��4��Ԫ�آݵ�������ΪAl2O3��Ԫ�آܵ�����������Ӧˮ����ΪNaOH�����߷�Ӧ����ƫ��������ˮ����Ӧ����ʽΪ��Al2O3+2NaOH=2NaAlO2+H2O���ʴ�Ϊ��Al2O3+2NaOH=2NaAlO2+H2O��
��5���ǽ�����Cl��S���ǽ�����Խǿ����Ӧ�⻯��Խ�ȶ������⻯���ȶ��ԣ�HCl��H2S���������õ���֮����û��Ƚ�����֤����Ӧ����ʽΪ��Cl2+H2S=S��+2HCl��
�ʴ�Ϊ��HCl��Cl2+H2S=S��+2HCl��
��6����ʾ������γɵĻ�����ΪCO2�ȣ�������̼����ʽΪ ����������������
����������������
�ʴ�Ϊ�� ���������ȣ�
���������ȣ�
���� ���⿼��Ԫ�����ڱ���Ԫ�������ɣ��Ƚϻ�����ע������ԡ��ǽ�����ǿ���Ƚ�ʵ����ʵ��

 ��У����ϵ�д�
��У����ϵ�д�ʵ����Ʊ�NaNO2
��С���������֪��2NO+Na2O2=2NaNO2��2NO2+Na2O2=2NaNO3
����Ʊ�װ�����£��г�װ����ȥ����

��1�����û��Bװ�ã�C�з����ĸ���Ӧ��2Na2O2+2H2O=4NaOH+O2����2NO+O2=2NO2��
��2��װ��D�ɽ�ʣ���NO������NO3-��������Ӧ�����ӷ���ʽΪ5NO+3MnO4-+4H+=5NO3-+3Mn2++2H2O��
��3����ͬѧ�����װ�����������ú����ʵ�飬�����Ƶõ�NaNO2�л��н϶��NaNO3���ʣ�������A��Bװ�ü���������A��B֮������װ��ˮ��ϴ��ƿװ�ã��Ľ��������NaNO2�Ĵ��ȣ�
ʵ���ⶨ��ȡ����Ʒ��NaNO2�ĺ���
���裺a����5���б�ŵĴ��̶��Թܣ���ɫ�ܣ��зֱ���벻ͬ����NaNO2��Һ��������1mL��M��Һ��M��NaNO2���Ϻ�ɫ��NaNO2Ũ��Խ����ɫԽ����ټ�����ˮ���������Ϊ10mL�����Ƴɱ�ɫ�ף�
b������0.10g�Ƶõ���Ʒ����ˮ���500mL��Һ����ȡ5mL����Һ������1mL M��Һ���������ˮ��10mL�����ɫ�ױȽϣ�
| �Թܱ�� | �� | �� | �� | �� | �� |
| NaNO2����/mg•L-1 | 0 | 20 | 40 | 60 | 80 |
��5����Ŀ�ӱ�ɫ��֤��ά����C������Ч����NaNO2�ĺ�������Ʋ��������ʵ�鱨�森
| ʵ�鷽�� | ʵ������ | ʵ����� |
| ȡ5mL ����Һ������һ������ά����C���� �ټ���1mL M��Һ������������ˮ10mL������ ��ܺ�ɫ�Աȣ� | �Ϻ�ɫ�Ȣ�ɫ��dz | ά����C ������Ч����NaNO2�ĺ����� |
 �����£���20mL 0.2mol��L H2A��Һ�еμ�0.2mol/L NaOH��Һ���й��������ʵ����仯��ͼ������1����H2A��II����HA-��III����A2-��������ͼʾ�жϣ�����˵����ȷ�ģ�������
�����£���20mL 0.2mol��L H2A��Һ�еμ�0.2mol/L NaOH��Һ���й��������ʵ����仯��ͼ������1����H2A��II����HA-��III����A2-��������ͼʾ�жϣ�����˵����ȷ�ģ�������| A�� | ��ʹNaHA��Һ�����ԣ����������м������� | |
| B�� | ��NaHA��Һ����ˮ�Ĺ����У�pH��������Ҳ���ܼ�С | |
| C�� | ��V��NaOH��=20mLʱ����Һ������Ũ�ȴ�С��ϵ��c��Na+����c��HA-����c��H+����c��A2-����c��OH-�� | |
| D�� | �������Ũ�ȵ�NaOH��Һ��H2A��Һ��Ϻ�����Һ��ˮ�ĵ���̶ȱȴ�ˮ�� |
 ��
��
 H2O2+
H2O2+ 6H+=
6H+= Mn2++
Mn2++ O2��+
O2��+ 8H2O��
8H2O��