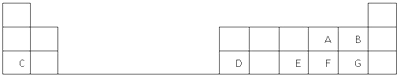
��Ŀ����
��ͼ��Ԫ�����ڱ���һ���֣���Ա��еĢ١���Ԫ�أ���д���пո�
��1������9��Ԫ���У��ǽ�������ǿ���� ����Ԫ�ط��ţ�����ѧ��������õ�Ԫ���� ���Ԫ�ط��š�����
��2��Ԫ�آݵ�ԭ�ӽṹʾ��ͼΪ ��Ԫ�آٵ����������ĵ���ʽΪ�� ��
��3��Ԫ�آ���ߵ���̬�⻯���н��ȶ��������� ���ѧʽ����
��4������ЩԪ�ص�����������Ӧˮ�����У�������ǿ���� ���ѧʽ����������ǿ���� ���ѧʽ����
��5��Ԫ�آ��������Ӱ뾶�ϴ���� �������ӷ��ţ���
| �� | �� | �� | |||||||||||||||
| �� | �� | �� | �� | �� | �� | ||||||||||||
��2��Ԫ�آݵ�ԭ�ӽṹʾ��ͼΪ
��3��Ԫ�آ���ߵ���̬�⻯���н��ȶ���������
��4������ЩԪ�ص�����������Ӧˮ�����У�������ǿ����
��5��Ԫ�آ��������Ӱ뾶�ϴ����
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ã�֪��ΪC����ΪO����ΪF����ΪNa����ΪAl����ΪSi����ΪS����ΪCl����ΪAr��
��1��ͬ����������ҷǽ�������ǿ��ͬ�������϶��·ǽ����Լ�����ϡ�����廯ѧ��������ã�
��2��Alԭ�Ӻ��������Ϊ13����3�����Ӳ㣬���������Ϊ2��8��3��̼�����������Ϊ������̼��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ�
��3���ǽ�����Խǿ���⻯��Խ�ȶ���
��4��FԪ��û����ۺ����ᣬ�ʸ������������ǿ������Ԫ����Na�Ľ�������ǿ�����������Ƽ�����ǿ��
��5�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС��
��1��ͬ����������ҷǽ�������ǿ��ͬ�������϶��·ǽ����Լ�����ϡ�����廯ѧ��������ã�
��2��Alԭ�Ӻ��������Ϊ13����3�����Ӳ㣬���������Ϊ2��8��3��̼�����������Ϊ������̼��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ�
��3���ǽ�����Խǿ���⻯��Խ�ȶ���
��4��FԪ��û����ۺ����ᣬ�ʸ������������ǿ������Ԫ����Na�Ľ�������ǿ�����������Ƽ�����ǿ��
��5�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС��
���
�⣺��Ԫ�������ڱ���λ�ã�֪��ΪC����ΪO����ΪF����ΪNa����ΪAl����ΪSi����ΪS����ΪCl����ΪAr��
��1��ͬ����������ҷǽ�������ǿ��ͬ�������϶��·ǽ����Լ�������FԪ�ؽ�������ǿ��ϡ������Ar�Ļ�ѧ��������ã��ʴ�Ϊ��F��Ar��
��2��Alԭ�Ӻ��������Ϊ13����3�����Ӳ㣬���������Ϊ2��8��3��ԭ�ӽṹʾ��ͼΪ��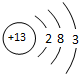 ��̼�����������Ϊ������̼��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ������ʽΪ��
��̼�����������Ϊ������̼��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ������ʽΪ��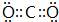 ���ʴ�Ϊ��
���ʴ�Ϊ��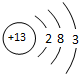 ��
��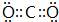 ��
��
��3��ͬ�������϶��·ǽ����Լ������ǽ�����Խǿ���⻯��Խ�ȶ������⻯���ȶ���H2O��H2S���ʴ�Ϊ��H2O��
��4��FԪ��û����ۺ����ᣬ��HClO4��������ǿ������Ԫ����Na�Ľ�������ǿ����NaOH������ǿ���ʴ�Ϊ��HClO4��NaOH��
��5��S2-��Cl-���Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�������Ӱ뾶ΪS2-��Cl-���ʴ�Ϊ��S2-��
��1��ͬ����������ҷǽ�������ǿ��ͬ�������϶��·ǽ����Լ�������FԪ�ؽ�������ǿ��ϡ������Ar�Ļ�ѧ��������ã��ʴ�Ϊ��F��Ar��
��2��Alԭ�Ӻ��������Ϊ13����3�����Ӳ㣬���������Ϊ2��8��3��ԭ�ӽṹʾ��ͼΪ��
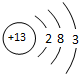 ��̼�����������Ϊ������̼��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ������ʽΪ��
��̼�����������Ϊ������̼��������Cԭ����Oԭ��֮���γ�2�Թ��õ��Ӷԣ������ʽΪ��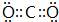 ���ʴ�Ϊ��
���ʴ�Ϊ��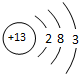 ��
��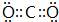 ��
����3��ͬ�������϶��·ǽ����Լ������ǽ�����Խǿ���⻯��Խ�ȶ������⻯���ȶ���H2O��H2S���ʴ�Ϊ��H2O��
��4��FԪ��û����ۺ����ᣬ��HClO4��������ǿ������Ԫ����Na�Ľ�������ǿ����NaOH������ǿ���ʴ�Ϊ��HClO4��NaOH��
��5��S2-��Cl-���Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�������Ӱ뾶ΪS2-��Cl-���ʴ�Ϊ��S2-��
���������⿼��Ԫ�����ڱ���Ԫ���������ۺ�Ӧ�ã��ѶȲ����ضԻ���֪ʶ�Ĺ��̣�ע���������ȽϵıȽϣ�

��ϰ��ϵ�д�
 ���ſ����ϵ�д�
���ſ����ϵ�д� ���Ŀ����ϵ�д�
���Ŀ����ϵ�д� ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д�
�����Ŀ
�����й��Ȼ�ѧ����ʽ��������ȷ���ǣ�������
| A����ϡ��Һ�У�H+��aq��+OH-��aq���TH2O��l����H=-57.3kJ/mol��������0.6mol H2SO4��ϡ�����뺬1mol NaOH����Һ��ϣ��ų�����������57.3kJ |
| B����֪C��ʯī��s���TC�����ʯ��s����H��0������ʯ��ʯī�ȶ� |
| C����Ҫ���ȵķ�Ӧ˵���������ȷ�Ӧ |
| D����֪2C��s��+2O2��g���T2CO2��g����H1��2C��s��+O2��g���T2CO��g����H2�����H1����H2 |

 ����
���� ����CH3CH2Cl����CCl4����CH��CH����
����CH3CH2Cl����CCl4����CH��CH���� �� ��CH3CH2OH��
�� ��CH3CH2OH��