��Ŀ����
2�������������г��������ʣ������ϳɷֵ�Ħ�����������������ݼ��ȣ�����Ħ�����������кܶ࣬��CaCO3��Al��OH��3��SiO2����Щ���ʵĻ���ij��ѧ��ȤС��ͬѧ��̽������Ħ�����ijɷ֣�ʵ��̽��һ��ijƷ���������Ƿ���CaCO3��
[��������]�������裨SiO2������ϡ���ᷴӦ��
[���ʵ��]
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��ȡ������Ʒ���Թ��У��μ�ϡ���ᣮ | ��������ð�� | ��Ʒ�������к���CaCO3�� |
| �ڽ�����������ͨ�����ʯ��ˮ�� | ��ʯ��ˮ����� |
��ȤС��ͬѧȡ��Ʒ��������Ʒ8.00g������������ϡ���ᣬ������ͼ1��ʾ��ʵ��װ�ý���ʵ�飬������װ�÷��ڵ�����ƽ�ϣ���������ƽ���������ݻ����ͼ2��
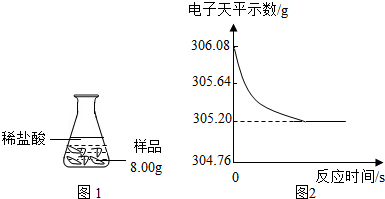
[���ݴ���]ʵ����������������0.88g�����Ʒ��������CaCO3��������Ϊ25%��
[�����뷴˼]
��1��д����ƿ�з�Ӧ�Ļ�ѧ����ʽCaCO3+2HCl�TCaCl2+H2O+CO2����дһ�����ɣ���
��2�����������Ҫ��������������CaCO3������������ƫС��ѡ�ƫ�������䡱��ƫС������
��3�������������ж�ϡ�����ѹ����ķ���ȡ��Ӧ����ƿ�е�������Һ���Թ��У��μ���ɫʯ����Һ����Һ���ɫ��д��ʵ�鲽�������
��4��ͬѧ�Ǿ�������һ����Ϊ������ͼ1װ�ô��ڿ�ѧ�����⣬�������ʵ��������˵�������������һ����������ӷ���
���� ʵ��̽��һ
[���ʵ��]����̼������ӵļ��鷽���������ʵ�飮
ʵ��̽������
[���ݴ���]��������������Һ�����ն�����̼������������㣮
[�����뷴˼]
��1������̼���������ķ�Ӧ��д����ƿ������������Ļ�ѧ����ʽ��
��2�����������Ҫ��������������CaCO3������������ƫС��
��3������ϡ��������ʣ����м�⣮
��4������������Ļӷ��ԣ�
��� �⣺ʵ��̽��һ
[���ʵ��]����̼��������ᷴӦ�����˶�����̼���壬������̼��ʹ�����ʯ��ˮ����ǣ����ԣ����ʵ�����£�
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��ȡ������Ʒ���Թ��У��μ� ϡ���� | �������ݲ��� | ��������Ʒ�к���CaCO3�� |
| �ڽ�����������ͨ�����ʯ��ˮ | �ڳ���ʯ��ˮ����� |
���������������������̼��Ӧ������̼���ƺ�ˮ�����ԣ�װ��Dƽ�����ӵ�����Ϊ0.88g�������ӵ�������Ϊ��Ӧ�����Ķ�����̼��������
��������Ʒ��̼��Ƶ�����������Ϊx
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
x 0.88g
$\frac{100}{44}=\frac{x}{0.88g}$����ã�x=2.00g
������Ʒ��̼��Ƶ���������Ϊ��$\frac{2.00g}{8g}$��100%=25%��
[�����뷴˼]
��1����ƿ�з�Ӧ�Ļ�ѧ����ʽ CaCO3+2HCl�TCaCl2+H2O+CO2����Al��OH��3+3HCl�TAlCl3+3H2O��
��2����Ϊ�����С�Ħ�����������кܶ࣬��CaCO3��Al��OH��3��SiO2����Щ���ʵĻ�������Al��OH��3����ϡ���ᷴӦ���ʼ��������Ҫ��������������CaCO3������������ƫС��
��3�������������ж�ϡ�����ѹ����ķ���Ϊ��ȡ��Ӧ����ƿ�е�������Һ���Թ��У��μ���ɫʯ����Һ����Һ���ɫ��
��4��ͬѧ�Ǿ�������һ����Ϊ������ͼ1װ�ô��ڿ�ѧ�����⣬�������ʵ��������˵�������������һ������������ӷ���
�ʴ�Ϊ��
[���ʵ��]ϡ��� ������ð����ʯ��ˮ����ǣ�
[���ݴ���]0.88g�� 25%
[�����뷴˼]��1��CaCO3+2HCl�TCaCl2+H2O+CO2����Al��OH��3+3HCl�TAlCl3+3H2O����2��ƫС��
��3��ȡ��Ӧ����ƿ�е�������Һ���Թ��У��μ���ɫʯ����Һ����Һ���ɫ����������ȷ�𰸣���
��4������ӷ�����������ȷ�𰸣���
���� ʵ���ǻ�ѧ����Ҫ��ɲ��֣���ȷ��ʵ������ǵó���ѧ���۵�ǰ������֮һ�����Ҫѧ�����ʵ�顢����ʵ�顢����ʵ�飬Ϊ�ó���ȷ�Ľ��۵춨������

��ϰ��ϵ�д�
�����Ŀ
12�������������ʵķ�������ȷ���ǣ�������
| A�� | �÷���ˮ����Ӳˮ����ˮ | |
| B�� | ��ˮ��������粒�����������ƹ��� | |
| C�� | ��ϡ����������������Ƿ���� | |
| D�� | �÷�̪��Һ����ϡ�����ʳ��ˮ |
17��ʳ���ǵ�ζƷ�����к��д��ᣨC2H4O2�������й��ڴ������������ȷ���ǣ�������
| A�� | ������� | |
| B�� | ̼���⡢��Ԫ�ص�������Ϊ6��1��8 | |
| C�� | ���������������� | |
| D�� | ���������̼���⡢��ԭ�ӵĸ�����Ϊ1��2��1 |
7�������Ƕ��������ʵ��������ٻ���ɫ�����д̼�����ζ��������ˮ������Ӧ���ܳ�������һ�����壻������ǿ����Һ������Ӧ�����������������Ӧ�����������������ʵ��ǣ�������
| A�� | �٢ڢ� | B�� | �٢ڢ� | C�� | �ڢܢ� | D�� | �ۢܢ� |
14������ͼ������ȷ��ӳ��Ӧ�仯��ϵ���ǣ�������
| A�� |  ��ͭ��п������ĩ�м������� | |
| B�� |  ��ͭƬ���뵽һ��������������Һ�� | |
| C�� |  ��������ȫ��ͬ��ϡ�����зֱ����п�ۡ�þ�� | |
| D�� |  ��������ȫ��ͬ��ϡ�����зֱ����п�ۡ�þ�� |
12�����г�����ѡ�õ��Լ��У���ȷ���ǣ�������
| ���� | �������� | �����Լ� | |
| A | ���� | ͭ�� | ϡ���� |
| B | NaCl | Na2CO3 | ϡH2SO4 |
| C | CO2 | CO | ���ȵ�CuO |
| D | KCl | KNO3 | H2O |
| A�� | A | B�� | B | C�� | C | D�� | D |
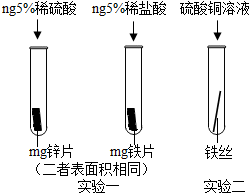 ��Ԫ��������Ĺ�ϵ���У�
��Ԫ��������Ĺ�ϵ���У�