��Ŀ����
 ��һ�����Ϊ2L������ܱ������м���0.5molCaCO3��������ӦCaCO3��s���TCaO ��s��+CO2��g������ö�����̼�����ʵ���Ũ�����¶ȵı仯��ϵ��ͼ��ʾ��ͼ��A��ʾCO2��ƽ��Ũ�����¶ȵĹ�ϵ���ߣ�B��ʾ��ͬ�¶��·�Ӧ������ͬʱ��ʱCO2�����ʵ���Ũ�ȵı仯���ߣ��밴Ҫ��ش��������⣺
��һ�����Ϊ2L������ܱ������м���0.5molCaCO3��������ӦCaCO3��s���TCaO ��s��+CO2��g������ö�����̼�����ʵ���Ũ�����¶ȵı仯��ϵ��ͼ��ʾ��ͼ��A��ʾCO2��ƽ��Ũ�����¶ȵĹ�ϵ���ߣ�B��ʾ��ͬ�¶��·�Ӧ������ͬʱ��ʱCO2�����ʵ���Ũ�ȵı仯���ߣ��밴Ҫ��ش��������⣺��1���÷�Ӧ����ӦΪ
��2������÷�Ӧ��ƽ�ⳣ��Kֵ��÷�Ӧ
a��һ�����淴Ӧ�����ƶ�
b����ƽ���ƶ�ʱ����Ӧ������������С
c��һ��������Ӧ�����ƶ�
d����ƽ���ƶ�ʱ�淴Ӧ�����ȼ�С������
��3����˵�����¶ȵ����ߣ�����B������A�ƽ���ԭ��
��4����T5���£�ά���¶Ⱥ�����������䣬������ƽ����ϵ���ٳ���0.5molN2�������ƽ��ʱ�����е�CaCO3������Ϊ
| ���� | ����ƽ�ⳣ����25�棩 |
| C6H5OH | Ki=1.28��10-10 |
| H2CO3 | Ki1=4.3��10-7 |
| Ki2=5.6��10-11 |
���㣺���ʵ�����Ũ����ʱ��ı仯����,��ѧƽ���Ӱ������
ר�⣺
��������1����ͼ���ж��¶���ƽ���ƶ���Ӱ�죬���жϷ�Ӧ�е���ЧӦ�����ݻ�ѧ��Ӧ����ʽ��֪��ƽ�ⳣ��K=c��CO2�����㣻
��2��ƽ�ⳣ������˵��ƽ�������ƶ�������¶ȶ�ƽ�ⳣ����Ӱ���жϣ�
��3�������¶ȶԷ�Ӧ���ʵ�Ӱ���жϣ�
��4����������������䣬ƽ����ϵ���ٳ���0.5molN2��ƽ�ⲻ�ƶ����ݴ˼��㣻
��5���ɵ���ƽ�ⳣ����֪�����ӵ���������̼�ᵫ��̼�����ǿ���ݴ˴��⣮
��2��ƽ�ⳣ������˵��ƽ�������ƶ�������¶ȶ�ƽ�ⳣ����Ӱ���жϣ�
��3�������¶ȶԷ�Ӧ���ʵ�Ӱ���жϣ�
��4����������������䣬ƽ����ϵ���ٳ���0.5molN2��ƽ�ⲻ�ƶ����ݴ˼��㣻
��5���ɵ���ƽ�ⳣ����֪�����ӵ���������̼�ᵫ��̼�����ǿ���ݴ˴��⣮
���
�⣺��1����ͼ���֪�������¶ȵ����ߣ�������̼��ƽ��Ũ������˵��ƽ��������Ӧ�����ƶ������Ը÷�ӦΪ���ȷ�Ӧ�����ݻ�ѧ��Ӧ����ʽ��֪��ƽ�ⳣ��K=c��CO2��������T5��ʱ��K=c��CO2��=0.2��
�ʴ�Ϊ�����ȣ� 0.2��
��2��ƽ�ⳣ������˵��ƽ�������ƶ�����ʱ�ı������Ϊ�����¶ȣ�����÷�Ӧ��ƽ�ⳣ��Kֵ�����
a��һ��������Ӧ�����ƶ�����a����
b����ƽ���ƶ�ʱ�������������¶ȣ����������淴Ӧ���ʶ��������ڸ÷�Ӧ�����ȷ�Ӧ������ƽ���ƶ����¶Ȼ��������ͣ��������淴Ӧ���ʶ����С����b��ȷ��
c��һ��������Ӧ�����ƶ�����c��ȷ��
d������b�ķ�����֪��d����
��ѡbc��
��3�������¶ȶԷ�Ӧ���ʵ�Ӱ���֪�������¶����ߣ���Ӧ���ʼӿ죬�ﵽƽ������Ҫ��ʱ���̣���������B������A�ƽ���
�ʴ�Ϊ�������¶����ߣ���Ӧ���ʼӿ죬�ﵽƽ������Ҫ��ʱ���̣�
��4����������������䣬ƽ����ϵ���ٳ���0.5molN2��ƽ�ⲻ�ƶ���������T5��ʱ��������̼�����ʵ���Ϊ2L��0.2mol/L=0.4mol���ֽ��CaCO3������Ϊ100��0.4g=40g������ƽ��ʱ�����е�CaCO3������Ϊ0.5mol��100g/mol-40g=10g��
�ʴ�Ϊ��10��
��5��������̼ͨ�뱽������Һ�Ļ�ѧ��Ӧ���ӷ���ʽΪC6H5O-+CO2+H2O��C6H5OH+HCO3-���ɵ���ƽ�ⳣ����֪�����ӵ���������̼�ᵫ��̼�����ǿ��������̼��ˮ�������̼�ᣬ��̼�����Աȱ�������ǿ���������ɱ��ӣ�̼��ĵ������������һ����H2CO3 H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3-
H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3- H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣬
H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣬
�ʴ�Ϊ��C6H5O-+CO2+H2O��C6H5OH+HCO3-���ɵ���ƽ�ⳣ����֪�����ӵ���������̼�ᵫ��̼�����ǿ��������̼��ˮ�������̼�ᣬ��̼�����Աȱ�������ǿ���������ɱ��ӣ�̼��ĵ������������һ����H2CO3 H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3-
H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3- H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣮
H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣮
�ʴ�Ϊ�����ȣ� 0.2��
��2��ƽ�ⳣ������˵��ƽ�������ƶ�����ʱ�ı������Ϊ�����¶ȣ�����÷�Ӧ��ƽ�ⳣ��Kֵ�����
a��һ��������Ӧ�����ƶ�����a����
b����ƽ���ƶ�ʱ�������������¶ȣ����������淴Ӧ���ʶ��������ڸ÷�Ӧ�����ȷ�Ӧ������ƽ���ƶ����¶Ȼ��������ͣ��������淴Ӧ���ʶ����С����b��ȷ��
c��һ��������Ӧ�����ƶ�����c��ȷ��
d������b�ķ�����֪��d����
��ѡbc��
��3�������¶ȶԷ�Ӧ���ʵ�Ӱ���֪�������¶����ߣ���Ӧ���ʼӿ죬�ﵽƽ������Ҫ��ʱ���̣���������B������A�ƽ���
�ʴ�Ϊ�������¶����ߣ���Ӧ���ʼӿ죬�ﵽƽ������Ҫ��ʱ���̣�
��4����������������䣬ƽ����ϵ���ٳ���0.5molN2��ƽ�ⲻ�ƶ���������T5��ʱ��������̼�����ʵ���Ϊ2L��0.2mol/L=0.4mol���ֽ��CaCO3������Ϊ100��0.4g=40g������ƽ��ʱ�����е�CaCO3������Ϊ0.5mol��100g/mol-40g=10g��
�ʴ�Ϊ��10��
��5��������̼ͨ�뱽������Һ�Ļ�ѧ��Ӧ���ӷ���ʽΪC6H5O-+CO2+H2O��C6H5OH+HCO3-���ɵ���ƽ�ⳣ����֪�����ӵ���������̼�ᵫ��̼�����ǿ��������̼��ˮ�������̼�ᣬ��̼�����Աȱ�������ǿ���������ɱ��ӣ�̼��ĵ������������һ����H2CO3
 H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3-
H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3- H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣬
H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣬�ʴ�Ϊ��C6H5O-+CO2+H2O��C6H5OH+HCO3-���ɵ���ƽ�ⳣ����֪�����ӵ���������̼�ᵫ��̼�����ǿ��������̼��ˮ�������̼�ᣬ��̼�����Աȱ�������ǿ���������ɱ��ӣ�̼��ĵ������������һ����H2CO3
 H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3-
H++HCO3-���ò��������H+�������C6H5O-�������Ը�����C6H5OH�����µ�һ������ƽ�������ƶ����ڶ������룺HCO3- H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣮
H++CO32-��C6H5OH���Դ���HCO3-�����Եڶ����������H+�������CO32-���������C6H5O-���ʲ�Ӱ��ڶ�������ƽ�⣮
������������Ҫ������Ӱ��ƽ�ⳣ�������ء�Ӱ��ƽ���ƶ������ء���ѧƽ�ⳣ���ļ��㡢����ƽ�ⳣ����Ӧ�õ�֪ʶ���е��Ѷȣ�����ʱע��ѧ���ͼ�л�ȡ��Ϣ����ע���ƽ�ⳣ����������⣮

��ϰ��ϵ�д�
�����Ŀ
ij��ɫ��Һ�к��У���K+����Ba2+����Cl-����Br-����SO32-����CO32-����SO42-�е�һ�ֻ��֣����ν�������ʵ�飬��ÿ�������Լ����������۲쵽���������£����н�����ȷ���ǣ�������
| ���� | ���� | ���� |
| �� | ��pH��ֽ���� | ��Һ��pH����7 |
| �� | ����Һ�еμ���ˮ���ټ���CCl4������ | CCl4��ʳ�ɫ |
| �� | ���������Һ�м���Ba��NO3��2��Һ��ϡHNO3 | �а�ɫ�������� |
| �� | ���ˣ�����Һ�м���AgNO3��Һ��ϡHNO3 | �а�ɫ�������� |
| A�����ܺ��е������Ǣ٢ڢ� |
| B���϶����е������Ǣ٢� |
| C������ȷ���������Ǣ٢ۢ� |
| D���϶�û�е������Ǣڢݢ� |
���ʵ���Ũ����ͬ��������Һ�У�NH4+Ũ�������ǣ�������
| A��NH4Cl |
| B��NH4HCO3 |
| C��CH3COONH4 |
| D��NH4HSO4 |
ij�л�����ҩ���������м��壬��ṹ��ʽ��ͼ�������й�������ȷ���ǣ�������
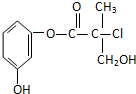

| A�����л�������ˮ�ɷ��ӳɷ�Ӧ |
| B�����л�����Ũ�����Ϲ��ȿɷ�����ȥ��Ӧ |
| C��1 mol���л���������NaOH��Һ��Ӧ�������3 mol NaOH |
| D�����л��ᆳ��������������������ͭ����Һ��������ש��ɫ���� |
����Ϊǿ������ǣ�������
��NaOH���� ��CH3COOH ��BaSO4���� ��������Һ��
��NaOH���� ��CH3COOH ��BaSO4���� ��������Һ��
| A���٢� | B���٢� | C���٢� | D���٢ۢ� |
���й��ڻ�ѧ����ı�ʾ�У���ȷ���ǣ�������
A��������ĵ���ʽ |
| B����ˮ�ķ���ʽH218O |
| C������ĽṹʽCH3COOH |
D��Mg2+�Ľṹʾ��ͼ |
����������ʯ�ҡ�ˮ��������Ϊ1��0.56��100��������ͭɱ����������Һ����ɷֵĻ�ѧʽ�ɱ�ʾΪCuSO4?xCaSO4?xCu��OH��2?yCa��OH��2����x=3ʱ��yΪ��������
| A��1 | B��3 | C��5 | D��7 |
��Na2O2��Na2CO3��NaHCO3��NaCl�����е�ij������ɵĻ����������м������������ᣬ������ų������ų�������ͨ��������NaOH��Һ����������������٣���������������ڿ����г�ּ��ȣ�Ҳ������ų��������ж���ȷ���ǣ�������
| A���������һ������Na2O2��NaHCO3 |
| B���������һ��������Na2CO3��NaCl |
| C�������һ��������Na2O2��NaCl |
| D���������һ������Na2O2��Na2CO3��NaHCO3 |