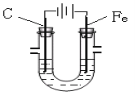
��Ŀ����
����Ŀ���������Ժʿ����Ժʿѧ��ͬ���ġ����ӹ��������ն�����������ˣ�ȫ��������������˵��BF3��TiO2��CH3COOH��CO2��NO����ï����NH3��HCN��H2S��O3�������ϩ���Ƶ��ڶ������ӹ������е����ǡ�
(1)д��Fe2���ĺ�������Ų�ʽ________________________________��
(2)����˵����ȷ����________��
a��H2S��O3���Ӷ���ֱ����
b��BF3��NH3��������
c��CO2��HCN���ӵĽṹʽ�ֱ���O=C=O��H��C��N
d��CH3COOH������̼ԭ�ӵ��ӻ���ʽ�У�sp2��sp3
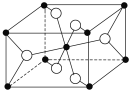
(3)TiO2����Ȼ�����У����ȶ���һ�־���ṹ��ͼ�� �����ʾ________ԭ�ӡ�
(4)����(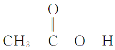 )�۷е�ܸߣ������ڴ����Է��Ӽ�����ϵĶ�����(��һ����״�ṹ)���뻭���ö�����Ľṹ��_________________________________________��
)�۷е�ܸߣ������ڴ����Է��Ӽ�����ϵĶ�����(��һ����״�ṹ)���뻭���ö�����Ľṹ��_________________________________________��
���𰸡�1s22s22p63s23p63d6��[Ar]3d6 cd ��(O) 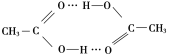
��������
(1)����26��Ԫ��,��ԭ�Ӻ�����26�����ӣ���ԭ��ʧȥ2�����ӱ�ΪFe2+,���ݹ���ԭ��֪�������Ӻ�������Ų�ʽΪ1s22s22p63s23p63d6��[Ar]3d6��
�ʴ�Ϊ��1s22s22p63s23p63d6��[Ar]3d6��
(2)a.H2S��O3���Ӷ���V�Σ� a�����
b.BF3Ϊƽ�������Σ�NH3��Ϊ�����Σ�b�����
c.CO2��HCN���ӵĽṹʽ�ֱ��ǣ�O=C=O��HC��N��c����ȷ��
d. CH3COOH�����м���-CH3����ԭ���γ�4���������ӻ������ĿΪ4�����õ���sp3�ӻ����Ȼ���-COOH����̼ԭ���γ�3���������ӻ������ĿΪ3�����õ���sp2�ӻ���d����ȷ��
�ʴ�Ϊ��c d
(3) �������=![]() ,�������=
,�������=![]() ,������ͺ��������Ϊ2:1,���ݶ�������Ļ�ѧʽ֪,�����ʾ��ԭ��.
,������ͺ��������Ϊ2:1,���ݶ�������Ļ�ѧʽ֪,�����ʾ��ԭ��.
�ʴ�Ϊ����(O)
(4)����������Ӽ���,�Ȼ��ϵ���ԭ������һ�����������̼��˫������ԭ���γ����,���Ըö�����ĽṹΪ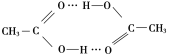 ��
��
�ʴ�Ϊ��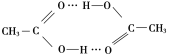 ��
��

����Ŀ������ʯ����Կ�����MgO��![]() ��
��![]() ��
��![]() ��ɡ�ijʵ��С����������������ֱ��Ƶõ���Al��Fe��Mg��Si��
��ɡ�ijʵ��С����������������ֱ��Ƶõ���Al��Fe��Mg��Si��
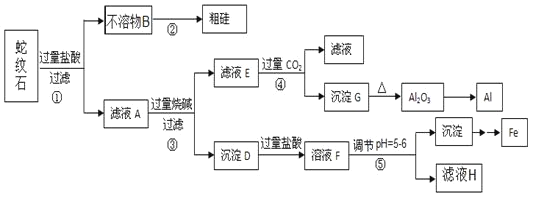
�й��������������pH���±���
���������� | �������↑ʼ����ʱ��pH | ����������ȫ����ʱ��pH |
| 1.9 | 3.2 |
| 9.4 | 11.6 |
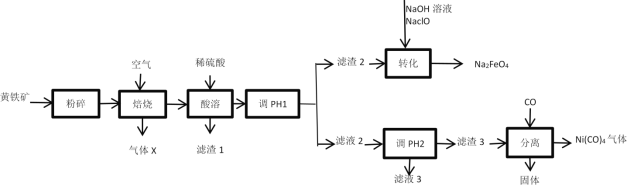
��1������ʯ������������þ�������⼸��Ԫ�صļ����ӵİ뾶��С�����˳��_____����ҺA�к��е��������� _________��
��2����Ԫ�������ڱ��е�λ��______��������̼�ĵ���ʽ___________��
��3���������з�Ӧ�Ļ�ѧ����ʽΪ ____�������������ɳ���G�����ӷ���ʽΪ _____��
��4���������е���pH��![]() ʱ�������õ����Լ�
ʱ�������õ����Լ�![]() �����
�����![]() ____________��
____________��
a��NaOH b����ˮ c��MgO d��Mg��OH��2
��5������ҺH��ȡ����Mg���������£�
![]()
������ұ��þ�ķ�����__________���ڸ����HCl�����м���MgCl2��6H2O��ȡ��ˮ�Ȼ�þ��ԭ����__________��
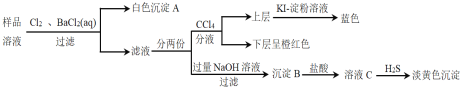
����Ŀ������п�����̵���(��Ҫ�ɷ�ΪZnO����������Fe2O3��CuO��SiO2��MnO��)Ϊԭ�Ͽ���������п����(ZnC2O4��2H2O)��
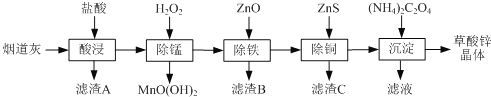
�й��������↑ʼ�����ͳ�����ȫ��pH���±���
�������� | Fe(OH)3 | Fe(OH)2 | Cu(OH)2 | Zn(OH)2 |
��ʼ������pH | 1.5 | 6.5 | 4.2 | 5.4 |
������ȫ��pH | 3.3 | 9.7 | 6.7 | 8.2 |
���ʴ��������⣺
��1������B����Ҫ�ɷ�Ϊ________ ��
��2�����̹����в���MnO(OH)2���������ӷ���ʽΪ________��
��3���ٳ���(����Cu2�����ܱ���ȥ)ʱ����ZnO���Ʒ�ӦҺpH�ķ�ΧΪ___________��
�����������г������ͭ��˳���ܵߵ�����������ʻ��С����ԭ����________��
��4�����������̲���Na2C2O4�������茶�����������п�������ļ��Ϸ�ʽ��________��
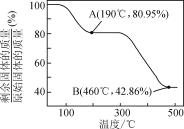
��5��������п������ȷֽ�ɵõ�һ�����ײ��ϡ����ȹ����й�����������¶ȵı仯��ͼ��ʾ��300 �桫460 �淶Χ�ڣ�������Ӧ�Ļ�ѧ����ʽΪ________��