��Ŀ����
������16�֣���ijʵ��С��ͬѧΪ��̽��ͭ��Ũ����ķ�Ӧ������������ϵ��ʵ�顣
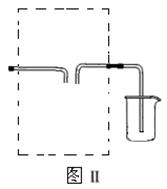
��ʵ��1��ͭ��Ũ���ᷴӦ��ʵ��װ����ͼ��ʾ��
ʵ�鲽�裺
�������Ӻ�װ�ã����������ԣ������Լ���
�ڼ���A�Թ�ֱ��B��Ʒ����ɫ��Ϩ��ƾ��ƣ�
�۽�Cu˿�����뿪Һ�档
��1��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2��Ϩ��ƾ��ƺ���Ϊ�е���D�Ĵ��ڣ�B�е�Һ�岻�ᵹ������ԭ���� ��
��3�����װ��ǰ������������Ϳ�ʹװ���в���������ȫ�����գ�Ӧ����ȡ�IJ����� ��
���͵�NaHSO3��Һ������ȩ�����ӳɷ�Ӧ������ˮ���ԵĦ�-�ǻ������ơ������ķ�ӦΪ��
R-CHO + NaHSO3 R-CH��OH��SO3Na
R-CH��OH��SO3Na
��Ӧ�ǿ���ģ���ͨ����������70%--90%������Ӧ����ת����
��4�����â���װ����ȡ����NaHSO3��Һ��Ӧ��ȡ��ʩ����װ�ý��в��ָı䡣���ִ�ʩ�ǣ�_____________________________________________________________________��ȷ����NaHSO3���ɵ�������:_______________________________________.
��5�����屽�л���������ȩ,���������ʳ�ȥ,�ɲ��õ��Լ���:__________,�����ķ�����:_____________________.
��6������CH3-CH(OH)SO3Na ˮ��Һ�м�����������,�л���ת��Ϊ:___________,����ת�������������ķ�������__________________.
��1��2H2SO4(Ũ)+Cu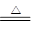 CuSO4+SO2��+2H2O (2��)
CuSO4+SO2��+2H2O (2��)
��2���Թ�A������ѹǿ��С��������D���ܽ����Թ�A�� (2��)
��3����D�ܿ���A�д������� (2��)
��4����B��C��λ�õ�����(2��)Ʒ����Һ��ɫ��(2��)
��5������NaHSO3��Һ(2��) ��Һ(2��) ��6��CH3CHO(2��)����(2��)
�������������
��1��Ũ������ͭ��Ӧ���ɲ��տα�Ũ��������ʡ�
��2��D�����������ǵ�A�е���ѹ��Сʱ����������D���ܽ���A�У���ֹ���������á�
��3��ͨ����Ŀ��֪����D�ܹ������ԴﵽĿ�ġ�
��4��Ҫ�Ʊ����������ƣ��ͱ���������������Һ��ͨ������Ķ�������ͬʱ��Ҫ����֤������������Ѿ��������뵽�����B��C���Թܵ����Ϳɣ����۲쵽Ʒ����Һ��ɫʱ��˵�����������Ѿ�����������������Ҳ�Ͳ����ˡ�
��5����С����Ҫ�����Ϣ����Ȼѡ�õ��DZ���������������Һ����ȥ����ȩ�����ڻ������ܵ�Һ�壬�����÷�Һ�ķ�����
��6����������������������Ƴ���ȩ��һ�����淴Ӧ����������������������Ƶ�Ũ���½���ƽ�����淴Ӧ�����ƶ���������ȩ����Ϊ��ȩ����ˮ��Һ���ʱ��Ҫ���÷е㲻ͬ��������ķ�����
���㣺Ũ��������ʣ�������������ʣ�������ᴿ�ķ�����

��15�֣�
���Ȼ��ף�PCl3����һ����Ҫ���л��ϳɴ�����ʵ���ҳ��ú���������Cl2��ȡPCl3��װ������ͼ��ʾ��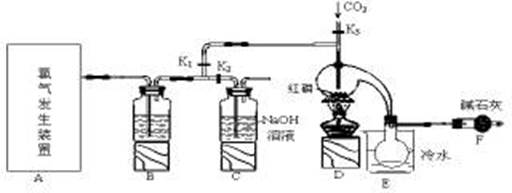
��֪������������Cl2��Ӧ����PCl3�������Cl2��Ӧ����PCl5��PCl3��O2������POCl3(��������)�� POCl3����PCl3��PCl3��ˮ��ǿ��ˮ������H3PO3��HCl��PCl3��POCl3���۷е���±���
| ���� | �۵�/�� | �е�/�� |
| PCl3 | -112 | 75.5 |
| POCl3 | 2 | 105.3 |
��1��д��A�з�Ӧ�����ӷ���ʽ ��
��2��Bװ������ʢ�Լ��� ��E���ձ�����ˮ�������� ��
��3��ʵ��ǰ������֮��������Ҫ�õ���Ƥ�ܣ������ӷ����ǣ��Ȱ� ��Ȼ�������������ɰѲ����ܲ�����Ƥ���С�
��4�����װ�������Ժ���Dװ�õ�����ƿ�м�����ף���K3ͨ������CO2��һ��ʱ���ر�K3����������ƿ���ϲ��л�ɫ���������ʱͨ����������Ӧ�������С�����ͨ�����CO2��Ŀ���� ��
��5��ʵ���ƵõĴֲ�Ʒ�г�����POCl3��PCl5�ȣ���������ȳ�ȥPCl5����ͨ�� ����ʵ�����ƣ����ɵõ��ϴ�����PCl3��
��6��ʵ���ر�K1����K2����A��B������ͨ��300ml 1mol/L��NaOH��Һ�С���NaOHǡ����ȫ��Ӧ�����������������ʵ���Ϊ�����跴Ӧ������NaCl �� NaClO �� NaClO3�����Σ� mol����Ӧ��ת�Ƶ��ӵ����ʵ���(n)�ķ�Χ�� ��
��֪��ICl���۵�Ϊ13��90C���е�Ϊ97��40C����ˮ�⣬���ܷ�����Ӧ��ICl(l��+ Cl2(g��=ICl3(l)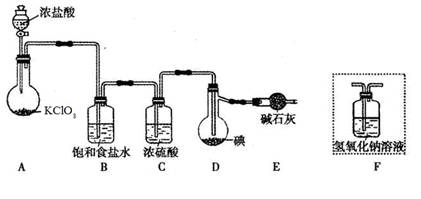
��1��װ��A�з�����Ӧ�Ļ�ѧ����ʽ��____________ ��
��2��װ��B��������______��������װ��F����װ��E������____________ ��
��3�����Ƶõ�ICl����������ICl3���ʣ��ᴿ�ķ�����______ (���ţ���
| A������ | B�������ᾧ | C������ | D����Һ |
��

��ICl+KI��I2+KCl
��I2��2Na2S2O3��2NaI+Na2S4O6
ʵ��1:��5��00g����֬��Ʒ�������Ȼ�̼���γ�100mL��Һ������ȡ��ʮ��֮һ������20mLijICl�ı�������Һ(����������ַ�Ӧ��������KI��Һ�����ɵĵⵥ����a mol��L-1��Na2S2O3����Һ�ζ�����ƽ��ʵ�飬������ĵ�Na2S2O3��Һ��ƽ�����ΪV1mL��
ʵ��2(�հ�ʵ�飩��������֬��Ʒ�������������衢�����Լ���������ʵ��1��ȫ��ͬ��������ĵ�Na2S2O3��Һ��ƽ�����ΪV2mL��
�ٵζ������п���______ ��ָʾ����
�ڵζ���������Ҫ����������ᵼ��V1______(�ƫ��ƫС����
��5��00g����֬��Ʒ�����ĵ�ICl�����ʵ���Ϊ__mol���ɴ����ݾ����㼴����ø���֬�IJ����Ͷȡ�
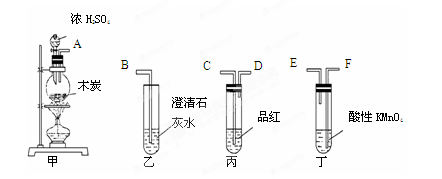
��ͼ���о�ͭ��Ũ����ķ�Ӧװ�ã�

��1��A�Թ��з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2����Ӧһ��ʱ��ɹ۲쵽B�Թ��е�����Ϊ ��
��3��C�Թܿڽ���NaOH��Һ������������ ��
��4���罫B�Թܻ���D�Թܣ�����ֱ����������BaCl2��Һ��ͨ����һ�����壬������ɫ����������������� �� ����Ҫ����һ�ֻ������һ�ֵ��ʵĻ�ѧʽ��������Ҫ���ɼ�װ������װ�á���
��5��ʵ�������֤��A�Թ��з�Ӧ���ò����Ƿ���ͭ���ӵIJ��������� ��
��6����ͭ��Ũ���ᷴӦ�Ĺ����У������к�ɫ���ʳ��֣�������������������ϡ�
����
| ����1 |  ����ͭ��Ũ���ᷴӦ������ɫ���ʵ�������� |
| ����2 | X���߾������������ͭ��Ũ���ᷴӦ���ɵĺ�ɫ����ΪCu2S��CuS��Cu7S4�е�һ�ֻ��֡� |
�������Ͽɵó�����ȷ������ ��
a��ͭ��Ũ���ᷴӦʱ���漰�ķ�Ӧ���ܲ�ֹһ��
b������Ũ��ѡ���ʵ����ɱ����������г��ֺ�ɫ����
c���÷�Ӧ����������֮һ������Ũ�ȡ�15 mol��L
d������Ũ��Խ��ɫ����Խ����֡�Խ����ʧ