��Ŀ����
7��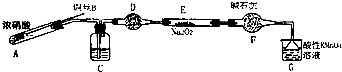 ����������һ�ֹ�ҵ�Σ������ʳ�ηdz����ƣ����Խ�ǿ��ij��ѧ��ȤС���ʳ�����������ƽ��ж�Ƕ�̽����
����������һ�ֹ�ҵ�Σ������ʳ�ηdz����ƣ����Խ�ǿ��ij��ѧ��ȤС���ʳ�����������ƽ��ж�Ƕ�̽������һ������NaCl��NaNO2
��ͬѧ�ó�����������
���飺������Ksp��AgNO2��=2��10-8��Ksp��AgCl��=1.8��10-10���ֱ���ʢ��5mL 0.0001mol/L��������Һ���Ժ���ͬʱ��εμ�0.0001mol/L��������Һ�������ɳ�������װ��NaCl��Һ���Թܣ�
��ͬѧ�ඨ��ҺpH��
��pH��ֽ�ֱ�ⶨ0.1mol/L��������Һ��pH�����NaNO2��Һ�ʼ��ԣ�����Һ�ʼ��Ե�ԭ����NO2-+H2O?HNO2+OH-�������ӷ���ʽ���ͣ���
��������С������ͼװ�ã���ȥ�г��������Ʊ���������
��֪����2NO+Na2O2=2NaNO2��
�����������£�NO��NO2������MnO4һ��Ӧ����NO3һ��Mn2+•
��1��ʹ��ͭ˿���ŵ��ǿ��Կ��Ʒ�Ӧ�ķ�����ֹͣ��
��2��װ��A�з�Ӧ����ʽΪCu+4HNO3��Ũ��=Cu��NO3��2+2NO2��+2H2O��
װ��C ��ʢ�ŵ�ҩƷ��C��������ĸ���ţ�
A��Ũ���� B��NaOH ��Һ C��ˮ D�����Ȼ�̼
����F�����÷�ֹˮ�������룮
��3����С���ȡ5.000g��ȡ����Ʒ����ˮ���250.0mL��Һ��ȡ25.00mL��Һ����ƿ�У���0.1000mol/L����KMnO4��Һ���еζ���ʵ���������������ʾ��
| ����� | 1 | 2 | 3 | 4 |
| ����KMnO4��Һ���/mL | 20.70 | 20.12 | 20.00 | 19.88 |
a����ƿϴ����δ����
b����ʽ�ζ���������ˮϴ����δ�ñ�Һ��ϴ
c���ζ��յ�ʱ���Ӷ���
������KMnO4��Һ�ζ�����������Һ�����ӷ���ʽΪ2MnO4-+5NO2-+6H+=2Mn2++5NO3-+3H2O��
�۸���Ʒ���������Ƶ���������Ϊ69.0%��
���� ��һ�������ܶȻ�С�ij����ȳ�����NaNO2˵��Ϊǿ�������Σ������������ˮ�����Һ��ʾ���ԣ�
������װ��A����Ũ�����ͭ���ȷ����ķ�Ӧ����Ӧ���ɶ�������������ͭ��ˮ��װ��C����Aװ�����ɵĶ���������ˮ��Ӧ���������һ��������ͨ��װ��D�еĸ��������ˮ������ͨ��װ��F�еĸ������ֹˮ�������룬ͨ��װ��E�еĹ���������һ��������Ӧ�����ͨ�����Ը��������Һ��ȥʣ��һ��������ֹ��Ⱦ������
��1����ͨ�������ƶ�ͭ˿���Ʒ�Ӧ�Ƿ���У�
��2��Ũ�����ͭ���ȷ�����Ӧ���ɶ�������������ͭ��ˮ��װ��C����Aװ�����ɵĶ���������ˮ��Ӧ���������һ��������ͨ��װ��F�еĸ������ֹˮ�������룮
��3���ٸ���c�����⣩=$\frac{c����ע����V������}{V�����⣩}$��������������V��������Ӱ�죬�Դ��ж�Ũ�ȵ���
�ڸ÷�Ӧ������������ӱ�����Ϊ��������ӣ�����������ӱ���ԭΪ�����ӣ�
�۸��ݷ���ʽ5NO2-+2MnO4-+6H+=5NO3-+2Mn2++3H2O������������Ƶ����ʵ�����Ȼ������Ʒ���������Ƶ�����������
��� �⣺��һ��Ksp��AgNO2��=2��10-8��Ksp��AgCl��=1.8��10-10���ֱ���ʢ��5mL 0.0001mol/L��������Һ���Ժ���ͬʱ��εμ�0.0001mol/L��������Һ��������AgCl������
�ʴ�Ϊ��NaCl��NO2-+H2O?HNO2+OH-��
��������1����ͨ�������ƶ�ͭ˿���Ʒ�Ӧ�Ƿ���У�����ʹ��ͭ˿���ŵ��ǿ��Կ��Ʒ�Ӧ�ķ�����ֹͣ��
�ʴ�Ϊ�����Կ��Ʒ�Ӧ�ķ�����ֹͣ��
��2��Ũ�����ͭ���ȷ�����Ӧ���ɶ�������������ͭ��ˮ������ʽΪ��Cu+4HNO3��Ũ��=Cu��NO3��2+2NO2��+2H2O��ͨ��װ��C�е�ˮ�����������Ӧ���������һ��������ͨ��װ��F�еĸ������ֹˮ�������룬��������Ʒ�Ӧ��
�ʴ�Ϊ��Cu+4HNO3��Ũ��=Cu��NO3��2+2NO2��+2H2O��C����ֹˮ�������룻
��3����a����ƿϴ����δ�������Һ�����ʵ������䣬��V��������Ӱ�죬����c�����⣩=$\frac{c����ע����V������}{V�����⣩}$������c���������䣬��a����
b����ʽ�ζ���������ˮϴ����δ�ñ�Һ��ϴ����Һ��Ũ��ƫС�����V������ƫ����c�����⣩=$\frac{c����ע����V������}{V�����⣩}$������c������ƫ��b��ȷ��
c���ζ��յ�ʱ���Ӷ��������V������ƫ����c�����⣩=$\frac{c����ע����V������}{V�����⣩}$������c������ƫ��c��ȷ��
��ѡbc��
�ڸ÷�Ӧ������������ӱ�����Ϊ��������ӣ�����������ӱ���ԭΪ�����ӣ����ӷ���ʽΪ2MnO4-+5NO2-+6H+=2Mn2++5NO3-+3H2O��
�ʴ�Ϊ��2MnO4-+5NO2-+6H+=2Mn2++5NO3-+3H2O��
�����ĸ�����ص����ʵ�����0.1mol/L��0.02L=0.002mol������ݷ���ʽ5NO2-+2MnO4-+6H+=5NO3-+2Mn2++3H2O��֪���������Ƶ����ʵ�����0.002mol��$\frac{5}{2}$=0.005mol����ԭ��Ʒ���������Ƶ����ʵ�����0.005mol��$\frac{100mL}{25mL}$=0.02mol��������Ϊ0.02mol��69g/mol=1.38g��������Ʒ���������Ƶ���������$\frac{1.38g}{2g}$��100%=69.0%��
�ʴ�Ϊ��69.0%��
���� ���⿼������ˮ�⡢��������ʡ�β��������ʵ���������ơ�ʵ�鷽������������Լ����ʺ����ⶨ�ȣ�ע��ʵ������еķ�Ӧ������������ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

| A�� | 1 | B�� | l-a | C�� | 2-lga | D�� | 3-lga |
| A�� | 50mL18.4mol/LŨ����������ͭ�ȷ�Ӧ������SO2���ӵ���ĿΪ0.46NA | |
| B�� | ij�ܱ�����ʢ��0.1mol N2��0.3mol H2����һ�������³�ַ�Ӧ��ת�Ƶ��ӵ���ĿΪ0 6NA | |
| C�� | ����CO2��O2��ɵĻ�����й���NA�����ӣ������е���ԭ����Ϊ2 NA | |
| D�� | �����£�1L 0�� 1 mol/L NH4NO3��Һ�е���ԭ����Ϊ0.4 NA |
| A�� | ��ԭFe�� | B�� | FeO | C�� | Fe3O4 | D�� | Fe2O3 |
| A�� | ԭ�Ӱ뾶��W��Z��Y | |
| B�� | ��ҵ��ͨ�����Y����ˮ�����Ʊ�Y�ĵ��� | |
| C�� | ����������Ӧˮ��������ԣ�W��Z��X | |
| D�� | ��Ԫ�طֱ���X��Y��Z��W�γɵĻ����ﶼ��ֹһ�� |
| A�� | CH3CH2Br������ˮ | |
| B�� | �÷�Ӧ����ȡ����Ӧ | |
| C�� | CH3CH2Br��HBr�����ڵ���� | |
| D�� | ��ԭ����ȫ��Ӧ�����������CH3CH2Br��NH3�����ʵ���֮��Ϊ2��1 |
| A�� | �����ݲ�������������ʹ�����ǵĻ��ȼ | |
| B�� | ��Ӧ����Һ���о������� | |
| C�� | ��Ӧǰ����Һ�������ӵ�Ũ�Ȳ��� | |
| D�� | ��Ӧǰ����Һ�������ӵ��������� |
 �������и��������У�����֮��ͨ��һ����Ӧ����ʵ����ͼ��ʾת�����ǣ�������
�������и��������У�����֮��ͨ��һ����Ӧ����ʵ����ͼ��ʾת�����ǣ�������| ���� ѡ�� | a | b | c |
| A | Al | AlCl3 | Al��OH��3 |
| B | HNO3 | NO | NO2 |
| C | Si | SiO2 | H2SiO3 |
| D | H2SO4 | SO2 | SO3 |
| A�� | A | B�� | B | C�� | C | D�� | D |