��Ŀ����
4����֪X��Y��Z����Ԫ�ص�ԭ������֮�͵���42��XԪ��ԭ�ӵ�4p�������3��δ�ɶԵ��ӣ�YԪ��ԭ�ӵ������2p�������2��δ�ɶԵ��ӣ�X��Y���γɻ�����X2Y3��ZԪ�ؿ����γɸ�һ�����ӣ���ش��������⣺��1��XԪ��ԭ�ӻ�̬ʱ�ĵ����Ų�ʽΪ1s22s22p63s23p63d104s24p3����Ԫ�صķ�����As��
��2��ZԪ�������ڱ�����s������s��p��d���������Ԫ����3��ͬλ�أ�
��3��X��Z���γɻ�����XZ3���û�����Ŀռ乹��Ϊ�����Σ�
��4����֪������X2Y3��ϡ������Һ�пɱ�����п��ԭΪXZ3�����ﻹ��ZnSO4��H2O���÷�Ӧ�Ļ�ѧ����ʽ��As2O3+6Zn+6H2SO4�T2AsH3��+6ZnSO4+3H2O��
��5��X���⻯����ͬ��ڶ�����������Ԫ�����γɵ��⻯���ȶ��ԡ��е�ߵ�˳��ֱ��ǣ��û�ѧʽ��ʾ��NH3��PH3��AsH3��NH3��AsH3��PH3��
���� XԪ��ԭ�ӵ�4p�������3��δ�ɶԵ��ӣ�XԪ��ԭ�ӵĺ�������Ų�ʽΪ 1s22s22p63s23p63d104s24p3�����ڵ������ڵڢ�A�壬��XΪAsԪ�أ�YԪ��ԭ�ӵ������2p�������2��δ�ɶԵ��ӣ�Y��2p�������2�����ӻ�4�����ӣ�����YΪ̼Ԫ�ػ���Ԫ�أ�X��Y���γɻ�����X2Y3����YΪ��Ԫ�أ�X��Y��Z����Ԫ�ص�ԭ������֮�͵���42����Z��������Ϊ42-8-33=1����ZΪ��Ԫ�أ���ԭ�ӿ����γɸ�һ�����ӣ��������⣬�ݴ˽��
��� �⣺XԪ��ԭ�ӵ�4p�������3��δ�ɶԵ��ӣ�XԪ��ԭ�ӵĺ�������Ų�ʽΪ 1s22s22p63s23p63d104s24p3�����ڵ������ڵڢ�A�壬��XΪAsԪ�أ�YԪ��ԭ�ӵ������2p�������2��δ�ɶԵ��ӣ�Y��2p�������2�����ӻ�4�����ӣ�����YΪ̼Ԫ�ػ���Ԫ�أ�X��Y���γɻ�����X2Y3����YΪ��Ԫ�أ�X��Y��Z����Ԫ�ص�ԭ������֮�͵���42����Z��������Ϊ42-8-33=1����ZΪ��Ԫ�أ���ԭ�ӿ����γɸ�һ�����ӣ��������⣮
��1��XԪ��ԭ�ӵ�4p�������3��δ�ɶԵ��ӣ�XԪ��ԭ�ӵĺ�������Ų�ʽΪ 1s22s22p63s23p63d104s24p3����XΪAs��
�ʴ�Ϊ��1s22s22p63s23p63d104s24p3��As��
��2��ZΪHԪ�أ�Ԫ�������ڱ�����s���������Ԫ����뭡�뮡��3����ͬλ�أ�
�ʴ�Ϊ��s��3��
��3��X��Z���γɻ�����XZ3ΪAsH3���백�����ӽṹ���ƣ��û�����Ŀռ乹��Ϊ�������Σ�
�ʴ�Ϊ�������Σ�
��4��������X2Y3ΪAs2O3��XZ3ΪAsH3��As2O3��ϡ������Һ�пɱ�����п��ԭΪAsH3�����ﻹ��ZnSO4��H2O����Ӧ��ZnԪ�ػ��ϼ���0������Ϊ+2�ۣ�AsԪ�ػ��ϼ���+3�۽���Ϊ-3�ۣ��μӷ�Ӧ��Zn��As2O3�����ʵ���֮��Ϊ6��2��2=6��1����Ӧ����ʽΪAs2O3+6Zn+6H2SO4�T2AsH3��+6ZnSO4+3H2O��
�ʴ�Ϊ��As2O3+6Zn+6H2SO4�T2AsH3��+6ZnSO4+3H2O��
��5��AsH3��NH3��PH3�У�����N-H��P-H��As-H������Խ�̣�����Խ�������ȶ��������ȶ���NH3��PH3��AsH3����Ϊ���Ӿ��壬NH3����֮�����������е���ߣ�AsH3��Է�����������PH3��AsH3���Ӽ���������ǿ����е����PH3���ʷе㣺NH3��AsH3��PH3��
�ʴ�Ϊ��NH3��PH3��AsH3��NH3��AsH3��PH3��
���� �����Ƕ����ʽṹ�Ŀ��飬�漰�ṹ��λ�ù�ϵ����������Ų����ɡ�������ԭ��Ӧ��Ԫ�������ɵȣ��Ѷ��еȣ��ƶ�Ԫ��Y�ǹؼ���ע�⣨4����������ԭ��Ӧ����ʽ����д��ƽ��


| A�� | ����ʽΪC8H6O3 | |
| B�� | �Ҵ���������һ�������¾���������ᷢ��������Ӧ | |
| C�� | 1mol���������4molH2�����ӳɷ�Ӧ | |
| D�� | 1mol��������NaOH��Һ��Ӧ�������2mol NaOH |
 �������ʺڼ��أ��ݱ���������������������ٽ�˯�ߵȹ��ܣ���ṹ��ʽΪ�����й����˵������ȷ���ǣ�������
�������ʺڼ��أ��ݱ���������������������ٽ�˯�ߵȹ��ܣ���ṹ��ʽΪ�����й����˵������ȷ���ǣ�������| A�� | �ܷ����ӳɷ�Ӧ | B�� | ���ڷ����廯���� | ||
| C�� | ����ʽΪC14C16N2O2 | D�� | ����ʹ�������������Һ��ɫ |
| A�� | ��ú��Һ������ָ��ú���ȱ�ΪҺ̬ | |
| B�� | ���Ҵ����͡���Ϊ������ȼ�յ���ֵ���������м����Ҵ� | |
| C�� | �������Ҫ�õ�������Ԫ����һ�ָ���Ľ���Ԫ�� | |
| D�� | ��ȼ�ϵ�ȼ�շ�Ӧ���Ϊԭ��أ�������������������� |
| A�� | �廯������ɫ���壬�۵�98�棬����̬�����磩 | |
| B�� | ̼��������ɫ���壬�۵�2200�棬����̬�����磩 | |
| C�� | ����������ɫ���壬����ʱ������ˮʱ�ƻ�����ͬ���������� | |
| D�� | �����������ɫ���壬�۵�19.5�棬�������Ҵ��� |
| A�� | ������ˮ�ķ�Ӧ��C12+H2O=2H++Cl-+ClO- | |
| B�� | ����������ʵ���Ũ�ȵ�����������Һ��̼�������Һ��ϣ�Ba2++2OH-+NH4++HCO3-=BaCO3��+NH3•H2O+H2O | |
| C�� | ̼������ڴ��CaCO3+2H+=Ca2++CO2��+H2O | |
| D�� | ����������������Һ��Ӧ��Ba2++OH-+SO42-+H+=BaSO4��+H2O |
 ��ͼΪ��100mL 2mol/L AlCl3��Һ�м���2mol/L NaOH��Һ�����У�����Al��OH��3��ɫ������������NaOH�����ʵ���֮��Ĺ�ϵ���ߣ��Իش�
��ͼΪ��100mL 2mol/L AlCl3��Һ�м���2mol/L NaOH��Һ�����У�����Al��OH��3��ɫ������������NaOH�����ʵ���֮��Ĺ�ϵ���ߣ��Իش�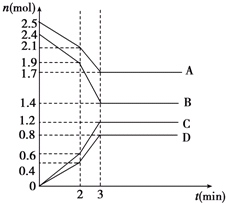 ��ijһ�ݻ�Ϊ2L�ĺ����ܱ������У�A��B��C��D�������ʵ����ʵ���n��mol����ʱ��t��min���ı仯������ͼ��ʾ���ش��������⣺
��ijһ�ݻ�Ϊ2L�ĺ����ܱ������У�A��B��C��D�������ʵ����ʵ���n��mol����ʱ��t��min���ı仯������ͼ��ʾ���ش��������⣺