��Ŀ����
2�� ij��ѧѧϰС���ͬѧ ����������ԭ��Ӧ��MnO4-+5Fe2++8H+�TMn2++5Fe3++4H2O���ɲ��õζ��ķ����ⶨFeSO4������������ʵ�鲽�����£�
ij��ѧѧϰС���ͬѧ ����������ԭ��Ӧ��MnO4-+5Fe2++8H+�TMn2++5Fe3++4H2O���ɲ��õζ��ķ����ⶨFeSO4������������ʵ�鲽�����£��ٳ����̷���Ʒ30.4g�����100mL������Һ
��ȡ10.0ml����Һ������ƿ�У�������һ����������
�۽�0.1mol•L-1��KMnO4��Һװ��ζ����У�����Һ����8.00mL��
�ܵζ�����Һ���ζ��յ�ʱ���ζ��ܵ�Һ�����18.00mL�����е����ʲ���������غ�ϡ���ᷴӦ������ش��������⣺
��1���ζ�ʱ�üף�����ͼ�ġ��ס����ҡ����ζ���ʢװKMnO4����Һ��
��2���ζ�ǰ�Ƿ�Ҫ�μ�ָʾ��������ǡ�������˵�����ɸ�����ر�������ɫ������ԭ����Һ������ɫ��
��3����ʵ��ﵽ�յ�ı�־�ǵ������һ��KMnO4��Һǡ���ɻ���ɫ��dz�Ϻ�ɫ���Ұ�����ڲ���ɫ��
��4����Ʒ��FeSO4����������Ϊ25%��
��5����������ѡ�ƫ�ߡ���ƫ�͡�����Ӱ�족��
�ٿ�ʼʵ��ʱ��ʽ�ζ��ܼ��촦�����ݣ��ڵζ�������������ʧƫ��
�����ζ�ǰ���ӵζ��̶ܿȶ���ʱ�����ʹ�ζ����ƫ�ͣ�
���� ��1��KMnO4����Һ����ǿ�����ԣ�ʢװ�ĵζ���Ӧѡȡ��ʽ�ζ��ܣ�
��2��������ر�������ɫ������ָʾ����
��3���ζ��յ�ʱ����ɫ����Ϻ�ɫ�����Ұ�����ڲ���ɫ���ɣ�
��4����0.1mol•L-1��KMnO4��Һװ��ζ����У�����Һ����8.00mL�����ζ�����Һ���ζ��յ�ʱ���ζ��ܵ�Һ�����18.00mL���������֪10.0ml����Һ��10.0ml0.1mol•L-1��KMnO4��ȫ��Ӧ������5Fe2++MnO4-+8H+�T5Fe3++Mn2++4H2O���㣻
��5������c�����⣩V�����⣩=c��������V�������жϲ��������������������Ӱ�죮
��� �⣺��1��KMnO4����Һ����ǿ�����ԣ��ܸ�ʴ�ܣ�Ӧʢװ����ʽ�ζ����У�ѡ��ף�
�ʴ�Ϊ���ף�
��2��������ر�������ɫ���ζ�ǰ����μ�ָʾ����
�ʴ�Ϊ��������ر�������ɫ������ԭ����Һ������ɫ��
��3���ζ��յ�ʱ����ɫ����Ϻ�ɫ�����Ұ�����ڲ���ɫ����ʵ��ﵽ�յ�ı�־�ǣ��������һ��KMnO4��Һǡ���ɻ���ɫ��dz�Ϻ�ɫ���Ұ�����ڲ���ɫ��
�ʴ�Ϊ���������һ��KMnO4��Һǡ���ɻ���ɫ��dz�Ϻ�ɫ���Ұ�����ڲ���ɫ��
��4����0.1mol•L-1��KMnO4��Һװ��ζ����У�����Һ����8.00mL�����ζ�����Һ���ζ��յ�ʱ���ζ��ܵ�Һ�����18.00mL���������֪10.0ml����Һ��10.0ml0.1mol•L-1��KMnO4��ȫ��Ӧ��
5Fe2++MnO4-+8H+�T5Fe3++Mn2++4H2O��
5 1
0.01L��C 0.1mol•L-1��0.01L
��C=0.5mol•L-1����10.0ml����Һ��FeSO4�����ʵ���Ϊ0.5mol•L-1��0.01L=0.005mol��FeSO4������Ϊ0.005mol��152g/mol=0.76g��100mL������Һ��FeSO4������Ϊ7.6g��������Ʒ��FeSO4����������Ϊ$\frac{7.6g}{30.4g}$��110%=25%��
�ʴ�Ϊ��25%��
��5���ٵζ�ǰ�ζ��ܼ��촦������δ�ų����ζ���������ʧ������v������ƫ����c�����⣩=$\frac{c������V������}{V�����⣩}$�жϿ�֪c�����⣩ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
�����ζ�ǰ���ӵζ��̶ܿȶ���ʱ������v������ƫС������c�����⣩=$\frac{c������V������}{V�����⣩}$�жϿ�֪c�����⣩ƫ�ͣ����ʹ�ζ����ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
���� ������Ҫ�����˷�Ӧ�ȵļ��㣬����ˮ��������Լ�������ԭ�ζ��ⶨ���ʵĺ�������Ŀ�Ѷ��еȣ�

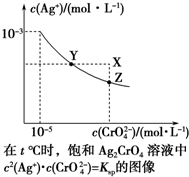 ��֪t��ʱAgCl��Ksp=4��10-10����t��ʱ��Ag2CrO4��ˮ�еij����ܽ�ƽ��������ͼ��ʾ������˵��������ǣ�������
��֪t��ʱAgCl��Ksp=4��10-10����t��ʱ��Ag2CrO4��ˮ�еij����ܽ�ƽ��������ͼ��ʾ������˵��������ǣ�������| A�� | ��t��ʱ��Ag2CrO4��KspΪ1��10-11 | |
| B�� | �ڱ�����Һ�м���K2CrO4��s����ʹ��Һ��Y�㵽Z�� | |
| C�� | ��t�棬Ag2CrO4��s��+2Cl-��aq��?2AgCl��s��+CrO${\;}_{4}^{2-}$��aq��ƽ�ⳣ��K=6.25��107 | |
| D�� | ��t��ʱ����0.001 mol•L-1 AgNO3��Һ�ζ�20 mL 0.001 mol•L-1 KCl��0.001 mol•L-1��K2CrO4�Ļ����Һ��CrO${\;}_{4}^{2-}$�ȳ��� |
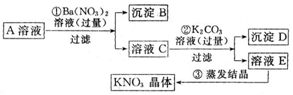
������ʵ��ܶȻ��������±���
| ���� | Cu��OH��2 | Fe��OH��3 | CuCl | CuI |
| Ksp | 2.2��10-20 | 2.6��10-39 | 1.7��10-7 | 1.3��10-12 |
��2���ڿ�����ֱ�Ӽ���CuCl2•2H2O����ò���������ˮCuCl2��ԭ����2CuCl2•2H2O$\frac{\underline{\;\;��\;\;}}{\;}$Cu��OH��2•CuCl2+2HCl+2H2O���û�ѧ����ʽ��ʾ����
��CuCl2•2H2O����õ�������ˮCuCl2�ĺ����������ڸ����HCl�����м�����ˮ��
��3��ijѧϰС���á���ӵ��������ⶨ����CuCl2•2H2O�������������������I-������Ӧ�����������ʣ��Ĵ��ȣ��������£�ȡ0.36g��������ˮ���������KI���壬��ַ�Ӧ�����ɰ�ɫ��������0.100 0mol•L-1 Na2S2O3����Һ�ζ�������ζ��յ�ʱ������Na2S2O3����Һ20.00mL��
�ٿ�ѡ�õ�����Һ���ζ�ָʾ�����ζ��յ����������ɫ��ȥ������һ��ʱ��ָ�ԭɫ��
��CuCl2��Һ��KI��Ӧ�����ӷ���ʽΪ2Cu2++4I-=2CuI��+I2��
�۸�������CuCl2•2H2O�������ٷ���Ϊ95%��
 1-����鳣�����л���Ӧ���ܼ���ʵ�����Ʊ�1-����飨CH3CH2CH2Br���ķ�Ӧ����Ҫʵ��װ�����£�
1-����鳣�����л���Ӧ���ܼ���ʵ�����Ʊ�1-����飨CH3CH2CH2Br���ķ�Ӧ����Ҫʵ��װ�����£���֪��
��CH3CH2CH2OH+HBr$\stackrel{��}{��}$CH3CH2CH2Br+H2O
��.2CH3CH2CH2OH$��_{140��}^{Ũ����}$��CH3CH2CH2��2O�������ѣ�+H2O
�����õ�������������£�
| ��Է� ������ | �ܶ� /g•mL-1 | �е�/�� | ˮ�� �ܽ��� | |
| ������ | 60 | 0.896 | 97.1 | �� |
| ������ | 102 | 0.74 | 90 | �������� |
| 1-����� | 123 | 1.36 | 71 | ���� |
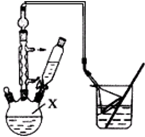
����A�м���50g��������һ������Ũ���ᡢ�廯��һ����ȣ����뼸����ʯ�����ڱ���69��72���������2Сʱ���������ռ�68��90�����Һ ����̼������Һϴ�����ԣ���Һ�����������ռ�68��76�����Һ���õ�����1-�����66g����ش�
��1��Bװ�������ǣ����Σ������ܣ���ʯ�������Ƿ�ֹҺ�屩�У�
��2������Aǰ�����ȴ�b���a����b��������B��ͨ��ˮ��
��3������ܵ�Ŀ����Ҫ��ϴȥŨ���ᣬ��ҡ���ã��ֲ���Ӧ�ӷ�Һ©�����£���ϡ����¡����ڷ������
��4����ʵ�����õ���1-����������C������ȷ����ţ���
A.41%������B.50%������C.64%������D.70%
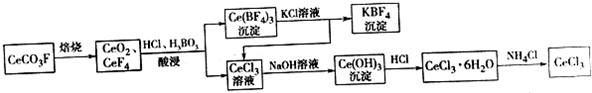
 ���嶡�����ӣ�
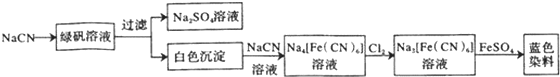
���嶡�����ӣ�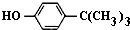 ����ҵ��;�㷺�����������������Է�ȩ��֬���ȶ��������ϵȣ�ʵ�����Ա��ӡ��嶡����[��CH3��3CCl]��Ϊԭ���Ʊ����嶡�����ӣ�
����ҵ��;�㷺�����������������Է�ȩ��֬���ȶ��������ϵȣ�ʵ�����Ա��ӡ��嶡����[��CH3��3CCl]��Ϊԭ���Ʊ����嶡�����ӣ� ��
�� ��
��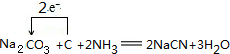 ��
��
 ij��ѧѧϰС����о������ǣ�̽���ⶨ���ᾧ�壨H2C2O4•xH2O����x��ֵ������ͬѧͨ���������ϲ�Ѱ�ã�����������ˮ��ˮ��Һ����������KMnO4��Һ���еζ���
ij��ѧѧϰС����о������ǣ�̽���ⶨ���ᾧ�壨H2C2O4•xH2O����x��ֵ������ͬѧͨ���������ϲ�Ѱ�ã�����������ˮ��ˮ��Һ����������KMnO4��Һ���еζ���