��Ŀ����
7����þ���仯�����ڹ�ũҵ������Ӧ�ù㷺����֪����þ����Ҫ�ɷ�ΪMg2B2O5•H2O����ɰ�Ļ�ѧʽΪNa2B4O7•10H2O��������þ����ȡ����þ������Ĺ�������Ϊ��
�ش������й����⣺
��1����ɰ��B�Ļ��ϼ�Ϊ+3��������ˮ����H2SO4��pH2��3��ȡH3BO3����Ӧ�����ӷ���ʽΪ3Mg+B2O3$\frac{\underline{\;����\;}}{\;}$2B+3MgO��XΪH3BO3���������ˮ�IJ������Mg��ȡ����Ļ�ѧ����ʽΪ3Mg+B2O3$\frac{\underline{\;����\;}}{\;}$2B+3MgO��
��2��MgCl2•7H2O��Ҫ��HCl��Χ�м��ȣ���Ŀ���Ƿ�ֹMgCl2ˮ������Mg��OH��2��
��3��þ-H2O2����ȼ�ϵ�صķ�ӦΪMg+H2O2+2H+=Mg2++2H2O����������ӦʽΪH2O2+2H++2e-=2H2O����֪Ksp[Mg��OH��2]=5.6��10-12������ҺpH=6ʱ�ޣ���С����ޡ���Mg��OH��2����������
��4���ƵõĴ�����һ������������BI3��BI3���ȷֽ���Եõ������ĵ������ֽ�0.020g�����Ƴɵ�BI3��ȫ�ֽ⣬���ɵ�I2��0.30mol•L-1 Na2S2O3��Һ�ζ����յ㣬����Na2S2O3��Һ18.00mL����ȡ����Һʹ�õ�����ӦΪ��ʽ�ζ��ܣ����ʽ����ʽ�������ô�����Ʒ�Ĵ���Ϊ97.2% ����ʾ��I2+2S2O32-=2I-+S4O62-����
��5���绯ѧ�����ڹ�ҵ����������Ҫ�������⣬����ˮ�Ĵ�����Ҳ�кܴ�������磺
�������ƺͶ��ȷӣ�
 ���ķ�ˮ�������������س�ȥ����ԭ����ͼ2��ʾ��
���ķ�ˮ�������������س�ȥ����ԭ����ͼ2��ʾ����B�ǵ�صĸ��������������������A���ĵ缫��ӦʽΪ
 +2e-+H+�T
+2e-+H+�T +Cl-��
+Cl-��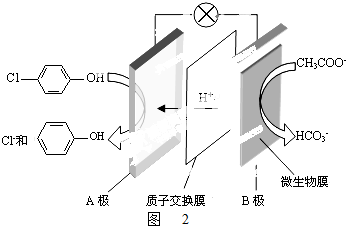
���� ��þ����Ҫ�ɷ�ΪMg2B2O5•H2O����ɰ�Ļ�ѧʽΪNa2B4O7•10H2O��������þ����ȡ����þ������Ĺ�����������þ�������������Ũ��Һ���˵õ��Ȼ�þ������Ũ�����ܽ�ͨ������Ũ���õ��Ȼ�þ�ᾧˮ������Ȼ��������м��ȵõ��Ȼ�þ���壬���õ�þ����Һ����Ҫ��NaBO2��ͨ������������̼����õ���ɰ�������Ⱥ���H2SO4��pH2��3��ȡH3BO3�����ȵõ�B2O3��
��1����ɰ�Ļ�ѧʽΪNa2B4O7•10H2O������Ԫ�ػ��ϼ۱�ע������Ԫ�ػ��ϼۣ���H2SO4��pH2��3����ɰ�е�Na2B4O7������Һ������H3BO3 ��XΪH3BO3���������ˮ�IJ���õ�B2O3��þ�͵õ�B2O3��Ӧ��������þ����
��2���Ȼ�þ��ˮ��Һ��ˮ������������þ��
��3��ȼ�ϵ�����������ǹ�������õ���������ˮ��PH=6��������������Ũ�ȣ��������þ����Ũ�ȼ���Ũ���̺��ܶȻ������ȽϷ����Ƿ�����������þ������
��4��H2S2O3Ϊ���ᣬNa2S2O3��Һ�Լ��ԣ��ݴ�ѡ��ζ��ܣ����ݹ�ϵʽB��BI3��$\frac{3}{2}$I2��3S2O32-���ζ����ݼ������������ĺ�����
��5���ٸ���ԭ����������ӵ��ƶ�����ȷ����������
�ڸ��������Ϸ����õ��ӵĻ�ԭ��Ӧ����д��
��� �⣺��1����ɰ�Ļ�ѧʽΪNa2B4O7•10H2O����Ԫ�ػ��ϼ�Ϊ+1�ۣ���Ԫ�ػ��ϼ�-2�ۣ����ݻ��ϼ۴����ͼ���õ���Ԫ�ػ��ϼ�Ϊ+3�ۣ���H2SO4��pH2��3����ɰ�е�Na2B4O7������Һ������H3BO3 ����Ӧ�����ӷ���ʽΪ��B4O72-+2H++5H2O=4H3BO3��XΪH3BO3���������ˮ�IJ�����B2O3��þ�͵õ�B2O3��Ӧ��������þ���𣬷�Ӧ�Ļ�ѧ����ʽΪ��3Mg+B2O3$\frac{\underline{\;����\;}}{\;}$2B+3MgO����Ӧ��Ԫ�ػ��ϼ۽��͵�������������Ԫ�ػ��ϼ�+3�۱仯Ϊ0�ۣ���B2O3����������
�ʴ�Ϊ��+3��B4O72-+2H++5H2O=4H3BO3��3Mg+B2O3$\frac{\underline{\;����\;}}{\;}$2B+3MgO��
��2��MgCl2•7H2O��Ҫ��HCl��Χ�м��ȣ���Ϊ�˷�ֹ�Ȼ�þˮ������������þ��
�ʴ�Ϊ����ֹMgCl2ˮ������Mg��OH��2��
��3��þ-H2O2����ȼ�ϵ�صķ�Ӧ����ΪMg+H2O2+2H+�TMg2++2H2O���������ǹ�������õ���������ˮ�ķ�Ӧ��������ӦʽH2O2+2H++2e-=2H2O��Ksp[Mg��OH��2]=5.6��10-12������ҺpH=6ʱ��c��OH-��=10-8mol/L����Qc=c��Mg2+����c2��OH-��=0.045mol/L��10-16mol/L=4.5��10-18��Ksp[Mg��OH��2]��˵����������þ�������ɣ�
�ʴ�Ϊ��H2O2+2H++2e-=2H2O���ޣ�
��4��H2S2O3Ϊ���ᣬNa2S2O3��Һ�Լ��ԣ��ݴ�ѡ��ζ���ӦΪ��ʽ�ζ��ܣ�
��������Ƶ����ʵ���Ϊ��0.30mol/L��0.018L=0.0054mol��I2+2S2O32-=2I-+S4O62-��
���ݹ�ϵʽ��B��BI3��$\frac{3}{2}$I2��3S2O32-��
n��B��=$\frac{1}{3}$n��S2O32-��=0.0018mol��
�������Ϊ��10.81g/mol��0.0018mol=0.01944g����������ĺ���Ϊ��$\frac{0.01944g}{0.0200g}$��100%=97.2%��
�ʴ�Ϊ����ʽ��97.2��
��5����ԭ����������ӵ��ƶ������ǴӸ�����������������B�ǵ�صĸ�����
�ʴ�Ϊ������
��A�������������Ϸ����õ��ӵĻ�ԭ��Ӧ�� +2e-+H+�T
+2e-+H+�T +Cl-��
+Cl-��
�ʴ�Ϊ�� +2e-+H+�T
+2e-+H+�T +Cl-��
+Cl-��
���� ���⿼���������ˮ�⡢ԭ���ԭ���͵���ԭ���ķ����������ܽ�ƽ��ļ���Ӧ�ã���Ŀ�Ѷ��Դ������ѵ㣬����ʱҪ���������Ŀ����������ϵ�ͱ������ݣ�����������ϵ�����������ĸ�����ٽ��⣮

 ������״Ԫ���Ծ�ϵ�д�
������״Ԫ���Ծ�ϵ�д��� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |

��2���ǽ�������ǿ��Ԫ���Ƿ�����Ԫ�����ƣ�
��3��д���٢߶�Ӧ����Ԫ�صķ��ŷֱ�ΪN��Cl������Ԫ�ط��ţ�Ԫ�آۢݵ�����������Ӧ��ˮ�������Ӧ�����ӷ���ʽΪAl��OH��3+OH-=AlO2-+H2O
��4����ЩԪ���е�����������Ӧ��ˮ�����У�������ǿ���ǣ����Ӧ������Ļ�ѧʽ����ͬ��HClO4��������ǿ����KOH�������Ե�����������Al��OH��3��
��5��д��ȷ��ijһ��Һ�к���Ԫ�آ���ڵ�ʵ��������ýྻ�IJ�˿պȡ�������������ھƾ����������գ�����ɫ�ܲ����۲�������ɫ���������ɫ��˵������������K+��
| A�� | 90 | B�� | 27 | C�� | 10.8 | D�� | 9 |
 ��֪������ˮ�Ȼ��ƿ����Ҵ��γ�������ˮ��CaCl2•6C2H5OH��
��֪������ˮ�Ȼ��ƿ����Ҵ��γ�������ˮ��CaCl2•6C2H5OH�����й��л���ķе������ʾ��
| �Լ� | ���� | �Ҵ� | ���� | �������� |
| �е�/�� | 34.7 | 78.5 | 118 | 77.1 |
�Իش��������⣺
��1��Ũ��������ã���ˮ�����������������Ӧ�м���������Ҵ���Ŀ������������ת���ʣ�
��2������ͬλ��18Oʾ�ٷ�ȷ������ˮ��������ԭ�ӵ��ṩ�ߣ�д���ܱ�ʾ18Oλ�õĻ�ѧ����ʽ��CH3CO18OH+CH3CH2OH
 CH3COOCH2CH3+H218O��
CH3COOCH2CH3+H218O����3������A�������Ƿ�Һ©����C��������������ȴ�����ͷ�ֹ����������Ӧǰ��D�м��뼸�η�̪����Һ�ʺ�ɫ�������������ԭ���ǣ������ӷ���ʽ��ʾ��CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-��ʵ����Ϻ��ձ�D����Һ�������ֹ������Ϊ��Һ�ֲ㣬�ϲ�Ϊ��ɫ��״Һ�壬�²���Һ��ɫ��dz������ȥ����
��4����D�з���������������л�����һ�������Ҵ������Ѻ�ˮ��Ӧ�ȼ�����ˮ�Ȼ��ƣ�������Ҵ��������ƣ����ټ���ˮ�����ƣ�Ȼ�������������Ϳ��Եõ��ϴ���������������
| A�� | ��ҵ�ϳ��õ������D�����������Ʊ�D�ĵ��� | |
| B�� | ����C��D�Ż����ʱC��������ĭ���������D��������ĭ�������� | |
| C�� | ������A2B2��C2B2������ѧ��������ȫ��ͬ | |
| D�� | A��B��C��ɵĻ��������ҺŨ��Ϊ0.01mol/L����������PHΪ12 |
��1����ͬѧ�����ʵ��������������£�
| ��� | ���� | ���� |
| I | ����2mL 0.1mol/L FeCl2��Һ�еμ�KSCN��Һ���ٵμ�������ˮ | |
| II | ����2mL 0.1mol/L FeCl3��Һ�еμ�KSCN��Һ���ٵμ�0.1mol/L KI��Һ |
��FeCl2��Һ��������ˮ��Ӧ�����ӷ���ʽΪ2Fe2++Cl2=2Fe3++2Cl-��
��2����ͬѧ�ڽ���ʵ��Iʱ���������ˮ���࣬���³��ֵĺ�ɫ����ȥ������ܵ�ԭ���ǹ�����ˮ��KSCN��Fe3+��������

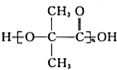
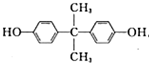 ��G��һ�ָ߷��ӣ����������������۾��IJ��ϣ�HΪ��Ԫ������
��G��һ�ָ߷��ӣ����������������۾��IJ��ϣ�HΪ��Ԫ������ ��
�� ��
�� ��
��
 ��MnO2��Ũ������ȡ�������������������������ͭ�۷�Ӧ��ȡ��������ˮCuCl2��װ����ͼ��ʾ��
��MnO2��Ũ������ȡ�������������������������ͭ�۷�Ӧ��ȡ��������ˮCuCl2��װ����ͼ��ʾ��
 ��
�� ��
��