��Ŀ����
12��X��Y��Z��WΪԭ��������������Ķ���������Ԫ�أ�������������֮��Ϊ20��X�ǵڶ�����Ԫ�أ�Zԭ����������������Xԭ���ڲ����������3��������Yԭ��������������2��������˵����ȷ���ǣ�������| A�� | ԭ�Ӱ뾶��W��Z��Y | |
| B�� | ��ҵ��ͨ�����Y����ˮ�����Ʊ�Y�ĵ��� | |
| C�� | ����������Ӧˮ��������ԣ�W��Z��X | |
| D�� | ��Ԫ�طֱ���X��Y��Z��W�γɵĻ����ﶼ��ֹһ�� |
���� X��Y��Z��W��ԭ��������������Ķ���������Ԫ�أ�X�ǵڶ�����Ԫ�أ�Zԭ����������������Xԭ���ڲ����������3��������Yԭ��������������2������Zԭ������������Ϊ6��Yԭ������������Ϊ3����ԭ��������������֮��Ϊ20����X��Wԭ������������֮��Ϊ20-6-3=11������������Ϊ4��7������X��ԭ��������С��Wԭ�����������֪XΪCԪ�ء�YΪAl��ZΪS��WΪCl��
��� �⣺X��Y��Z��W��ԭ��������������Ķ���������Ԫ�أ�X�ǵڶ�����Ԫ�أ�Zԭ����������������Xԭ���ڲ����������3��������Yԭ��������������2������Zԭ������������Ϊ6��Yԭ������������Ϊ3����ԭ��������������֮��Ϊ20����X��Wԭ������������֮��Ϊ20-6-3=11������������Ϊ4��7������X��ԭ��������С��Wԭ�����������֪XΪCԪ�ء�YΪAl��ZΪS��WΪCl��
A��ͬ�����������ԭ�Ӱ뾶��С����ԭ�Ӱ뾶��Y��Al����Z��S����W��Cl������A����
B����ҵ��ͨ���������������ұ��Al����B����
C���ǽ�����W��Cl����Z��S����X��C��������������Ӧˮ��������ԣ���������̼�ᣬ��C��ȷ��
D����Ԫ����Y��Al��ֻ���γ�����������D����
��ѡ��C��
���� ���⿼��ṹ����λ�ù�ϵӦ�ã��ƶ�Ԫ���ǽ���ؼ����������պ�������Ų���������Ԫ�����ڱ��ṹ�����ؿ�������ƶ�������

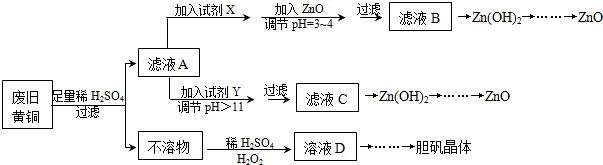
��֪��Zn���������������Al����������������ƣ�pH��11ʱZn��OH��2������NaOH��Һ����[Zn��OH��4]2-������г��˼������������������������pH����ʼ������pH����������Ũ��Ϊ1.0mol•L-1���㣩��
| Fe3+ | Fe2+ | Zn2+ | |
| ��ʼ������pH | 1.1 | 5.8 | 5.9 |
| ������ȫ��pH | 3.0 | 8.8 | 8.9 |
��1���Լ�X������H2O2���������ǽ�Fe2+����ΪFe3+��
��2������ZnO����pH=3��4��Ŀ���ǽ���H+Ũ�ȣ���ʹFe3+����ˮ������ Fe��OH��3��������ȥ��
��3���ɲ�����������ҺD�Ļ�ѧ����ʽΪCu+H2O2+H2SO4=CuSO4+2H2O��
��4������ҺD�Ƶ��������������Ҫ��������������Ũ������ȴ�ᾧ�����ˣ�
��5�������Լ�����ΪY�Լ�����B��A��ZnO B��NaOH C��Na2CO3 D��ZnSO4
������ҺC����μ�������ֱ����������������������Ȳ�����ɫ�������ܽ⣮
��6���ⶨ��������Ĵ��ȣ���������I-������Ӧ�����������ʣ���ȷ��ȡ0.5000g��������������ƿ�У�������ˮ�ܽ⣬�ټ������KI����0.1000mol•L-1Na2S2O3����Һ�ζ����յ㣬����Na2S2O3����Һ19.40mL����֪�������ζ������е����ӷ���ʽ���£�Cu2++4I-�T2CuI����ɫ����+I2��I2+2S2O32-�T2I-+S4O62-
�ٵ�������Ĵ���Ϊ97.00%��
���ڵζ������о���ҡ������Һ���⽦����ƿ��������õĴ��Ƚ���ƫ�ߣ��ƫ�ߡ�����ƫ�͡����䡱����
| A�� | ������ | B�� | �������� | C�� | ������������ | D�� | ���Ȼ���ϡ��Һ |
 ����������һ�ֹ�ҵ�Σ������ʳ�ηdz����ƣ����Խ�ǿ��ij��ѧ��ȤС���ʳ�����������ƽ��ж�Ƕ�̽����
����������һ�ֹ�ҵ�Σ������ʳ�ηdz����ƣ����Խ�ǿ��ij��ѧ��ȤС���ʳ�����������ƽ��ж�Ƕ�̽������һ������NaCl��NaNO2
��ͬѧ�ó�����������
���飺������Ksp��AgNO2��=2��10-8��Ksp��AgCl��=1.8��10-10���ֱ���ʢ��5mL 0.0001mol/L��������Һ���Ժ���ͬʱ��εμ�0.0001mol/L��������Һ�������ɳ�������װ��NaCl��Һ���Թܣ�
��ͬѧ�ඨ��ҺpH��
��pH��ֽ�ֱ�ⶨ0.1mol/L��������Һ��pH�����NaNO2��Һ�ʼ��ԣ�����Һ�ʼ��Ե�ԭ����NO2-+H2O?HNO2+OH-�������ӷ���ʽ���ͣ���
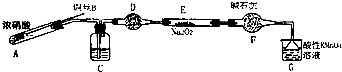
��������С������ͼװ�ã���ȥ�г��������Ʊ���������
��֪����2NO+Na2O2=2NaNO2��
�����������£�NO��NO2������MnO4һ��Ӧ����NO3һ��Mn2+•
��1��ʹ��ͭ˿���ŵ��ǿ��Կ��Ʒ�Ӧ�ķ�����ֹͣ��
��2��װ��A�з�Ӧ����ʽΪCu+4HNO3��Ũ��=Cu��NO3��2+2NO2��+2H2O��
װ��C ��ʢ�ŵ�ҩƷ��C��������ĸ���ţ�
A��Ũ���� B��NaOH ��Һ C��ˮ D�����Ȼ�̼
����F�����÷�ֹˮ�������룮
��3����С���ȡ5.000g��ȡ����Ʒ����ˮ���250.0mL��Һ��ȡ25.00mL��Һ����ƿ�У���0.1000mol/L����KMnO4��Һ���еζ���ʵ���������������ʾ��
| ����� | 1 | 2 | 3 | 4 |
| ����KMnO4��Һ���/mL | 20.70 | 20.12 | 20.00 | 19.88 |
a����ƿϴ����δ����
b����ʽ�ζ���������ˮϴ����δ�ñ�Һ��ϴ
c���ζ��յ�ʱ���Ӷ���
������KMnO4��Һ�ζ�����������Һ�����ӷ���ʽΪ2MnO4-+5NO2-+6H+=2Mn2++5NO3-+3H2O��
�۸���Ʒ���������Ƶ���������Ϊ69.0%��
| A�� | ��SO2Ư���IJ�ñ���վû��ɫ | |
| B�� | �轺��ף�������ʳƷ������ʹ��������� | |
| C�� | �����պ�����ζ�ķ���������֯��ʹ�ë֯�� | |
| D�� | ������������ˮ�����ļ���ɱ��Ч�����ڶ����� |
ijͬѧѡ������װ�����Ȼ����Ʊ������Ȼ�������װ�ò������ظ�ʹ�ã�����������֪���Ȼ�����ˮ����ˮ�⣬�ڼ���������������ԭ�Ȼ�������FeCl2��HCl��
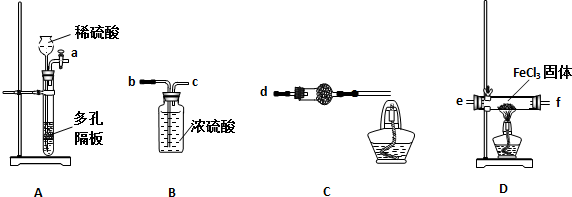
��1��ʵ�鿪ʼǰӦ�ȼ��װ�õ������ԣ�������װ��A�����Լ���ķ������رջ�������©����עˮ���γ�һ��ˮ����Һ��߶Ȳ��䣬˵��װ������������
��2�������������ң�װ�ýӿ�����˳����ab c e f d
��3��װ��C�и��������ʢ�Լ��������ǣ���ʯ�һ���ʯ�ң������ǣ���ȥHCl������ֹ������ˮ�Ľ���
��4��װ��C�оƾ��Ƶ������ǣ�ȼ��H2����ֹ��Ⱦ����
��5��Ϊ�˲ⶨ�Ȼ�����������������ijͬѧ����������ʵ�飺
��ȡʵ���Ʊ����Ȼ�������Ʒ5.435g�������Һ����1.00mol•L-1����KMnO4����Һ�ζ����յ㣮����֪��ԭ��Fe2+��Cl-���ζ���������ʾ��
| �ζ����� | ����Һ����� | |
| �ζ�ǰ�̶�/mL | �ζ���̶�/mL | |
| 1 | 1.04 | 25.03 |
| 2 | 1.98 | 25.99 |
| 3 | 3.20 | 25.24 |
����Ʒ���Ȼ���������������Ϊ70.1%��