��Ŀ����
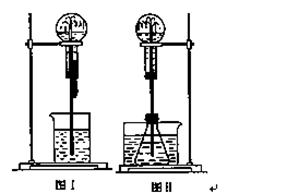
��ͼ��ʾ�����ס�������װ�в�ͬ���ʵ���Ͳ�õ�������������������Ͳ�ڵ�����ѹ������Ͳ�ڣ������±����еIJ�ͬʵ��(������ͬ��ͬѹ�²ⶨ)���Իش��������⣺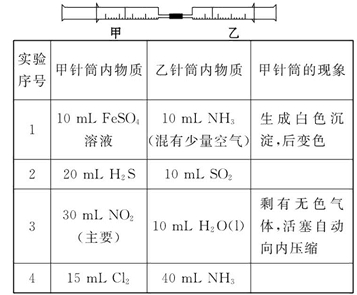
(1)ʵ��1�У��������ձ�Ϊ_______ɫ��д��������ɫ�Ļ�ѧ����ʽ_____________________��
(2)ʵ��2����Ͳ�ڵ������ǣ���_______���ɣ���Ͳ����_______�ƶ�(����⡱�������ڡ�����)����Ӧ�����Ͳ���������IJ������壬��ȷ�Ĵ��������ǽ���ͨ��_______��Һ�С�
(3)ʵ��3�У����е�30 mL������NO2��N2O4�Ļ�����壬��ô�������ʣ�����ɫ������_______��д��NO2��H2O��Ӧ�Ļ�ѧ����ʽ____________________________��
(4)ʵ��4�У���֪��3Cl2+2NH3=N2+6HCl������Ͳ���������ƶ�����Ͳ���а��̲����⣬�������ɫ�仯Ϊ_______�������Ͳ��ʣ����������ԼΪ_______mL��
(1)���
4Fe(OH)2+O2+2H2O=4Fe(OH)3
(2)��ɫ���� ���� NaOH(��KOH)
(3)NO 3NO2+H2O=2HNO3+NO
(4)�ɻ���ɫ��Ϊ��ɫ 5
����

 ����ȫ���ִʾ��ƪ��ϵ�д�
����ȫ���ִʾ��ƪ��ϵ�д����г���I��II��ȷ�����������ϵ���ǣ� ��
| ѡ�� | ����I | ����II |
| A | Fe2(SO4)3��������ˮ�� | Fe 3+ˮ��ɽ��������ˮ�е�ϸС������ |
| B | ��Ba(NO3)2��Һ��ͨ������SO2������������ | �������ǿ�� |
| C | Cl2��ʹʪ��IJ�����ɫ | Cl2����Ư���� |
| D | ���NaCl��Һ�Ʊ������� | NaCl�����ӻ����� |
�й�Ԫ��X��Y��Z��D��E����Ϣ���£�
| | �й���Ϣ |
| X | Ԫ����Ҫ���ϼ�Ϊ��2��ԭ�Ӱ뾶Ϊ0.074 nm |
| Y | ��������������������������֮��Ϊ4 |
| Z | ԭ�Ӱ뾶Ϊ0.102 nm���䵥����X�ĵ�����ȼ�գ���������������ɫ���� |
| D | ����������Ӧ��ˮ���������ˮ�����ܵ������������ȵ����������� |
| E | �����������г�������������Ʒ�ڳ�ʪ�������ױ���ʴ���� |
��ش��������⣺
��1��X��һ���⻯�������ʵ������ȡX�ĵ��ʣ��䷴Ӧ�Ļ�ѧ����ʽΪ___________��
��2��EԪ����YԪ�ؿ��γ�EY2��EY3���ֻ��������˵����ȷ����(�����)_______��
�ٱ���EY2��Һʱ��������Һ�м�������E����
��EY2ֻ��ͨ���û���Ӧ���ɣ�EY3ֻ��ͨ�����Ϸ�Ӧ����
��ͭƬ��̼����EY3��Һ���ԭ��أ�������ͭƬ�ص�������̼��
��3����Z������X�ĵ�����ȼ�յIJ���͵����ʵ�����Y����ͬʱͨ��������ˮ�У���ַ�Ӧ�����Һ�е���Ʒ����Һ��������__________________���йط�Ӧ�����ӷ���ʽΪ________________________________________________________________________��

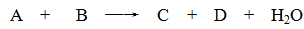
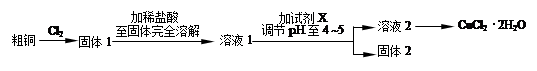

 CuCl42��(aq) +4H2O(l)
CuCl42��(aq) +4H2O(l)