��Ŀ����
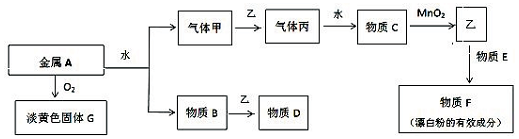
17����������Ϊ��ɫ���壬��Ϊһ�ֻ���ԭ�ϣ��ɹ㷺����Ϳ�ϡ��մɡ���������ȵ���ɫ���Լ����͵�ز��ϡ��й���ϵ��������ϳɲ���������������ͼ1��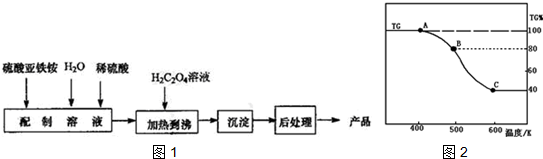
��1�����ƣ�NH4��2Fe��SO4��2•6H2O��Һʱ�����������ϡ���ᣬĿ��������Fe2+��NH4+����ˮ�⣮
��2���õ��IJ���������������ϴ�ӣ������Ƿ�ϴ�Ӹɾ��ķ�����ȡ�������һ�ε�ϴ����Һ���Թ��У������еμ������ữ�� BaCl2��Һ�����ް�ɫ�����������������ϴ�Ӹɾ���
��3�����ƵõIJ�Ʒ��FeC2O4•2H2O������������н��м��ȷֽ⣬�����ͼ2��TG%��ʾ������������ռԭ��Ʒ�������İٷ�������
����A-B������Ӧ�Ļ�ѧ����ʽΪFeC2O4•2H2O$\frac{\underline{\;\;��\;\;}}{\;}$FeC2O4+2H2O��
����֪B��C�������е����ʵ�����������̬���������ɣ�д��B��C�Ļ�ѧ����ʽFeC2O4 $\frac{\underline{\;\;��\;\;}}{\;}$FeO+CO��+CO2����
��4��ij����������Ʒ�к�����������泥�Ϊ�˲ⶨ������Ʒ�в�����ĺ�����ijͬѧ�������·���ʵ�飺
��ȷ����m g��Ʒ����������2mol/L�����в���100mL����ƿ���ݣ�
��ȡ������Һ20mL����c mol/L������ر���Һ�ζ�����Һ��Ϊ����ɫ�����ĸ��������Һ�����ΪV1 mL��
����������Һ�м�������Zn�ۣ�ʹ��Һ�е�Fe3+ǡ��ȫ����ԭΪFe2+�����ˣ�
����ϴ��ʣ���п�ۺ���ƿ��ϴ��Һ������Һ
������c mol/L KMnO4��Һ�ζ�����Һ����Һ���ֵ���ɫ������KMnO4��Һ�����V2 mL��
�ش��������⣺
����֪�����ᣨH2C2O4�������Ը��������Һ��Ӧ�������������ݲ�������ɫ��ʧ��д���÷�Ӧ�����ӷ���ʽ��2MnO4-+5H2C2O4+6H+=2Mn2++10CO2+8H2O��
����ʡ�Բ��������ⶨ�IJ�������Ӻ���ƫ���ƫ����ƫС�����䡱��
��m g��Ʒ�в�������ӵ����ʵ���Ϊc��V1-V2����10-3��$\frac{25}{2}$mol����c��V1��V2��ʽ�ӱ�ʾ�����ػ���
���� ��1����Һ���������Ӻ�笠�����ˮ�⣬�����������ᣬ����Fe2+��NH4+����ˮ�⣻
��2��ϴ�Ӳ����ľ��巽��Ϊ���ز�������©���м�����������ˮ����û������������ˮ��Ȼ���£��ظ�2-3�Σ�ȡ���һ��ϴ��Һ������Ƿ�����������ӽ����ж��Ƿ�ϴ�Ӹɾ���
��3����B�������������ռԭ��Ʒ�������İٷ���λ80%����ʧ20%��FeC2O4.2H2O�нᾧˮ������Ϊ$\frac{36}{180}$��100%=20%����A��B������Ӧ��FeC2O4.2H2O����ʧȥ�ᾧˮ��
����ͼ��֪��������Cʱ���ֽ���ȫ���ٶ�������������Ϊ180g�������ʵ���Ϊ1mol����C���������Ϊ180g��40%=72g����Ԫ������Ϊ1mol��56g/mol=56����Ӧ������Ԫ��Ϊ72g-56g=16g������ԭ������ԭ�����ʵ���֮��Ϊ1��1��ӦΪFeO��B�㵽C����FeC2O4�ֽ�ķ�Ӧ��
��4������ʵ��ԭ����2MnO4-+5H2C2O4+6H+�T2Mn2++10CO2+8H2O��MnO4-+8H++5Fe2+�T5Fe3++Mn2++4H2O������cmol/L������ر���Һ�ζ���Ʒ��Һ����Ϊ��������������Ӷ��ܱ�������ر���Һ���������Ե�һ�����ĸ��������Һ�����ΪV1mLΪ������������������ӹ�ͬ���ģ�Ȼ��������� Zn �ۣ�ʹ��Һ�е� Fe3+ǡ��ȫ����ԭΪ Fe2+������c mol/L KMnO4��Һ�ζ�����Һ����Һ���ֵ���ɫ������KMnO4��Һ�����V2 mL�������������������ĵģ�����������������ĸ��������Һ�����Ϊ��V1-V2 ��mL���ݴ˷�����
��� �⣺��1����Һ���������Ӻ�笠�����ˮ�������������ӣ�ˮ�������ԣ������������ᣬ����Fe2+��NH4+����ˮ�⣬�ʴ�Ϊ������Fe2+��NH4+����ˮ�⣻
��2��ϴ�Ӳ����ľ��巽��Ϊ���ز�������©���м�����������ˮ����û������������ˮ��Ȼ���£��ظ�2-3�Σ������Ƿ�ϴ�Ӹɾ��ķ�����ȡ�������һ�ε�ϴ����Һ���Թ��У������еμ������ữ�� BaCl2��Һ�����ް�ɫ�����������������ϴ�Ӹɾ���
�ʴ�Ϊ��ȡ�������һ�ε�ϴ����Һ���Թ��У������еμ������ữ�� BaCl2��Һ�����ް�ɫ�����������������ϴ�Ӹɾ���
��3������ͼ��֪����������Bʱʣ�����Ϊ80%����ʧ20%��FeC2O4.2H2O�нᾧˮ������Ϊ$\frac{36}{180}$��100%=20%����A��B������Ӧ��FeC2O4•2H2O����ʧȥ�ᾧˮ����Ӧ����ʽΪ��FeC2O4•2H2O$\frac{\underline{\;\;��\;\;}}{\;}$FeC2O4+2H2O��
�ʴ�Ϊ��FeC2O4•2H2O$\frac{\underline{\;\;��\;\;}}{\;}$FeC2O4+2H2O��
����ͼ��֪B��������Cʱ���ֽ���ȫ���ٶ�������������Ϊ180g�������ʵ���Ϊ1mol����C���������Ϊ180g��40%=72g����Ԫ������Ϊ1mol��56g/mol=56����Ӧ������Ԫ��Ϊ72g-56g=16g����ԭ�����ʵ���Ϊ1mol������ԭ������ԭ�����ʵ���֮��Ϊ1��1��ӦΪFeO������������ԭ��Ӧ�����غ�������㣬̼Ԫ�ػ��ϼ۴�+3�۱仯Ϊ+2�ۺ�+3�ۣ�����һ����̼�Ͷ�����̼���壬ԭ���غ���ƽд����ѧ����ʽ��FeC2O4 $\frac{\underline{\;\;��\;\;}}{\;}$FeO+CO��+CO2�����ʴ�Ϊ��FeC2O4 $\frac{\underline{\;\;��\;\;}}{\;}$FeO+CO��+CO2����
��4���ٲ��ᣨH2C2O4�������Ը��������Һ��Ӧ�������������ݲ�������ɫ��ʧ����Ӧ�����ӷ���ʽΪ��2MnO4-+5H2C2O4+6H+=2Mn2++10CO2+8H2O��
�ʴ�Ϊ��2MnO4-+5H2C2O4+6H+=2Mn2++10CO2+8H2O��
�ڸ������Ϸ�������ʡ�Բ��������ʹ����������ƫ�٣��������ĸ��������Һ�����ҲV2 ƫС����������������ĸ��������Һ�����Ϊ��V1-V2 ��mLƫ�����Բⶨ�IJ�������Ӻ���Ҳƫ�ʴ�Ϊ��ƫ��
�۸������Ϸ�����������������ĸ��������Һ�����Ϊ��V1-V2 ��mL����2MnO4-+5H2C2O4+6H+�T2Mn2++10CO2+8H2O������mg��Ʒ�в�������ӵ����ʵ���Ϊc��V1-V2����10-3��$\frac{5}{2}$��$\frac{100ml}{20ml}$=c��V1-V2����10-3��$\frac{25}{2}$mol���ʴ�Ϊ��c��V1-V2����10-3��$\frac{25}{2}$mol��
���� ���⿼���Ϊ�ۺϣ��漰������ʽ����д��ʵ������������ζ��ȼ��㣬ע�����ʵ��֪ʶ�Ļ��ۣ�����ʵ�鲽�衢ԭ����ע����������⣬��Ŀ�Ѷ��еȣ�

| A�� |  | B�� |  | ||
| C�� |  | D�� |  |
| ѡ�� | ��Ŀ | ��� | �����Ƽ |
| A�� | ԭ�� | ʳ�Ρ���������ʯ�� | ʳ�Ρ�������������̼ |
| B�� | ���ܵĸ����� | �Ȼ��� | �Ȼ�� |
| C�� | ѭ������ | ������������̼ | �������Ȼ��� |
| D�� | ���� | ԭ���á����ʸ� | �豸���ܺĵ� |
| A�� | A | B�� | B | C�� | C | D�� | D |
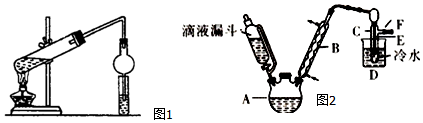
��ijͬѧ���Ҵ��������Ũ������ȡ����������װ����ͼ1��ʾ��
��1��װ���и���ܵ������Ƿ�ֹС�Թ���Һ�巢������
��2��С�Թ��е��Լ�����˳��ΪB
A��Ũ���� �Ҵ� ���� B���Ҵ� Ũ���� ���� C������ Ũ���� �Ҵ�
II����֪��R-OH+HX��R-X+H2O��ͼ2��ʵ�������Ҵ���Ũ������廯�Ʒ�Ӧ�Ʊ��������װ�ã�ͼ��ʡȥ�˼���װ�ã��Ҵ��������顢���йز������ݼ�����
| �Ҵ� | ������ | �� | |
| ״̬ | ��ɫҺ�� | ��ɫҺ�� | �����ɫҺ�� |
| �ܶ�/g•cm-3 | 0.79 | 1.44 | 3.1 |
| �е�/�� | 78.5 | 38.4 | 59 |
��4��Ϊ��ȥ��Ʒ�е�Br2�����ѡ��������Һ��ϴ�Ӳ�ƷB��
A���������� B���������� C���⻯��
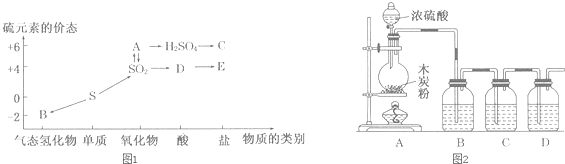
 ���᳧�����ջ�����FeS2������ȡ���ᣬʵ�����������᳧��������Ҫ�ɷ���Fe2O3������FeS��SiO2���Ʊ��̷���
���᳧�����ջ�����FeS2������ȡ���ᣬʵ�����������᳧��������Ҫ�ɷ���Fe2O3������FeS��SiO2���Ʊ��̷���
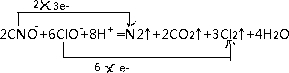 ��
��