��Ŀ����
�ڱ�״���½��мס��ҡ�������ʵ�飮����ʵ�����ȡ30mLͬŨ�ȵ����ᣬ����ͬһ��þ���Ͻ��ĩ���������壬�й����ݼ��±���
��1���ס�������ʵ���У���һ�������Dz������ģ� ����ס����ҡ�����
��2��Ҫ�����������ʵ���Ũ�ȣ����п���Ϊ�������ݵ������� ����õ���������ʵ���Ũ��Ϊ ��
��3����Ͻ���Mg��Al�����ʵ���֮�ȣ����п���Ϊ�������ݵ������� ����õ�Mg��Al���ʵ���֮��Ϊ ��
| ʵ����� | �� | �� | �� |
| �Ͻ�����/mg | 255 | 385 | 459 |
| �����������/mL | 280 | 336 | 336 |
��2��Ҫ�����������ʵ���Ũ�ȣ����п���Ϊ�������ݵ�������
��3����Ͻ���Mg��Al�����ʵ���֮�ȣ����п���Ϊ�������ݵ�������
���㣺�йػ���ﷴӦ�ļ���
ר�⣺������
��������1������Ũ�ȡ����һ�������кϽ�����С�����кϽ��������Ҽ��������������С���������������˵���������������������ȫ��Ӧ�����ݼ��н�����������������ȹ�ϵ��������336mL������Ҫ������������ȷ����������������Ƿ�ǡ�÷�Ӧ��
��2���ҡ�����������ȫ�����Ը��ݷ�Ӧ�����������������������ʵ���Ũ�ȣ�����n=
�������������ʵ�����������Ԫ���غ��֪n��HCl��=2n��H2�����ݴ˼��㣻
��3������������ʣ�࣬������ȫ��Ӧ����ʱ��������280mL���ʿ��Ը��ݼ������ݼ�����������ʵ���֮�ȣ���þ���������ʵ����ֱ�Ϊxmol��ymol�����ݶ�������֮�������ת���غ��з��̼���x��y��ֵ���ݴ˽��
��2���ҡ�����������ȫ�����Ը��ݷ�Ӧ�����������������������ʵ���Ũ�ȣ�����n=
| V |
| Vm |
��3������������ʣ�࣬������ȫ��Ӧ����ʱ��������280mL���ʿ��Ը��ݼ������ݼ�����������ʵ���֮�ȣ���þ���������ʵ����ֱ�Ϊxmol��ymol�����ݶ�������֮�������ת���غ��з��̼���x��y��ֵ���ݴ˽��
���
�⣺��1������Ũ�����һ�������кϽ�����С�����кϽ��������Ҽ��������������С���������������˵���������������������ȫ��Ӧ�����кϽ�����С�ڱ��кϽ����������ҡ����������������ȣ�˵���ҡ�����������ȫ��Ӧ��
���ݼ�֪������336mL������Ҫ����������Ϊ��255mg��
=306mg�������н�����ʣ�࣬��������㣬
�ʴ�Ϊ���ң�
��2���ҡ�����������ȫ��Ӧ�����Ը��ݷ�Ӧ�����������������������ʵ���Ũ�ȣ�������ȫ��Ӧ��������336mL�����������ʵ���Ϊ��
=0.015mol��������Ԫ���غ��֪��n��HCl��=2n��H2��=2��0.015mol=0.03mol������������ʵ���Ũ��Ϊ��
=1mol/L��
�ʴ�Ϊ���һ����1mol/L��
��3������������ʣ�࣬������ȫ��Ӧ����ʱ��������280mL���ʿ��Ը��ݼ������ݼ�����������ʵ���֮�ȣ���þ���������ʵ����ֱ�Ϊxmol��ymol�����ݶ���������֪����24x+27y=0.255�����ݵ���ת���غ��У���2x+3y=
��2��
���ݢ٢�������ã�x=0.005��y=0.005��
���ԺϽ���þ���������ʵ���֮��Ϊ��0.005mol��0.005mol=1��1��
�ʴ�Ϊ���ף�1��1��
���ݼ�֪������336mL������Ҫ����������Ϊ��255mg��
| 336mL |
| 280mL |
�ʴ�Ϊ���ң�
��2���ҡ�����������ȫ��Ӧ�����Ը��ݷ�Ӧ�����������������������ʵ���Ũ�ȣ�������ȫ��Ӧ��������336mL�����������ʵ���Ϊ��
| 0.336L |
| 22.4L/mol |
| 0.03mol |
| 0.03L |
�ʴ�Ϊ���һ����1mol/L��
��3������������ʣ�࣬������ȫ��Ӧ����ʱ��������280mL���ʿ��Ը��ݼ������ݼ�����������ʵ���֮�ȣ���þ���������ʵ����ֱ�Ϊxmol��ymol�����ݶ���������֪����24x+27y=0.255�����ݵ���ת���غ��У���2x+3y=
| 0.28L |
| 22.4L/mol |
���ݢ٢�������ã�x=0.005��y=0.005��
���ԺϽ���þ���������ʵ���֮��Ϊ��0.005mol��0.005mol=1��1��
�ʴ�Ϊ���ף�1��1��
���������⿼�����ﷴӦ�ļ��㣬��Ŀ�Ѷ��еȣ����ݱ������ݹ�ϵ�жϷ�Ӧ�Ĺ��������ǹؼ���ע�������йػ���ﷴӦ�ļ��㷽��������������ѧ���ķ�����������������ѧ����������

��ϰ��ϵ�д�
�����Ŀ
���й��ڽ����˵������ȷ���ǣ�������
| A��������۲����� |
| B�����岻��ͨ����ֽ |
| C���������Ӳ�ͣ���������˶� |
| D�����岻�ȶ�����ֹ�������γɳ��� |
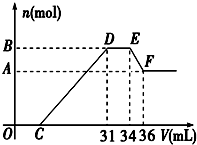 ��֪�������������������ԭ��Ӧ��ʱ��һ������Ũ��Խϡ����Ӧ�Ļ�ԭ�����е��Ļ��ϼ�Խ�ͣ�����һ�������������Ͻ���һ����ϡHNO3��ַ�Ӧ����Ӧ���������κ�����ų����ڷ�Ӧ���������Һ�У���μ���4mol/L NaOH��Һ������NaOH��Һ�������V����������������ʵ�����n����ϵ��ͼ��ʾ����
��֪�������������������ԭ��Ӧ��ʱ��һ������Ũ��Խϡ����Ӧ�Ļ�ԭ�����е��Ļ��ϼ�Խ�ͣ�����һ�������������Ͻ���һ����ϡHNO3��ַ�Ӧ����Ӧ���������κ�����ų����ڷ�Ӧ���������Һ�У���μ���4mol/L NaOH��Һ������NaOH��Һ�������V����������������ʵ�����n����ϵ��ͼ��ʾ���� ijУ�о���ѧϰС���ͬѧѧϰ�굪���й����ʵ�����֮�Ե�Ԫ�ص��⻯�������������ʽ����˸�������о���
ijУ�о���ѧϰС���ͬѧѧϰ�굪���й����ʵ�����֮�Ե�Ԫ�ص��⻯�������������ʽ����˸�������о���