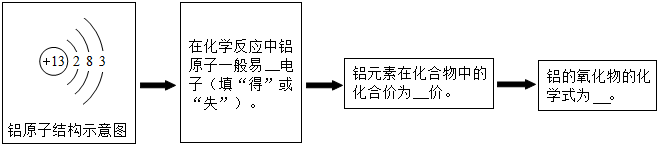
��Ŀ����
2���������ƣ�CaO2����һ�ֶ���������ͨ����Ca��OH��2Ϊԭ���Ƶã��Ʊ��������£�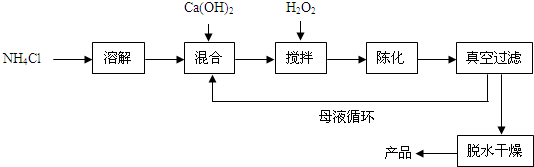
��1��ԭ����NH4Cl�е�Ԫ�صĻ��ϼ���-3��NH4Cl�е�Ԫ�ص���������Ϊ26.2%����ȷ�� 0.1%����
��2���뽫��������з�����Ӧ�Ļ�ѧ����ʽ����������
CaCl2+H2O2+2NH3•H2O+6H2O=CaO2•8H2O��+2NH4Cl���Ʊ������г�ˮ���ѭ��ʹ�õ�������NH4Cl���ѧʽ����
���� ��1�������ڻ��������������ϼ۴�����Ϊ����м��㣻���ݵ�Ԫ�ص����������ļ��㷽�����ǣ���2�����������غ㶨�ɽ��з��������ݼ��Ƿ�Ӧ��Ҳ������������ʿ���ѭ�����ý��з�����
��� �⣺��1��NH4Cl����Ԫ����+1�ۣ���Ԫ����-1�ۣ��赪Ԫ�ػ��ϼ���X��X+��+1����4+��-1��=0�����X=-3��NH4Cl�е�Ԫ�ص���������Ϊ��$\frac{14}{14+1��4+35.5}��100%$=26.2%��
��2�����������غ㶨�ɿ�֪����ѧ��Ӧǰ��ԭ�Ӹ������䣬������Ҫ�����������ˮ���Ȼ�識ȳ������˷�Ӧ���У�Ҳ���������������У������Ʊ������г�ˮ���ѭ��ʹ�õ������ǣ�NH4C1��
�ʴ�Ϊ����1��-3��26.2%����2��H2O�� NH4Cl��
���� �ڽ������ʱ�����ȷ������еķ�Ӧ���̣�Ȼ����ѧ����֪ʶ������������֪ʶ���з������

��ϰ��ϵ�д�
�����Ŀ
12���㴻������ѿ�㴻����Ϊ�������߲ˡ����㴻����Ӫ���ḻ�������е���߰���п�����������ҩ�ü�ֵ�������й���߰����ѧʽΪC27H42O3��˵���д�����ǣ�������
| A�� | ��߰�����л��� | B�� | һ����߰�����к���72��ԭ�� | ||
| C�� | ��߰������Ԫ����� | D�� | ��߰���⡢��Ԫ������Ϊ14��1 |
4����ͼʵ���������ȷ���ǣ�������
| A�� |  ��ȡҺ��ҩƷ | B�� |  ��ȡҺ��Ķ��� | C�� |  ����ҩƷ��ȡ�� | D�� |  Ϩ��ƾ��� |