��Ŀ����
17����ҵ�����÷�̼�����Ҫ�ɷ�CeCO3F����ȡCeCl3��һ�ֹ����������£�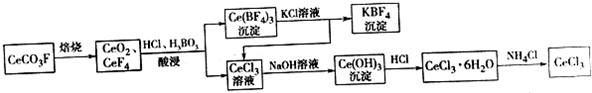
��ش��������⣺
��1��CeCO3F�У�CeԪ�صĻ��ϼ�Ϊ+3��
��2�������������ϡ�����H2O2�滻HCl������ɻ�����Ⱦ��д��ϡ���ᡢH2O2��CeO2��Ӧ�����ӷ���ʽ��H2O2+2CeO2+6H+=2Ce3++4H2O+O2����
��3����Ce��BF4��3�м���KCl��Һ��Ŀ���DZ�����������Ce��BF4��3��������ʽ��ʧ���ȥBF4-�����CeCl3�IJ��ʣ�
��4����Һ�е�C��Ce3+������1��10-5mol��l-1������ΪCe3+������ȫ����ʱ��Һ��PHΪ9������֪KSP[Ce��OH��3]=1��10-20��
��5������CeCl3.6H2O��NH4Cl�Ĺ�������ɵõ���ˮCeCl3������NH4Cl��������NH4Cl�������ȷֽ����HCl������CeCl3ˮ�⣮
��6��ȷ��ȡ0.7500gCeCl3��Ʒ������ƿ�У������������������Һ��Ce3+����ΪCe4+��Ȼ����0.1000mol��l-1��NH4��2Fe��SO4��2����Һ�ζ����յ㣬����25.00ml����Һ������֪��Fe2+Ce4+=Ce3++Fe3+��
�ٸ���Ʒ��CeCl3����������Ϊ82.2%��
����ʹ�þ��õģ�NH4��2Fe��SO4��2����Һ���еζ�����ø�CeCl3��Ʒ����������ƫ���ƫ����ƫС������Ӱ�족��
���� ���÷�̼�����Ҫ�ɷ�CeCO3F����ȡCeCl3��һ�ֹ�������Ϊ��CeCO3F���յ�CeO2��CeF4���������������������˵ó���Ce��BF��4��CeCl3��Һ��Ce��BF��4���Ȼ�����Һ��Ӧ�õ�CeCl3��KBF��CeCl3��ǰ����Һ��CeCl3�ĺϲ��ټ��������ƣ���Ce��OH��3������Ce��OH��3���������ᷴӦ�ɵ�CeCl3•6H2O������CeCl3•6H2O��NH4Cl�Ĺ�������ɵõ���ˮCeCl3��
��1�����ݻ��ϼ۴�����Ϊ0�жϣ�
��2��������������H2O2�ܻ�ԭCeO2����Ce3+������Ԫ���غ�͵���غ���д���ӷ���ʽ��
��3����Ce��BF4��3�м���KCl��Һ����CeCl3��KBF����ֹ����Ce��BF4��3������
��4����Һ�е�C��Ce3+������1��10-5mol•L-1������ΪCe3+������ȫ������KSP[Ce��OH��3]=C��Ce3+��•C3��OH-�� �ɼ������Һ�����������ӵ�Ũ�ȣ�����ȷ��PHֵ��
��5��NH4Cl�������ȷֽ����HCl����������CeCl3ˮ�⣬�ݴ˴��⣻
��6����0.1000mol•l-1��NH4��2Fe��SO4��2����Һ���25.00ml�������������ӵ����ʵ���Ϊ2.5��10-3mol�����ݷ�ӦFe2++Ce4+�TCe3++Fe3+������CeԪ���غ��֪���ɼ����CeCl3������������ȷ����Ʒ��CeCl3������������
����ʹ�þ��õģ�NH4��2Fe��SO4��2����Һ���еζ��������������ӱ��������������������Ի����ı�Һ�����Ϊƫ�ݴ˷�����
��� �⣺��1�����ݻ��ϼ۴�����Ϊ0��֪��CeCO3F�У�CeԪ�صĻ��ϼ�Ϊ+3�ۣ�
�ʴ�Ϊ��+3��
��2��ϡ���ᡢH2O2��CeO2��Ӧ�����ӷ���ʽΪ��H2O2+2CeO2+6H+=2Ce3++4H2O+O2����
�ʴ�Ϊ��H2O2+2CeO2+6H+=2Ce3++4H2O+O2����
��3����Ce��BF4��3�м���KCl��Һ����CeCl3��KBF����������Ŀ���DZ�����������Ce��BF4��3��������ʽ��ʧ���ȥBF4-�����CeCl3�IJ��ʣ�
�ʴ�Ϊ��������������Ce��BF4��3��������ʽ��ʧ���ȥBF4-�����CeCl3�IJ��ʣ�
��4����Һ�е�C��Ce3+������1��10-5mol•L-1������ΪCe3+������ȫ������KSP[Ce��OH��3]=C��Ce3+��•C3��OH-��=1��10-20��֪��C��OH-��=$\root{3}{\frac{1��1{0}^{-20}}{1��1{0}^{-5}}}$mol•L-1=1��10-5mol•L-1����ʱ��Һ��PHΪ9��
�ʴ�Ϊ��9��
��5������NH4Cl�������ȷֽ����HCl������CeCl3ˮ�⣬���Լ���CeCl3•6H2O��NH4Cl�Ĺ�������ɵõ���ˮCeCl3��
�ʴ�Ϊ��NH4Cl�������ȷֽ����HCl������CeCl3ˮ�⣻
��6����0.1000mol•l-1��NH4��2Fe��SO4��2����Һ���25.00ml�������������ӵ����ʵ���Ϊ2.5��10-3mol�����ݷ�ӦFe2++Ce4+�TCe3++Fe3+������CeԪ���غ��֪��CeCl3������Ϊ2.5��10-3mol��246.5g/mol=0.6163g��������Ʒ��CeCl3����������Ϊ$\frac{0.6163g}{0.7500g}$��100%=82.2%��
�ʴ�Ϊ��82.2%��
����ʹ�þ��õģ�NH4��2Fe��SO4��2����Һ���еζ��������������ӱ��������������������Ի����ı�Һ�����Ϊƫ�����ø�CeCl3��Ʒ����������ƫ��
�ʴ�Ϊ��ƫ��
���� ��������ȡCeCl3��һ�ֹ�������Ϊ���壬�����˻��ϼۡ����ӷ�Ӧ�������ܽ�ƽ��ļ��㡢ʵ�������������ѧ�����֪ʶ�����ط������ƶϼ�ʵ���������ۺϿ��飬��Ŀ�Ѷ��еȣ�

 ��Ȥ����¹�֪��ϵ�д�
��Ȥ����¹�֪��ϵ�д� Ӣ��СӢ������Ĭдϵ�д�
Ӣ��СӢ������Ĭдϵ�д� �����ҵ���������ͯ������ϵ�д�
�����ҵ���������ͯ������ϵ�д� NaH2PO4��Na2HPO4�����ںϳɻ���ԭ�����������ƣ�Na5P3O10����
NaH2PO4��Na2HPO4�����ںϳɻ���ԭ�����������ƣ�Na5P3O10������1����˵��NaH2PO4��Һ��������ԭ������ӷ���ʽΪH2PO4-?HPO42-+H+��
��2����˵��Na2HPO4��Һ��������ԭ������ӷ���ʽΪHPO42-+H2O?H2PO4-+OH-��
��3���ⶨij����������������Na5P3O10�ĺ������ü�ӵζ��������������£�
��Ʒ1.3000g$\stackrel{ϡ����}{��}$H3PO4$��_{��pH��3-4}^{��NaOH��aq��}$NaH2PO4��$\stackrel{���100.00mL}{��ҺA}$$\stackrel{ȡ��}{��}$$��_{ָʾ��X}^{0.1000mol��L-1NaOH��Һ��}$Na2HPO4
1�ζ�ʱ���õ�ָʾ��X��Ϊ��̪��
��NaOH����ҺӦʢ������ͼ��ʾ�ĵζ����ң�ѡ����ס����ҡ��� �У�
�۵ζ�ʵ���¼���±�����Ʒ�����ʲ������Ӧ����
| �ζ����� | ������ҺA�������/mL�� | 0.1000mol•L-1NaOH��Һ����� | |
| �ζ�ǰ������/mL�� | �ζ��������/mL�� | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 0.20 | 20.20 |
| A�� | ��ʽ�ζ���ֻ��ˮϴ����δ�ô���Һ��ϴ | |
| B�� | ��ƿ�в���������ˮ | |
| C�� | ��ʽ�ζ��ܵζ�ǰ���촦�����ݣ��ζ���������ʧ | |
| D�� | �ζ�ǰ���ӿ̶ȶ������ζ����ӿ̶ȶ��� |
| A�� | �ñ�FeCl3��Һ�ζ�KI��Һ��ѡ��KSCN��Һ | |
| B�� | ��I2��Һ�ζ�Na2SO3��Һ��������ָʾ�� | |
| C�� | ��AgNO3��Һ�ζ�NaCl��Һ��Na2CrO4��ָʾ�� | |
| D�� | ��H2O2��Һ�ζ�KI��Һ��������ָʾ�� |

 �����ƣ�NaClO3�������ι�ҵ����Ҫ��Ʒ֮һ��
�����ƣ�NaClO3�������ι�ҵ����Ҫ��Ʒ֮һ�� ij��ѧѧϰС���ͬѧ ����������ԭ��Ӧ��MnO4-+5Fe2++8H+�TMn2++5Fe3++4H2O���ɲ��õζ��ķ����ⶨFeSO4������������ʵ�鲽�����£�
ij��ѧѧϰС���ͬѧ ����������ԭ��Ӧ��MnO4-+5Fe2++8H+�TMn2++5Fe3++4H2O���ɲ��õζ��ķ����ⶨFeSO4������������ʵ�鲽�����£�