��Ŀ����
ij��ѧ��ȤС��Ϊ̽��ͭ��Ũ����ķ�Ӧ������ͼװ�ý����й�ʵ�顣��ش�ʵ���м�ͬѧ��a gͭƬ��12 mL 18mol/L��Ũ�������Բ����ƿ�м��ȣ�ֱ����Ӧ��ϣ��������ƿ�л���һ�����������ͭƬʣ�ࡣ
��1����д��ͭƬ��Ũ���ᷴӦ�Ļ�ѧ����ʽ
װ��E��D��ʢƷ����Һ����C�����強����D���п��ܹ۲쵽��������
ʵ��װ��D���п�����ɻ�����Ⱦ��������ķ��������ʵ����Ʒ��ѡ��
װ��B�������������������塣��D������������ر�����K����ȥ�ƾ��ƣ����������ȵ����ã�A�����������������ʱB�������� B��Ӧ���õ�Һ�壨����ĸ��Ϊ�� ��
A������ Na2SO3 ��Һ B������KMnO4 ��Һ C��Ũ��ˮ D������NaHSO3
��2���������ۣ�Ϊʲô��һ���������ᵫδ��ʹͭ��ȫ�ܽ⣬����Ϊԭ���� ��
��������ҩƷ�ܹ�����֤����Ӧ���������ƿ��ȴ��������� ��
A��Fe�� B��BaCl2 ��Һ C��Ag D�� Na2CO3 ��Һ
��1����д��ͭƬ��Ũ���ᷴӦ�Ļ�ѧ����ʽ
װ��E��D��ʢƷ����Һ����C�����強����D���п��ܹ۲쵽��������
ʵ��װ��D���п�����ɻ�����Ⱦ��������ķ��������ʵ����Ʒ��ѡ��
װ��B�������������������塣��D������������ر�����K����ȥ�ƾ��ƣ����������ȵ����ã�A�����������������ʱB�������� B��Ӧ���õ�Һ�壨����ĸ��Ϊ�� ��
A������ Na2SO3 ��Һ B������KMnO4 ��Һ C��Ũ��ˮ D������NaHSO3
��2���������ۣ�Ϊʲô��һ���������ᵫδ��ʹͭ��ȫ�ܽ⣬����Ϊԭ���� ��
��������ҩƷ�ܹ�����֤����Ӧ���������ƿ��ȴ��������� ��
A��Fe�� B��BaCl2 ��Һ C��Ag D�� Na2CO3 ��Һ
��1��ͭƬ��Ũ���ᷴӦ�Ļ�ѧ����ʽCu+2H2SO4(Ũ)===CuSO4+2H2O+SO2����2�֣�
D���п��ܹ۲쵽�������� Ʒ����Һ��ɫ ��1�֣�������ķ��������ʵ����Ʒ��ѡ�� ������������ʪ�����������Թܿ� ��2�֣� B�������� �Լ�ƿ��Һ���½�������©����Һ������ ��2�֣�B��Ӧ���õ�Һ��Ϊ��D ��1�֣���
��2��ԭ���� ������������ģ�Ũ�����Ũ�Ȳ��ϼ�С��ϡ������ͭ����Ӧ ����2�֣�
֤����Ӧ���������ƿ��ȴ��������� AD ��2�֣� ��
D���п��ܹ۲쵽�������� Ʒ����Һ��ɫ ��1�֣�������ķ��������ʵ����Ʒ��ѡ�� ������������ʪ�����������Թܿ� ��2�֣� B�������� �Լ�ƿ��Һ���½�������©����Һ������ ��2�֣�B��Ӧ���õ�Һ��Ϊ��D ��1�֣���
��2��ԭ���� ������������ģ�Ũ�����Ũ�Ȳ��ϼ�С��ϡ������ͭ����Ӧ ����2�֣�
֤����Ӧ���������ƿ��ȴ��������� AD ��2�֣� ��
��

��ϰ��ϵ�д�
�����Ŀ
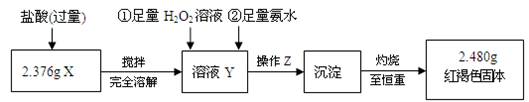
 �������ӷ���ʽ��ʾ����
�������ӷ���ʽ��ʾ����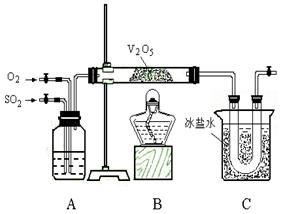
 ��ʱ�õ�b g ��������ʵ���ж��������ת���ʲ�С��_________________��
��ʱ�õ�b g ��������ʵ���ж��������ת���ʲ�С��_________________�� ��
��


 ��
��
 ������������ȷ��̼��Ƶ�����������
������������ȷ��̼��Ƶ�����������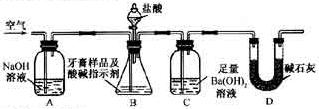
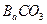 ������ֻҪ�ⶨװ��C������
������ֻҪ�ⶨװ��C������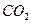 ǰ��������һ������ȷ��̼��Ƶ�����������ʵ��֤�����˷����ⶨ�Ľ������ƫ�ߣ�ԭ����______.
ǰ��������һ������ȷ��̼��Ƶ�����������ʵ��֤�����˷����ⶨ�Ľ������ƫ�ߣ�ԭ����______.