��Ŀ����
5��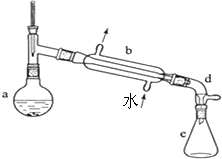 ijУʵ���Һϳɻ���ϩ�ķ�Ӧ��ʵ��װ�ã��гּ����Ȳ�����ʡ�ԣ����£�
ijУʵ���Һϳɻ���ϩ�ķ�Ӧ��ʵ��װ�ã��гּ����Ȳ�����ʡ�ԣ����£�
����������£�
| ��Է������� | �ܶ�/��g•cm-3�� | �е�/�� | �ܽ��� | |
| ���Ѵ� | 100 | 0.9618 | 161 | ����ˮ |
| ����ϩ | 82 | 0.8102 | 83 | ������ˮ |
��[�ϳ�]����a�м���20.00g����������2С���ʯ����ȴ�����»�������10mLŨ���ᣮb��ͨ����ȴˮ��������a�������������¶Ȳ�����90�森
��[�ᴿ]������Ӧ��ֲ��ﵹ���Һ©���У�����������5% Na2CO3��Һ��ˮϴ�ӣ�����������ˮCaCl2����������һ��ʱ�����ȥCaCl2���ٽ����������ջ��12.30g������ϩ��
�ش��������⣺
��1��װ��b��������ֱ�������ܣ������ʯ��Ŀ���Ƿ�ֹ���Һ���У�
��2����ʵ���Һ���ò���Ӧ���Ͽڵ�������Ͽڡ����¿ڡ�����
��3���ᴿ�����м���Na2CO3��Һ��Ŀ���dz�ȥ�����H2SO4��������ˮCaCl2�������Ǹ������û���ϩ�������
��4����ʵ�����û���ϩ�IJ�����75%������=$\frac{ʵ�ʲ���}{���۲���}$��100%��
���� ��1��װ��b�������������Ƭ�Ĵ��ڿ��Է�ֹ�ڼ��ȹ����в�����������
��2����Һ�����У����ڻ���ϩ���ܶȱ�ˮ���ܶ�С����Ӧ�ôӷ�Һ©�����Ͽڵ�����
��3������Ӧ��ֲ��ﵹ���Һ©���У�����������5% Na2CO3��Һ��ˮϴ�ӣ�Ŀ����ϴȥ��Ӧ�����е����ᣬ��ˮ�Ȼ����������ղ�����������ˮ��
��4��������ʵ�����ɵ������������ϵõ�����������ϻ�ѧ����ʽ����õ���
��� �⣺��1������װ��ͼ������֪װ��b������װ���е�������װ�ã���ʯ�Ĵ��ڿ��Է�ֹ�ڼ��ȹ����в�����������
�ʴ�Ϊ��ֱ�������ܣ���ֹ���Һ���У�
��2����Һ�����У����ڻ���ϩ���ܶȱ�ˮ���ܶ�С����Ӧ�ôӷ�Һ©�����Ͽڵ�����
�ʴ�Ϊ���Ͽڣ�
��3������Ӧ��ֲ��ﵹ���Һ©���У�����������5% Na2CO3��Һ��ˮϴ�ӣ�Ŀ����ϴȥ��Ӧ�����еĴ������ᣬ�����ᴿ�����м�����ˮ�Ȼ��Ƶ�Ŀ����������ˮ�Ȼ������ղ�����������ˮ��
�ʴ�Ϊ����ȥ�����H2SO4���������û���ϩ�������
��4��20.00g����������2С���ʯ����ȴ�����»�������10mLŨ������ȷ�Ӧ����12.30g������ϩ��
���ݻ�ѧ����ʽ���㷴Ӧ�õ�����ֵΪ��
C6H12O��C6H10
100 82
20.00g m
m=16.4g
����ϩ�IJ���=$\frac{12.3g}{16.4g}$��100%=75%��
�ʴ�Ϊ��75%��
���� ���⿼���������Ʊ�ʵ�鷽����ƣ������ʵķ���Ӧ�ã�ʵ�������ԭ���ķ����жϣ����ջ����ǽ���ؼ�����Ŀ�Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | Cu2O+H2SO4��CuSO4+Cu+H2O | B�� | FeO+HNO3��Fe��NO3��3+H2O | ||
| C�� | NH4NO3��N2+H2O | D�� | S+KOH��K2SO3+K2S+H2O |
| A�� | ��Һ�е������ӵ���Ŀ���� | |
| B�� | ��Һ��$\frac{c��C{H}_{3}COOH��}{c��C{H}_{3}CO{O}^{-}��}$��ֵ��С | |
| C�� | ����ĵ���̶�����[H+]������ | |
| D�� | �ټ���10mlpH=11��NaOH��Һ�����ҺpH=7 |
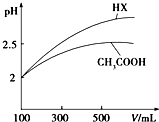 ��֪25��ʱ����������ʵĵ���ƽ�ⳣ�����������ʾ��
��֪25��ʱ����������ʵĵ���ƽ�ⳣ�����������ʾ��| ��ѧʽ | CH3COOH | H2CO3 | HClO | |
| ����ƽ�ⳣ�� | Ka=1.8��10-5 | Kal=4.3��10-7 | Ka2=5.6��10-11 | Ka=3.0��10-8 |
��1�����ʵ���Ũ�Ⱦ�Ϊ0.1mol•L-1��������Һ��a��CH3COONa b��Na2CO3 c��NaClO
pH��С�������е�˳����a��c��b���ñ����д����
��2�������Ϊ100mL pH=2��CH3COOH��һԪ��HX����ˮϡ������pH����Һ����Ĺ�ϵ��ͼ��ʾ����HX�ĵ���ƽ�ⳣ�����ڣ�����ڡ�����С�ڡ����ڡ���CH3COOH�ĵ���ƽ�ⳣ����ϡ�ͺ�HX��Һ��ˮ���������C��H+�����ڣ�����ڡ�����С�ڡ����ڡ���������Һ��ˮ���������C��H+����
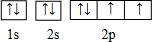 ������ӵ�����������2�֣��Ƚ�����ͬ����ǽ���Ԫ���γɵĵ��ʵ������ԣ��û�ѧ����ʽ��ʾ��2H2S+O2=2S��+2H2O
������ӵ�����������2�֣��Ƚ�����ͬ����ǽ���Ԫ���γɵĵ��ʵ������ԣ��û�ѧ����ʽ��ʾ��2H2S+O2=2S��+2H2O ����Ҫ��Br-��ʽ�����ں�ˮ�У���ˮ�������ԣ���ҵ���Ʊ���Br2�IJ�������Ϊ��
����Ҫ��Br-��ʽ�����ں�ˮ�У���ˮ�������ԣ���ҵ���Ʊ���Br2�IJ�������Ϊ��