��Ŀ����
�߷��ӻ�����G����Ϊ﮵���� Li+Ǩ�ƵĽ��ʣ��ϳ�G���������£�

��֪��
��1��D������������ ��
��2��A��B�ķ�Ӧ������ ��
��3��C�Ľṹ��ʽ�� ��
��4��D��E��Ӧ������ ��
��5��G�Ľṹ��ʽ�� ��
��6����֪��
M��N�Ļ�ѧ����ʽ�� ��
��7��D��ͬ���칹���У�д����˳���칹�Ľṹ��ʽ
ע�⣺�������ǻ�����ͬһ��̼�ϣ��ǻ�����˫����̼�ϵĽṹ���ȶ������迼�ǣ�
�ڲ����ǹ������ͻ�״�ṹ ��˳���ṹ��д����

��֪��

��1��D������������
��2��A��B�ķ�Ӧ������
��3��C�Ľṹ��ʽ��
��4��D��E��Ӧ������
��5��G�Ľṹ��ʽ��
��6����֪��

M��N�Ļ�ѧ����ʽ��
��7��D��ͬ���칹���У�д����˳���칹�Ľṹ��ʽ
ע�⣺�������ǻ�����ͬһ��̼�ϣ��ǻ�����˫����̼�ϵĽṹ���ȶ������迼�ǣ�
�ڲ����ǹ������ͻ�״�ṹ ��˳���ṹ��д����
���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������F�Ľṹ��ʽ���A��B��C��D�ķ�Ӧ��������֪C4H8�ĽṹΪ�� �����巢���ӳɷ�Ӧ����AΪ
�����巢���ӳɷ�Ӧ����AΪ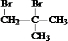 ��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ
��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ ����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ
����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ ����6����CΪ
����6����CΪ �������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪����N���ݴ˽��
�������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪����N���ݴ˽��
 �����巢���ӳɷ�Ӧ����AΪ
�����巢���ӳɷ�Ӧ����AΪ ��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ
��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ ����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ
����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ ����6����CΪ
����6����CΪ �������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪����N���ݴ˽��
�������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪����N���ݴ˽�����
�⣺��F�Ľṹ��ʽ���A��B��C��D�ķ�Ӧ��������֪C4H8�ĽṹΪ�� �����巢���ӳɷ�Ӧ����AΪ
�����巢���ӳɷ�Ӧ����AΪ ��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ
��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ ����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ
����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ ��
��
��1��DΪHOOCC��OH����CH3��2������������Ϊ���Ȼ����ǻ����ʴ�Ϊ���Ȼ����ǻ���
��2��AΪ ��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��
��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��
�ʴ�Ϊ��ˮ�ⷴӦ����ȡ����Ӧ����
��3��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������֪��֪CΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��4��DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪֪D��Ũ��������������E����Ӧ����ʽΪ�� ��
��
�ʴ�Ϊ��Ũ���ᡢ���ȣ�
��5��ͨ������F�Ľṹ��ʽ��֪��F��ֻ��ͨ��̼̼˫���ļӾ۷�Ӧ���ɸ߷��ӻ�����G����GΪ ��
��
�ʴ�Ϊ�� ��
��
��6��CΪ �������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪���ɶ���N�ķ�Ӧ����ʽΪ��
�������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪���ɶ���N�ķ�Ӧ����ʽΪ�� ��
��
�ʴ�Ϊ�� ��
��
��7��DΪHOOCC��OH����CH3��2��ͬ���칹������˳���칹��Ӧ����C=C˫�����Ң������ǻ�����ͬһ��̼�ϣ��ǻ�����˫����̼�ϵĽṹ���ȶ������迼�ǣ��ڲ����ǹ������ͻ�״�ṹ������˳���ṹ�У� ��
�� ��
��
 ��
��
�ʴ�Ϊ�� ��
�� ��
��
 ��
�� ��
��
 �����巢���ӳɷ�Ӧ����AΪ
�����巢���ӳɷ�Ӧ����AΪ ��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ
��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������Ϣ�ڿ�֪CΪ ����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ
����DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪D��Ũ��������������E��E�뻷�����鷴Ӧ����F����֪EΪCH2=C��CH3��COOH��F��ֻ��ͨ���Ӿ۷�Ӧ���ɸ߷��ӻ�����GΪ ��
����1��DΪHOOCC��OH����CH3��2������������Ϊ���Ȼ����ǻ����ʴ�Ϊ���Ȼ����ǻ���
��2��AΪ
 ��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B��
��A����������ˮ��Һ�����������·���ˮ�ⷴӦ����B���ʴ�Ϊ��ˮ�ⷴӦ����ȡ����Ӧ����
��3��BΪHOCH2C��OH����CH3��2��B����������C��C��������D�������֪��֪CΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����4��DΪHOOCC��OH����CH3��2��F�Ľṹ��ʽ�к���̼̼˫������֪֪D��Ũ��������������E����Ӧ����ʽΪ��
 ��
���ʴ�Ϊ��Ũ���ᡢ���ȣ�
��5��ͨ������F�Ľṹ��ʽ��֪��F��ֻ��ͨ��̼̼˫���ļӾ۷�Ӧ���ɸ߷��ӻ�����G����GΪ
 ��
���ʴ�Ϊ��
 ��
����6��CΪ
 �������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪���ɶ���N�ķ�Ӧ����ʽΪ��
�������֪��Ӧ�ƶ�MӦΪ��CH3��2C��OH��CH��NH2��COOH������֪���ɶ���N�ķ�Ӧ����ʽΪ�� ��
���ʴ�Ϊ��
 ��
����7��DΪHOOCC��OH����CH3��2��ͬ���칹������˳���칹��Ӧ����C=C˫�����Ң������ǻ�����ͬһ��̼�ϣ��ǻ�����˫����̼�ϵĽṹ���ȶ������迼�ǣ��ڲ����ǹ������ͻ�״�ṹ������˳���ṹ�У�
 ��
�� ��
�� ��
��
�ʴ�Ϊ��
 ��
�� ��
�� ��
�� ��
��
���������⿼���л����ƶϣ���Ҫѧ���Ը������Ϣ�������ã��ܽϺõĿ���ѧ���Ķ���ȡ��Ϣ���������ƶϹ�����ע�ⷴӦ���������ʵķ���ʽ���ṹ��ʽ����Ϣ���ۺ����ã���ȡ�������Ʒ����Ͻ����ƶϣ���7����ͬ���칹����дΪ�״��㡢�ѵ㣬�ѵ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
��һ������Fe2��SO4��3�루NH4��2Fe��SO4��2�Ļ��������ˮ�����2L��Һ����c��NH4+��=1mol/L��c��SO42-��=3mol/L�������Һ��Ͷ����������ۺ��������ӵ�Ũ��Ϊ������ˮ�⣩��������
| A��1.5mol/L |
| B��2mol/L |
| C��2.5mol/L |
| D��3mol/L |
�������е������Һ��˵���У���ȷ���ǣ�������
| A�������£�10mL 0.2mol/L NH4NO3��Һ��10mL 0.1mol/L NaOH��Һ��Ϻ�����pH=9.6����Һ�У�c��NO3-����c��NH4+����c��Na+����c��NH3?H2O����c��OH-����c��H+�� |
| B��0.1mol/L Na2S��Һ�У�c��Na+��+c��H+��=c��S2-��+c��HS-��+c��OH-�� |
| C��������a mL 0.1mol/L KOH��b mL 0.1mol/L HCN����Һ��Ϻ�pH��7����һ����a��b |
| D��10mL 0.1mol/L NaCl��Һ����������Ϊx��10mL 0.1mol/L CH3COONa��Һ����������Ϊy����x=y |
����ȷ��ʾ���з�Ӧ�����ӷ���ʽ�ǣ�������
| A������ˮ��Ӧ��Na+2H2O�TNa++OH-+H2�� | ||||
| B��KI��Һ�е���ϡ���ᣬ��������4H++4I-+O2�T2I2+2H2O | ||||
| C��CuCl2��Һ�м��백ˮCu2++2OH-�TCu��OH��2�� | ||||
D����NH4HCO3��Һ�мӹ�����NaOH��Һ�����ȣ�NH4++OH-
|


