��Ŀ����
ij��ѧ��ȤС��Ϊ�˲ⶨ����ʯ��ʯ�Ĵ��ȣ���������֪��ʯ��ʯ�к��ж������裬�������費����ˮ�����������ᷴӦ������ʱ�������ֽⷴӦ������ȡ��8g��ʯ��ʯ��Ʒ����ʵ�������е�δ֪��������������ϡ����40g��4�μ��룬��ַ�Ӧ�����ˡ�����Ȳ����������ÿ��ϡ����������ʣ�������������±�������ʵ�����ݻ��Ƶ�ͼ�����ͼ��| ϡ�������� | ʣ��������� |
| ��1���10g | 6g |
| ��2���10g | 4g |
| ��3���10g | 2g |
| ��4���10g | 1g |
��1���Ĵ�ʵ���е�______������࣬ͼ��aֵΪ______����ʯ��ʯ��Ʒ�Ĵ�����______��
��2�����õ�ϡ�������������������Ƕ��٣�

���𰸡���������1���Ա�ʵ�����ݣ���1��2��3�μ���10gϡ����ʱ��������������Ǽ���2g��˵��10gϡ����ǡ����2g̼�����ȫ��Ӧ������4���ټ���10gϡ����ʱ����ֻ������1g��˵�������е�̼����Ѿ�ȫ�����뷴Ӧ��ʣ���1g���岻�ܺ�ϡ���ᷴӦ����ʯ��ʯ��Ʒ��������1g��̼��Ƶ������ǣ�8g-1g��=7g������̼��Ƶ������������ʯ��ʯ��Ʒ�Ĵ��ȣ�
��2��ͨ������������֪��10gϡ����ǡ����2g̼��Ʒ�Ӧ������̼��������ᷴӦ�ķ���ʽ����̼��Ƶ����������ϡ���������ʵ��������ٸ����������������ļ��㹫ʽ�����ϡ��������������������
����⣺��1�����ݱ��е����ݿ�֪�����ڵ�1��2��3�μ���10gϡ����ʱ��������������Ǽ���2g��˵��10gϡ����ǡ���ܹ���2g̼�����ȫ��Ӧ������4���ټ���10gϡ����ʱ����ֻ������1g��ʣ���1g���岻�ܺ�ϡ���ᷴӦ��˵�������е�̼����Ѿ�ȫ�����뷴Ӧ��������ʣ�࣬ʯ��ʯ��Ʒ��������1g��a��ֵ����1g��̼��Ƶ������ǣ�8g-1g��=7g��ʯ��ʯ��Ʒ�Ĵ����ǣ�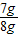 ×100%=87.5%��
×100%=87.5%��
��2����10gϡ���������ʵ�����Ϊx��
CaCO3+2HCl=CaCl2+CO2��+H2O
100 73
2g x
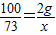
x=1.46g
���õ�ϡ�������������������ǣ�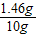 ×100%=14.6%��
×100%=14.6%��
�𣺣�1���Ĵ�ʵ���е�4������࣬ͼ��aֵΪ1����ʯ��ʯ��Ʒ�Ĵ����� 87.5%����2�����õ�ϡ��������������������14.6%��
�����������ѶȲ��Ǻܴ���Ҫ���������ȥ�������ݼ����ݻ�ѧ����ʽ�ļ��㣬����ѧ���������ݵ������ͽ�������������
��2��ͨ������������֪��10gϡ����ǡ����2g̼��Ʒ�Ӧ������̼��������ᷴӦ�ķ���ʽ����̼��Ƶ����������ϡ���������ʵ��������ٸ����������������ļ��㹫ʽ�����ϡ��������������������
����⣺��1�����ݱ��е����ݿ�֪�����ڵ�1��2��3�μ���10gϡ����ʱ��������������Ǽ���2g��˵��10gϡ����ǡ���ܹ���2g̼�����ȫ��Ӧ������4���ټ���10gϡ����ʱ����ֻ������1g��ʣ���1g���岻�ܺ�ϡ���ᷴӦ��˵�������е�̼����Ѿ�ȫ�����뷴Ӧ��������ʣ�࣬ʯ��ʯ��Ʒ��������1g��a��ֵ����1g��̼��Ƶ������ǣ�8g-1g��=7g��ʯ��ʯ��Ʒ�Ĵ����ǣ�
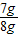 ×100%=87.5%��
×100%=87.5%����2����10gϡ���������ʵ�����Ϊx��
CaCO3+2HCl=CaCl2+CO2��+H2O
100 73
2g x
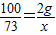
x=1.46g
���õ�ϡ�������������������ǣ�
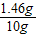 ×100%=14.6%��
×100%=14.6%���𣺣�1���Ĵ�ʵ���е�4������࣬ͼ��aֵΪ1����ʯ��ʯ��Ʒ�Ĵ����� 87.5%����2�����õ�ϡ��������������������14.6%��
�����������ѶȲ��Ǻܴ���Ҫ���������ȥ�������ݼ����ݻ�ѧ����ʽ�ļ��㣬����ѧ���������ݵ������ͽ�������������

��ϰ��ϵ�д�
 ���ɿ��õ�Ԫ����AB��ϵ�д�
���ɿ��õ�Ԫ����AB��ϵ�д� С�����ϵ�д�
С�����ϵ�д�
�����Ŀ
ij��ѧ��ȤС��Ϊ�˷������������ĺ�����������ʵ���о�����ȡ60g����������������800ϡ�����4�μ��뵽����Ʒ�У�������ݼ�¼���±���

��˵�������������ʲ�����ˮ��Ҳ�������ᷴӦ����
��1����2�β��ʣ���������Ϊ15.2g����ɷ�Ϊ ��
A���� B������̼ C��̼
��2��������������������Ϊ���٣���д��������̣����������С�����һλ��
��3����������ϡ�����������������Ϊ���٣���д��������̣����������С�����һλ����������Ӧ���ɵ��������������ϡ���������Ĺ�ϵͼ��
| ��1�� | ��2�� | ��3�� | ��4�� | |
| ����ϡ����������g�� | 200 | 200 | 200 | 200 |
| ʣ�����������g�� | 37.6 | 15.2 | 4 | 4 |

��˵�������������ʲ�����ˮ��Ҳ�������ᷴӦ����
��1����2�β��ʣ���������Ϊ15.2g����ɷ�Ϊ
A���� B������̼ C��̼
��2��������������������Ϊ���٣���д��������̣����������С�����һλ��
��3����������ϡ�����������������Ϊ���٣���д��������̣����������С�����һλ����������Ӧ���ɵ��������������ϡ���������Ĺ�ϵͼ��
����ij��ʢ��ʯ��ʯ�����к��в�������������ʣ�ij��ѧ��ȤС��Ϊ�˲ⶨʯ��ʯ��̼��Ƶĺ�����ȡ10.0gʯ��ʯ��Ʒ���������ȫ�������ձ��У�������������ϡ���ᣬ�ձ�����ʢ���ʵ�������Ϊ80.0g����Ӧ�����в���ձ�����ʢ���ʵ���������Ӧʱ���¼���±���
�Լ��㣺��1����Ӧ�������ų����ٿ˵Ķ�����̼��
��2����ʯ��ʯ��Ʒ��̼��Ƶ����������Ƕ��٣�
| ��Ӧʱ��/�� | 0 | 2 | 4 | 6 | 8 | 10 |
| �ձ�����ʢ��������/�� | 80.0 | 79.0 | 78.3 | 77.9 | 77.8 | 77.8 |
��2����ʯ��ʯ��Ʒ��̼��Ƶ����������Ƕ��٣�
 ij��ѧ��ȤС��Ϊ�˴��Բⶨһ��ʯ��ʯ��Ʒ��CaCO3��������������Ʋ���������ʵ�飮ʵ��װ����ͼ��ʾ����ȡ��ϸ��2.60gʯ��ʯ��Ʒ����4�μ���ϡ���ᣬ��ַ�Ӧ�����ٲ�������Ϊֹ����÷�Ӧǰ����й����������
ij��ѧ��ȤС��Ϊ�˴��Բⶨһ��ʯ��ʯ��Ʒ��CaCO3��������������Ʋ���������ʵ�飮ʵ��װ����ͼ��ʾ����ȡ��ϸ��2.60gʯ��ʯ��Ʒ����4�μ���ϡ���ᣬ��ַ�Ӧ�����ٲ�������Ϊֹ����÷�Ӧǰ����й����������
