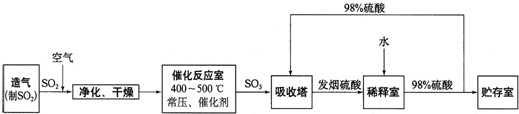
��Ŀ����
�ж�����˵����ȷ���ǣ� ��
A��1molFeCl3��ˮ��ȫ��Ӧת��Ϊ�����������壬���н������ӵ����ʵ���Ϊ1mol
B��һ���¶��£�1L0.50mol/L NH4Cl ��Һ��2L0.25mol/LNH4Cl��Һ���е�NH4+�����ʵ�����ͬ
C������480mL 0.5mol/L ��CuSO4��Һ����Ҫ��ȡ60.0g��CuSO4��5H2O����
D��10mL��������Ϊ98% ��H2SO4��Һ����ˮϡ����100mL��������������Ϊ9.8%
(ע��������Һ���ܶ���Ũ�ȵ����������)
B
����:
��

��ϰ��ϵ�д�
 ��ʦ������Ԫ��ĩ���100��ϵ�д�
��ʦ������Ԫ��ĩ���100��ϵ�д�
�����Ŀ
��2010?��������ģ��
|
���ֶ�����Ԫ�ص�ԭ�Ӱ뾶��ijЩ���ϼۼ��±��������ж�����˵����ȷ���ǣ�������
|
��֪25�桢101kPa�£�ʯī�ͽ��ʯȼ�յ��Ȼ�ѧ����ʽ��
C��ʯī��+O2��g��=CO2��g����H=-393.51kJ?mol��1
C�����ʯ��+O2��g��=CO2��g����H=-395.41kJ?mol��1
�ݴ��ж�����˵����ȷ���ǣ�������
C��ʯī��+O2��g��=CO2��g����H=-393.51kJ?mol��1
C�����ʯ��+O2��g��=CO2��g����H=-395.41kJ?mol��1
�ݴ��ж�����˵����ȷ���ǣ�������
| A��ʯī�Ƚ��ʯ�ȶ� | B�����ʯ��ʯī�ȶ� | C��ȼ�յ�����ʱʯī�ķų������Ƚ��ʯ�� | D��ȼ�յ�����ʱʯī�ķų���������ʯһ���� |