��Ŀ����
�����й���Һ������Ũ�ȵĹ�ϵʽ�У���ȷ����
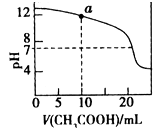
��0.1 mol/L CH2COOH��Һ�ζ� 20 mL0.1 moI/L NaOH��Һ�ĵζ�����
[ ]
A�����������pH�ֱ�Ϊ3��4�Ĵ�����Һ��ȫ�к�ʱ�����������Ƶ����ʵ�����ǰ���Ǻ��ߵ�10��
B����H2A�Ƕ�Ԫ���ᣬ��0.1 mol��L-1 NaHA��Һ�У�c(Na+) =2c(A2- )+c(HA-)+c(H2A)
C��CH3COOH��Һ��NaOH��Һǡ���к�ʱ����Һ��c(Na+)=c(CH3COO-)>c(OH-)=c(H+)
D����ͼ������a���Ӧ����Һ�и�����Ũ�ȵĹ�ϵ��c(OH-)=c(H+)+c(CH3COO-)+ 2c(CH3COOH)
B����H2A�Ƕ�Ԫ���ᣬ��0.1 mol��L-1 NaHA��Һ�У�c(Na+) =2c(A2- )+c(HA-)+c(H2A)
C��CH3COOH��Һ��NaOH��Һǡ���к�ʱ����Һ��c(Na+)=c(CH3COO-)>c(OH-)=c(H+)
D����ͼ������a���Ӧ����Һ�и�����Ũ�ȵĹ�ϵ��c(OH-)=c(H+)+c(CH3COO-)+ 2c(CH3COOH)
D

��ϰ��ϵ�д�
�����Ŀ
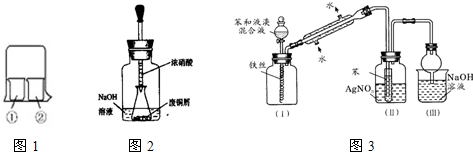
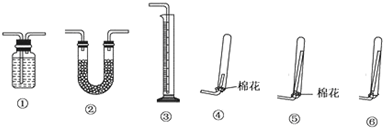

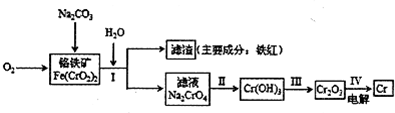
 Cr2O72-+H2O
Cr2O72-+H2O



