��Ŀ����
6��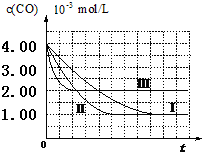 ��I��Ŀǰ���������̳����ô�������β���е�NO��COת����CO2��N2����ѧ����ʽ���£�2NO+2CO$\stackrel{����}{?}$2CO2+N2��Ϊ�о������߸�ת�����̷�Ӧ���ʣ�ij���������������ʵ��̽����
��I��Ŀǰ���������̳����ô�������β���е�NO��COת����CO2��N2����ѧ����ʽ���£�2NO+2CO$\stackrel{����}{?}$2CO2+N2��Ϊ�о������߸�ת�����̷�Ӧ���ʣ�ij���������������ʵ��̽���������ϲ��ġ�
�ٲ�ͬ�Ĵ�����ͬһ��Ӧ�Ĵ�Ч�ʲ�ͬ��
��ʹ����ͬ�Ĵ������������������ʱ�������ıȱ�����Դ�Ч����Ӱ�죮
��ʵ����ơ�������Ϊ̽��ijЩ�������������β��ת����Ӧ���ʵ�Ӱ����ɣ���������¶Ա�ʵ�飮
��1���������ʵ����Ʊ������в�Ҫ���ո�
| ʵ���� | ʵ��Ŀ�� | T/�� | NO��ʼŨ�� mol/L | CO��ʼŨ�� mol/L | ͬ�ִ����ıȱ����m2/g |
| �� | Ϊ����ʵ�������� | 280 | 6.50��10-3 | 4.00��10-3 | 80 |
| �� | ̽�������ȱ������β��ת�����ʵ�Ӱ�� | 6.50��10-3 | 4.00��10-3 | 120 | |
| �� | ̽���¶ȶ�β��ת�����ʵ�Ӱ�� | 360 | 6.50��10-3 | 4.00��10-3 | 80 |
��2������ڢ���ʵ���У���ƽ��ʱNO��Ũ��Ϊ3.5��10-3mol/L��
��3�������ߢ��֪����������ȱ����������β��ת�������������������С��������Ӱ�족����
��II������������ʳ��������������ũҵ�Ի�ѧ���ϵ�������Խ��Խ�����е���������������һ�ֻ��ʣ������ĺϳ�Ϊ���ʵ�������ҵ�춨�˻�������ԭ��Ϊ��N2+3H2?2NH3
��1����N2+3H2?2NH3�ķ�Ӧ�У�һ��ʱ���NH3��Ũ��������0.9mol•L-1����N2��ʾ�䷴Ӧ����Ϊ0.15mol•L-1•s-1������������ʱ��ΪB��
A��2s B��3s C��4s D��6s
��2������4���������ڲ�ͬ�����²�õĺϳɰ���Ӧ�����ʣ����з�Ӧ������D��
A��v��H2��=0.1mol•L-1•min-1 B��v��N2��=0.1mol•L-1•min-1
C��v��NH3��=0.15mol•L-1•min-1 D��v��N2��=0.002mol•L-1•s-1��
���� ��I����1��ʵ��Ŀ���ǣ�̽��ijЩ�������������β��ת����Ӧ���ʵ�Ӱ����ɣ�Ӧ��ȡ���Ʊ����������о���ʵ��I��������ȱ������ͬ������������ͬ��
��2���ɷ���ʽ��֪��c��CO��=��c��NO����ƽ��ʱNOŨ��=NO��ʼŨ��-NOŨ�ȱ仯����
��3�������ߢ��֪����������ȱ���������̵���ƽ���ʱ�䣬����β��ת����������
��II����1����������֮�ȵ��ڻ�ѧ������֮�ȣ�����N2��ʾ��ƽ����Ӧ����v��N2����������v��NH3�����ٸ������ʶ�����㷴Ӧʱ�䣻
��2��ͬһ��ѧ��Ӧ�У�ͬһʱ����ڣ������ʵķ�Ӧ����֮�ȵ����������֮�ȣ��ȰѲ�ͬ���ʵķ�Ӧ���ʻ����ͬһ���ʵķ�Ӧ���ʽ��бȽϣ�ע�ⵥλ����һ�£�
��� �⣺��I����1��ʵ��Ŀ���ǣ�̽��ijЩ�������������β��ת����Ӧ���ʵ�Ӱ����ɣ�Ӧ��ȡ���Ʊ����������о���ʵ��I��������ȱ������ͬ����������Ҫ��ͬ����̽�������ȱ������β��ת�����ʵ�Ӱ�죬�ʢ��е��¶�Ϊ280�棬
�ʴ�Ϊ��
| ʵ���� | ʵ��Ŀ�� | T/�� | NO��ʼŨ�� mol/L | CO��ʼŨ�� mol/L | ͬ�ִ����ıȱ����m2/g |
| �� | |||||
| �� | 280 | ||||
| �� |
�ʴ�Ϊ��3.5��10-3mol/L��
��3�������ߢ��֪����������ȱ���������̵���ƽ���ʱ�䣬����β��ת����������
�ʴ�Ϊ������
��II����1������v��N2��=0.15mol/��L•s��������֮�ȵ��ڻ�ѧ������֮�ȣ�����v��NH3��=2v��N2��=2��0.15mol/��L•s��=0.3mol/��L•s�������Է�Ӧ��������ʱ��Ϊ 0.9mol/L��0.3mol/��L•s��=3s��
�ʴ�Ϊ��B��
��2����ӦΪN2��g��+3H2��g��?2NH3��g�����������ķ�Ӧ����Ϊ�������жϣ�
A��v��H2��=0.1mol•L-1•min-1��
B��v��N2��=0.1mol•L-1•min-1����Ӧ����֮�ȵ����������֮�ȣ�����v��H2��=3v��N2��=0.3mol•L-1•min-1��
C��v��NH3��=0.15mol•L-1•min-1����Ӧ����֮�ȵ����������֮�ȣ�����v��H2��=1.5v��NH3��=0.225mol/��L•min����
D��v��N2��=0.002mol•L-1•s-1=0.12mol•L-1•min-1����Ӧ����֮�ȵ����������֮�ȣ�����v��H2��=3v��N2��=0.36mol/��L•min�������Է�Ӧ����������D��
��ѡD��
���� ���⿼�黯ѧƽ����㡢��������Է�Ӧ���ʵ�Ӱ�졢��ѧ��Ӧ���ʼ��㡢��Ӧ���ʿ����Ƚϵȣ�I��ע�����ÿ��Ʊ�����������𣬷�Ӧ���ʿ����Ƚϻ������������仯ѧ�������ı�ֵ��С�жϣ���ֵԽ��ʾ��Ӧ����Խ�죬�Ѷ��еȣ�

| A�� | ��ѹ | B�� | ���� | C�� | ��ѹ | D�� | ����B��Ũ�� |
��1������ʵ���з�����Ӧ�Ļ�ѧ����ʽ��Zn+CuSO4=ZnSO4+Cu��Zn+H2SO4=ZnSO4+H2����
��2������ͭ��Һ���Լӿ������������ʵ�ԭ����CuSO4 ��Zn��Ӧ������ Cu ��Zn�γ�ͭпԭ��أ��ӿ����������������ʣ�
��3��ʵ����������Na2SO4��MgSO4��AgNO3��K2SO4��4����Һ����������ʵ����CuSO4��Һ���������õ���AgNO3��
��4��Ҫ�ӿ�����ʵ����������������ʣ����ɲ�ȡ�Ĵ�ʩ�����߷�Ӧ�¶ȣ��ʵ����������Ũ�ȡ�����п���ı������ ���δ�һ�֣���
��5��Ϊ�˽�һ���о�����ͭ�����������������ʵ�Ӱ�죬��ͬѧ���������һϵ�е�ʵ�飮�����������Ļ����Һ�ֱ���뵽6��ʢ�й���Zn���ķ�Ӧƿ�У��ռ����������壬��¼�����ͬ�������������ʱ�䣮
| ��ʵ�� �����Һ | A | B | C | D | E | F |
| 4mol•L-1 H2SO4/mL | 30 | V1 | 30 | 30 | 30 | 30 |
| ����CuSO4��Һ/mL | 0 | 0.5 | 2.5 | 5 | V6 | 20 |
| H2O/mL | 20 | V8 | V9 | V10 | 10 | 0 |
�ڸ�ͬѧ���ó��Ľ���Ϊ������������CuSO4��Һʱ���������������ʻ�����ߣ����������CuSO4��Һ����һ����ʱ���������������ʷ������½���������������������½�����Ҫԭ����һ����������ͭ�����ɵĵ���ͭ�������п�ı��棬������п����Һ�ĽӴ������
��1��������һ������Ľྻȼ�ϣ���֪��
CH4��g��+2O2��g���TCO2��g��+2H2O��g������H=-802.3kJ•mol-1
H2O��1���TH2O��g������H=+44.0kJ•mol-l
��4.8g����������ȫȼ������Һ̬ˮ���ų�������Ϊ267.1kJ��
��2�����ü�����ˮ��Ӧ�Ʊ���������ԭ�����ۣ������ƹ��ֵ��
�÷�ӦΪCH4��g��+H2O��g��?CO��g��+3H2��g������H=+206.1kJ•mol-l��
����800��ʱ����Ӧ�Ļ�ѧƽ�ⳣ��K=l.0��ijʱ�̲�ø��¶����ܱ������и����ʵ����ʵ���Ũ�����±���
| CH4 ��g�� | H2O ��g�� | CO ��g�� | H2 ��g�� |
| 3.0mol•L-1 | 8.5mol•L-1 | 2.0mol•L-1 | 2.0mol•L-1 |
a��v��������v���棩 b��v��������v���棩
c��v������=v���棩 d�����ж�
��Ϊ��̽���¶ȡ�ѹǿ��������ѧ��Ӧ���ʵ�Ӱ�죬ijѧϰС���������������Ա�ʵ�飨�¶�Ϊ360���480�桢ѹǿΪ101kPa��303kPa������ʵ���������±�����
| ʵ����� | �¶�/�� | ѹǿ/kPa | V��CH4��/mol•L-1•s-1 | V��H2O��/mol•L-1•s-1 |
| 1 | 360 | P1 | 0.100 | 0.100 |
| 2 | 480 | 101 | 0.120 | 0.120 |
| 3 | 360 | P2 | 0.080 | 0.080 |
ʵ��2��3��Ŀ����̽���¶ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죮
ʵ��1��2��3�з�Ӧ�Ļ�ѧƽ�ⳣ���Ĵ�С��ϵ��K1=K3��K2 ����K1��K2��K3��ʾ����
| A�� | ±���ǵ��͵ķǽ���Ԫ�أ���˲����������ǽ���Ԫ�ػ��� | |
| B�� | ±�ص���Խ���ã����ۡ��е��Խ�� | |
| C�� | ±�ص��ʶ��ܺ�ˮ���ҷ�Ӧ | |
| D�� | ±�ص��ʶ��ܺ�H2��Ӧ������̬�⻯����ȶ����浥�������Ե���ǿ����ǿ |
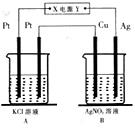 ��ͼ��ʾ�������µ��5min��ͭ�缫��������1��08g���Իش�
��ͼ��ʾ�������µ��5min��ͭ�缫��������1��08g���Իش�