��Ŀ����
1����98%��Ũ���ᣨ���ܶ�Ϊ1.84 g/cm������100mL 1.0mol•L-1ϡ���ᣬ������ʵ��������ѡ��A 100mL��Ͳ B ������ƽ C ������ D 50mL����ƿ
E 10mL��Ͳ F ��ͷ�ι� G.50mL�ձ� H 100mL����ƿ
��1������ȡŨ��������Ϊ5.4mL��
��2��ʵ��ʱ����ѡ�õ������У�����ţ�ABD
��3�����ƹ����У����������ʹ���ƽ��ƫ�ߵ��Ǣ٣�����ţ�
�ٶ���ʱ���ӿ̶��߹۲�Һ��
������ƿʹ��ʱδ����
�۶��ݺ���ҡ�ȡ����ã�����Һ����ڿ̶��ߣ��ټ�����ˮ�����̶���
��4��������ƿʹ�÷����У����в�����ȷ���ǣ�����ţ�AE
A��ʹ������ƿǰ������Ƿ�©ˮ
B������ƿ������ˮϴ�������ô���Һ��ϴ
C�����������ƹ��������ƽ���̵���ֽ�ϣ�ȷ�����������ձ����ܽ������ע������ƿ��
D����ȷ��ȡ��18.4mol•L-1�����ᣬע����ʢ��30mLˮ��100mL������ƿ�У���ˮ���̶���
E�����ݺ�����ƿ������ʳָ��סƿ��������һֻ�ֵ���ָ��סƿ�ף�������ƿ��תҡ�ȣ�
���� ��1���ȼ����Ũ��������ʵ���Ũ��Ϊc=$\frac{1000�Ѧ�}{M}$��Ȼ�������Һϡ�Ͷ���CŨVŨ=CϡVϡ�����㣻
��2���������Ʋ����Ǽ��㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��������Ҫ��������
��3������c=$\frac{n}{V}$��������ʵ����ʵ���n����Һ�����V�ı仯��������������
��4��A��ʹ������ƿǰҪ������Ƿ�©ˮ����ֹ��������Һ������©Һ��
B������ƿ������ˮϴ�������ô���Һ��ϴ���ᵼ��������ҺŨ��ƫ��
C�������������ܽ�����л���ȣ�Ҫ����ȴ��������ת�ƽ�����ƿ��
D������ƿ����������Һ��ϡ�ͣ�
E��ҡ��ʱʳָ��סƿ��������һֻ�ֵ���ָ��סƿ�ף�������ƿ��ת��������ҡ�ȣ�
��� �⣺��1��Ũ��������ʵ���Ũ��Ϊc=$\frac{1000�Ѧ�}{M}$=$\frac{1000��1.84��98%}{98}$���������Ũ��������ΪVmL��������Һϡ�Ͷ���CŨVŨ=CϡVϡ��֪��18.4mol/L��VmL=1mol/L��100mL�����V=5.4mL���ʴ�Ϊ��5.4mL��
��2���������Ʋ����Ǽ��㡢��ȡ��ϡ�͡���ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��֪����������У�10mL��Ͳ���ձ�����������100mL����ƿ�ͽ�ͷ�ιܣ��ʲ�ѡ�ø�������У�ABD���ʴ�Ϊ��ABD��
��3���ٶ���ʱ���ӿ̶��߹۲�Һ�棬����Һ���ƫС��Ũ��ƫ�ߣ��ʢ���ȷ��
������ƿʹ��ʱδ�������ҺŨ����Ӱ�죬�ʢڴ���
�۶��ݺ���ҡ�ȡ����ã�����Һ����ڿ̶����������ģ��ټ�����ˮ�����̶��ߣ���Ũ��ƫ�ͣ��ʢ۴���
��ѡ�٣�
��4��A��ʹ������ƿǰҪ������Ƿ�©ˮ����ֹ��������Һ������©Һ����A��ȷ��
B������ƿ������ˮϴ�������ô���Һ��ϴ���ᵼ��������ҺŨ��ƫ��B����
C�������������ܽ�����л���ȣ�Ҫ����ȴ��������ת�ƽ�����ƿ����C����
D������ƿ�������ȣ��ʲ���������Һ��ϡ�ͣ�������Һʱ��Ҫ���ܽ�ϡ�ͣ���ȴ�������Ժ���ת��������ƿ����D����
E��ҡ��ʱʳָ��סƿ��������һֻ�ֵ���ָ��סƿ�ף�������ƿ��ת��������ҡ�ȣ���E��ȷ��
��ѡ��AE��
���� ���⿼����������ƿ����һ�����ʵ���Ũ����Һ��ʵ�������ע������ѶȲ���ʵ���������������ڶද�ֲ������ද��˼����

| A�� | 0.500 mol/L | B�� | 1.00 mol/L | C�� | 2.00 mol/L | D�� | 3.00 mol/L |
| A�� | 1 mol•L-1 | B�� | 2 mol•L-1 | C�� | 3 mol•L-1 | D�� | 4 mol•L-1 |
| A�� | ʹ�÷�Һ©��������ƿʱ������Ҫ��© | |
| B�� | ����ʵ���У�Ҫ����ƿ�м��뼸����ʯ�����Ƭ���Է�ֹҺ�屩�� | |
| C�� | ���Թ��м�Һ��ʱ����ͷ�ιܽ����Թ��ڱ� | |
| D�� | ����������м�������ʱ��һ��Ҫ��ʯ���� |
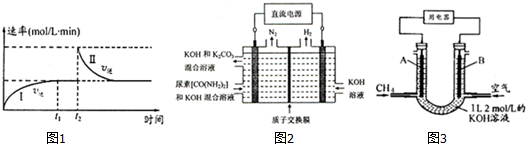
a��0��t1ʱ��v����v����t2ʱ��v����v��
b�����������ܶȲ��ٸı�ʱ���������̴ﵽƽ��
c��t2ʱ�̸ı���������������ܱ������м�C
d���������̴ﵽƽ��ʱ��ƽ�ⳣ����
��2����ҵ�ϳ���CO2��NH3ͨ�����·�Ӧ�ϳ�����[CO��NH2��2]��
CO2��g��+2NH3��g��$\stackrel{һ������}{?}$CO��NH2��2��1��+H2O��g����H��0
t��ʱ�����ݻ��㶨Ϊ2L���ܱ������м���0.10molCO2��0.40molNH3��70min��ʼ�ﵽƽ�⣮��Ӧ��CO2�� g�������ʵ�����ʱ��仯�����ʾ��
| ʱ��/min | ��0 | 20 | 70 | 80 | 100 |
| n��CO2��/mol | 0.10 | 0.060 | 0.020 | 0.020 | 0.020 |
����100minʱ�����������������䣬���������г���0.050mo1CO2��0.20molNH3�����½���ƽ���CO2��ת������ԭƽ����Ƚ���������������䡱��С������
���������淴Ӧ��ƽ�ⳣ��Ϊ277.8������һλС������
��ͼ2��ʾװ�ã�����������Ϊ���Ե缫�������ڵ�����ء�CO��NH2��2���ļ�����Һ��ȡ��������װ���������ĵ缫��ӦʽΪCO��NH2��2+8OH--6e-=CO32-+N2��+6H2O��
��3��CH4ȼ�ϵ�أ�װ��ʾ����ͼ3��A��BΪ�����̼����������ͨ����飬�ڱ�״���£����ļ������VL���� V=44.8Lʱ������ܷ�Ӧ����ʽΪCH4+2O2+KOH=KHCO3+2H2O��


 ��
��