��Ŀ����
2����NaOH��MgCl2��AlCl3���ֹ�����ɵĻ������������ˮ����1.16g��ɫ����������������Һ����μ���1.00mol/L HCl��Һ������HCl��Һ����������ɳ����Ĺ�ϵ��ͼ��ʾ��
��1��A��ij�����Ļ�ѧʽΪMg��OH��2��
��2��д��A����B�㷢����Ӧ�����ӷ���ʽH2O+AlO2-+H+=Al��OH��3����
��3��ԭ�������MgCl2��������1.9g��AlCl3��������2.67g��C��HCl��Һ�����Ϊ130mL��NaOH��������5.2g��
���� ��ˮ�й�����ɵĻ������Һ�м�������ʱ��0-Aʱû�г������ɣ�˵����Һ��NaOH����������������ȫת��Ϊƫ��������ӣ���Һ�д��ڵij���ΪMg��OH��2��
A-B�Σ������ƫ�����Ʒ�Ӧ���������������������ӷ�Ӧ����ʽΪH2O+AlO2-+H+=Al��OH��3����B����Һ�д��ڵ��������Ȼ��ƣ�����ΪMg��OH��2��Al��OH��3��
B-C�����У�������þ�����������������ᷴӦ�����Ȼ������Ȼ�þ������C����Һ�е��������Ȼ������Ȼ�þ���Ȼ��ƣ�
��� �⣺��1����ˮ�й�����ɵĻ������Һ�м�������ʱ��0-Aʱû�г������ɣ�˵����Һ��NaOH��������Һ����������ȫת��Ϊƫ��������ӣ���Һ�д��ڵij���ΪMg��OH��2���ʴ𰸣�Mg��OH��2��
��2��A-B�Σ������ƫ�����Ʒ�Ӧ���������������������ӷ�Ӧ����ʽΪH2O+AlO2-+H+=Al��OH��3�����ʴ𰸣�H2O+AlO2-+H+=Al��OH��3����
��3��A���������������þ����n��Mg��OH��2��=$\frac{1.16g}{58g/mol}$=0.02mol������Mgԭ���غ��n��MgCl2��=n��Mg��OH��2��=0.02mol���Ȼ�þ������=0.02mol��95g/mol=1.9g��A-B�Σ������ƫ�����Ʒ�Ӧ���������������������ӷ�Ӧ����ʽΪH2O+AlO2-+H+=Al��OH��3��������HCl�����֪��n��AlO2-��=n��HCl��=1mol/L����0.03-0.01��L=0.02mol���Ȼ���������=0.02mol��133.5g/mol=2.67g��������ԭ���غ��n��AlCl��=n��AlO2-��=0.02mol����Na+���Ӻ�Cl-�����غ�ã�ԭ�������n��NaOH��=n��NaCl��=n��Cl-��=2n��MgCl2��+3n��AlCl3��+n��HCl��=0.02mol��2+0.02mol��3+0.03L��1mol/L=0.13mol�������Ƶ�����=0.13mol��40g/mol=5.2g��
C����ҺΪMgCl2��AlCl3��NaCl���Һ��C���������������к�ԭ������е�NaOH����ʱ�����������ʵ�����
n��HCl��=n��NaOH��=0.13mol��P������ʾ��������Ϊ��V=$\frac{0.13mol}{1mol/L}$=0.13L=130ml�ʴ�Ϊ��1.9��2.67��130��5.2��
���� ����ͼ�������Ľ�����ɣ�Ҫ��ȷÿһ��ͼ�����Ļ�ѧ��Ӧ��֪���յ�����ĺ��弰��Һ�����ʵijɷ֣���Ϸ���ʽ���й�������з�����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���Pb+2Cl2�TPbCl4��Sn+2Cl2�TSnCl4��SnCl2+Cl2�TSnCl4
��PbO2+4HCl�TPbCl4+2H2O��Pb3O4+8HCl�T3PbCl2+Cl2��+4H2O��
| A�� | �ڢۢ� | B�� | �ڢۢ� | C�� | �٢ڢ� | D�� | �٢ڢܢ� |
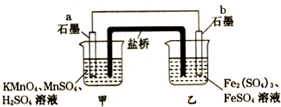 �����ͼԭ��أ�2KMnO4+10FeSO4+8H2SO4�T2MnSO4+5Fe2��SO4��3+K2SO4+8H2O����������װ�б�����Һ������˵����ȷ���ǣ�������
�����ͼԭ��أ�2KMnO4+10FeSO4+8H2SO4�T2MnSO4+5Fe2��SO4��3+K2SO4+8H2O����������װ�б�����Һ������˵����ȷ���ǣ�������| A�� | ���·���ӵ������Ǵ�a��b | |
| B�� | ��ع���ʱ�������е�SO42-������ձ� | |
| C�� | a�缫�Ϸ����ķ�ӦΪ��MnO4-+8H++5e-�TMn2++4H2O | |
| D�� | ˫Һԭ��ع���ʱ�������������� |
 BHT��һ�ֳ��õ�ʳƷ������������
BHT��һ�ֳ��õ�ʳƷ������������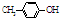 �����ϳ�BHT�ķ�������ͼ���֣�����˵���������
�����ϳ�BHT�ķ�������ͼ���֣�����˵�����������BHT����������
| A�� | BHT�� ����ʹ����KMnO4��ɫ ����ʹ����KMnO4��ɫ | |
| B�� | BHT��ˮ�е��ܽ��С�ڱ��� | |
| C�� | ���ַ����ķ�Ӧ���Ͷ��Ǽӳɷ�Ӧ | |
| D�� | ����ɫ��ѧ�Ƕȷ�������һ���ڷ����� |
��1���ϳɰ��õ��������Լ���Ϊԭ���Ƶã�CH4��g��+H2O��g��?CO��g��+3H2��g�����йػ�ѧ��Ӧ�������仯��ͼ��ʾ��CH4��g����H2O��g����Ӧ����CO��g����H2��g�����Ȼ�ѧ����ʽΪCH4��g��+H2O��g��=CO��g��+3H2 ��g����H=+161.1kJ•mol-1��
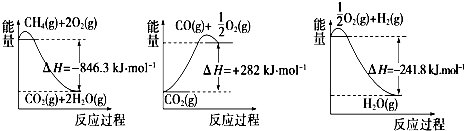
��2��CO�Ժϳɰ��Ĵ����ж������ã��������������ͭ������Һ������ԭ������CO���䷴Ӧԭ��Ϊ[Cu��NH3��2CH3COO]��l��+CO��g��+NH3��g��?[Cu��NH3��3]CH3COO•CO��l����H��0������CO�������ͭ��Һ�����ʵ��������ֿ��������ָ�������CO�������Թ�ѭ��ʹ�ã�����������������B��������ĸ��
A�����¡���ѹ B�����¡���ѹ C�����¡���ѹ D�����¡���ѹ
��3���ð�����ȡ����[CO��NH2��2]�ķ�ӦΪ2NH3��g��+CO2��g��?CO��NH2��2��l��+H2O��g����H��0��ij�¶��£����ݻ�Ϊ100L���ܱ�������ͨ��4molNH3��2molCO2���÷�Ӧ���е�40sʱ�ﵽƽ�⣬��ʱCO2��ת����Ϊ50%�����¶��´˷�Ӧƽ�ⳣ��K��ֵΪ2500 L2•mol-2��
��4��������ʩ����������ͨ��ת��Ϊ̼��炙�̼����狀�ű����������ã����ط�����������ڵ���ø�����£�ת��Ϊ̼��泥���֪���������ˮ�еĵ���ƽ�ⳣ����25�棩��������г�����0.1mol•L-1�ģ�NH4��2CO3��Һ��
| ������� | H2CO3 | NH3•H2O |
| Ka1 | 4.30��10-7 | 1.77��10-5 |
| Ka2 | 5.61��10-11 |
�ھ���Һ������֮�������й�ϵʽ������Ϊ���в���ȷ����B��
A��c��NH4+����c��CO32-����c��HCO3-����c��NH3•H3O��
B��c��NH4+��+c��H+��=c��HCO3-��+c��OH-��+c��CO32-��
C��c��CO32-��+c��HCO3-��+c��H2CO3��=0.1mol•L-1
D��c��NH4+��+c��NH3•H2O��=2c��CO32-��+2c��HCO3-��+2c��H2CO3��
 Ϊ��֤��һˮ�ϰ���NH3•H2O����������ʣ��ס��Ҷ��˷ֱ�ѡ�������Լ�����ʵ�飺��ˮ��0.10mol•L-1NH4Cl��Һ��NH4Cl���塢��̪��Һ��pH��ֽ������ˮ��
Ϊ��֤��һˮ�ϰ���NH3•H2O����������ʣ��ס��Ҷ��˷ֱ�ѡ�������Լ�����ʵ�飺��ˮ��0.10mol•L-1NH4Cl��Һ��NH4Cl���塢��̪��Һ��pH��ֽ������ˮ��
