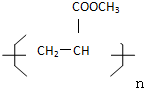ΧβΡΩΡΎ»ί
12Θ°œ¬Ν–”–ΙΊΒΡ–π ω÷–Άξ»Ϊ’ΐ»ΖΒΡ“ΜΉι «Θ®ΓΓΓΓΘ©ΔΌ÷±Ϋ”Φ”»κΒμΖέ»ή“ΚΦλ―ιΡ≥ ≥―Έ «ΖώΈΣΦ”Ββ―ΈΘΜ
ΔΎ≥ΘΈ¬≥Θ―Ιœ¬Θ§14gΒΡC3H6ΚΆC4H8ΜλΚœΤχΧε÷–Κ§”–ΒΡ‘≠Ή” ΐΈΣ3NAΘΜ
Δέ‘ΎΆ®≥ΘΉ¥Ωωœ¬Θ§0.5molNO”κ0.5molO2ΜλΚœΚσΤχΧεΖ÷Ή” ΐΈΣ0.75NAΘΜ
Δή»τ―τΦΪΈΣΜν–‘ΒγΦΪ‘ρ”≈œ»Ζ≈ΒγΘ§»γCuΓΔFeΓΔAlΓΔAgΒ»Ής―τΦΪΨυΈΣΜν–‘ΒγΦΪΘ§“ρ¥ΥΕΦ «ΒγΦΪ”≈œ» ßΒγΉ”ΘΜ
Δί1molC10H22Ζ÷Ή”÷–Κ§”–ΒΡΙ≤ΦέΦϋΒΡΉή ΐΈΣ31NAΘΜ
Δό±ξΉΦΉ¥Ωωœ¬Θ§22.4LSO3÷–Κ§”–ΒΡΖ÷Ή” ΐΈΣNAΘΜ
ΔΏΥήΝœ¥ϋΓΔΖœ÷ΫΓΔΟΪΖΔΓΔœπΫΚ÷ΤΤΖΒ»Ψυ τ”Ύ”–ΜζΈοΘΜ
ΔύΦν–‘―θΜ·ΈοΕΦ «”…Ϋπ τ‘ΣΥΊΚΆ―θ‘ΣΥΊΉι≥…Θ§»γK2OΓΔCuOΓΔNa2OΓΔNa2O2ΓΔMn2O7ΓΔAl2O3ΓΔFe2O3Β»»Ϊ≤Ω”…Ϋπ τ‘ΣΥΊΚΆ―θ‘ΣΥΊΉι≥…ΒΡΘ§“ρ¥ΥΕΦΈΣΦν–‘―θΜ·ΈοΘ°
| AΘ° | ΔΌΔΎΔέΔίΔύ | BΘ° | ΔΌΔέΔόΔΏΔύ | CΘ° | ΔΌΔήΔόΔΏ | DΘ° | ΔΎΔήΔίΔΏ |
Ζ÷Έω ΔΌ”ωΒΫΒμΖέœ‘ ΨάΕ…Ϊ «ΒβΒΞ÷ ΒΡΧΊ–‘Θ§≤Μ «Ββ‘ΣΥΊΒΡΧΊ–‘ΘΜ
ΔΎC3H6ΚΆC4H8ΒΡΉνΦρ ΫœύΆ§ΕΦ «CH2Θ§Ψί¥ΥΦΤΥψΥυΚ§‘≠Ή”Ηω ΐΘΜ
Δέ“Μ―θΜ·ΒΣ”κ―θΤχΖ¥”Π…ζ≥…Εΰ―θΜ·ΒΣΘ§Εΰ―θΜ·ΒΣ≤ΩΖ÷ΉΣΜ·≥…ΥΡ―θΜ·ΕΰΒΣΘΜ
ΔήΜν–‘ΒγΦΪ ß»ΞΒγΉ”ΡήΝΠ«Ω”Ύ»ή“Κ÷–“θάκΉ”ΘΜ
Δί1mol C10H22Ζ÷Ή”÷–Κ§”–22molΧΦ«βΦϋΓΔ9molΧΦΧΦΦϋΘ§ΉήΙ≤Κ§”–31molΙ≤ΦέΦϋΘΜ
ΔόΤχΧεΡΠΕϊΧεΜΐ ”ΟΕ‘œσΈΣΤχΧεΘΜ
ΔΏΗυΨίΥήΝœ¥ϋΓΔΖœ÷ΫΓΔΨ…œπΫΚ÷ΤΤΖΒΡ≥…Ζ÷Ζ÷±πΈΣΚœ≥…ΗΏΖ÷Ή”Μ·ΚœΈοΓΔœΥΈ§ΥΊΓΔΚœ≥…ΗΏΖ÷Ή”≈–ΕœΘΜ
ΔύΦν–‘―θΜ·Έο”κΥ°Ζ¥”Π÷Μ…ζ≥…ΦνΘ°
Ϋβ¥π ΫβΘΚΔΌΒμΖέ”κΒβΒΞ÷ œ‘ ΨάΕ…ΪΘ§ ≥―Έ÷–≤ΜΚ§ΒβΒΞ÷ Θ§≤ΜΡή÷±Ϋ”Φ”»κΒμΖέ»ή“ΚΦλ―ιΡ≥ ≥―Έ «ΖώΈΣΦ”Ββ―ΈΘ§Ι ΔΌ¥μΈσΘΜ
ΔΎ14gΒΡC3H6ΚΆC4H8ΜλΚœΤχΧε÷–Κ§”–1molΉνΦρ ΫCH2Θ§Κ§”–3mol‘≠Ή”Θ§Κ§”–ΒΡ‘≠Ή” ΐΈΣ3NAΘ§Ι ΔΎ’ΐ»ΖΘΜ
Δέ‘ΎΆ®≥ΘΉ¥Ωωœ¬Θ§0.5mol NO”κ0.5mol O2ΜλΚœΚσ…ζ≥…0.5molΕΰ―θΜ·ΒΣΘ§―θΤχ Θ”ύ0.25molΘ§”…”Ύ≤ΩΖ÷Εΰ―θΜ·ΒΣΉΣΜ·≥…ΥΡ―θΜ·ΕΰΒΣΘ§Υυ“‘ΤχΧεΖ÷Ή” ΐ–Γ”Ύ0.75NAΘ§Ι Δέ¥μΈσΘΜ
ΔήCuΓΔFeΓΔAlΓΔAgΒ»Ής―τΦΪΘ§ΕΦ «Μν–‘ΒγΦΪΘ§Μν–‘ΒγΦΪ‘ρ”≈œ»Ζ≈ΒγΘ§Ι Δή’ΐ»ΖΘΜ
Δί1mol C10H22Ζ÷Ή”÷–Κ§”–9molΧΦΧΦΦϋΓΔ22molΧΦ«βΦϋΘ§ΉήΙ≤Κ§”–Ι≤ΦέΦϋΒΡΈο÷ ΒΡΝΩΈΣ31molΘ§Κ§”–ΒΡΙ≤ΦέΦϋΒΡΉή ΐΈΣ31NAΘ§Ι Δί’ΐ»ΖΘΜ
Δό±ξΩωœ¬Θ§»ΐ―θΜ·Νρ≤Μ «ΤχΧεΘ§≤ΜΡή Ι”ΟΤχΧεΡΠΕϊΧεΜΐΘ§Ι Δό¥μΈσΘΜ
ΔΏΥήΝœ¥ϋΓΔΨ…œπΫΚ÷ΤΤΖ τ”ΎΗΏΖ÷Ή”Μ·ΚœΈοΘ§Ζœ÷ΫΒΡ÷ς“Σ≥…Ζ÷ «÷≤ΈοΒΡœΥΈ§ «œΥΈ§ΥΊΘ§ΥϋΟ«ΕΦ τ”Ύ”–ΜζΈοΘ§Ι ΔΏ’ΐ»ΖΘΜ
ΔύΙΐ―θΜ·ΡΤ”κΥ°Ζ¥”Π≥ΐΝΥ…ζ≥…ΦνΆβΜΙ…ζ≥…―θΤχΘ§≤Μ τ”ΎΦν–‘―θΜ·ΈοΘ§Ι Δύ¥μΈσΘΜ
Ι ―ΓΘΚDΘ°
ΒψΤά ±ΨΧβΩΦ≤ιΑΔΖϋΦ”Β¬¬ό≥Θ ΐΒΡ”–ΙΊΦΤΥψΚΆ≈–ΕœΓΔΥαΦν―ΈΦΑ―θΜ·ΈοΒΡΖ÷άύΓΔ”–ΜζΈοΒΡ≈–ΕœΘ§≤ύ÷ΊΩΦ≤ι―ß…ζΕ‘Μυ¥Γ÷Σ ΕΒΡ’ΤΈ’ λΝΖ≥ΧΕ»Θ§ΧβΡΩΡ―Ε»≤Μ¥σΘ°

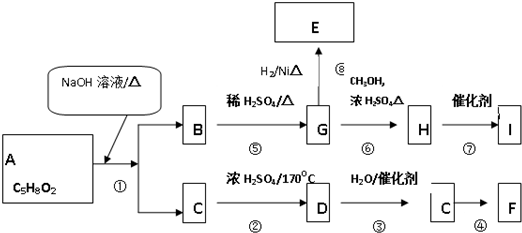
 ‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ
‘ΡΕΝΩλ≥ΒœΒΝ–¥πΑΗ| AΘ° | ”ΟFeCl3»ή“ΚΗ· ¥”ΓΥΔΒγ¬ΖΑεΘΚFe3++Cu®TFe2++Cu2+ | |
| BΘ° | œρΑ±Υ°÷–ΒΈΦ”¬»Μ·¬Ν»ή“ΚΘΚAl3++4OH-=AlO2-+2H2O | |
| CΘ° | ”Ο–ΓΥ’¥ρ÷ΈΝΤΈΗΥαΙΐΕύΘΚHCO3-+H+=CO2Γϋ+H2O | |
| DΘ° | ”Ο¥ΉΥα≥ΐ»ΞΥ°ΙΗΘΚCaCO3+2H+®TCa2++H2O+CO2Γϋ |
| AΘ° | ΦΉ±Ϋ | BΘ° | ΜΖΈλΆι | CΘ° | 2Θ§2-ΕΰΦΉΜυΕΓΆι | DΘ° | Ε‘ΕΰΦΉ±Ϋ |
| AΘ° | 8.1g | BΘ° | 6.75g | CΘ° | 5.4g | DΘ° | 2.7 g |
| AΘ° | dΓΔaΓΔbΓΔc | BΘ° | bΓΔaΓΔdΓΔc | CΘ° | dΓΔbΓΔaΓΔc | DΘ° | dΓΔcΓΔaΓΔb |
| AΘ° | ““¥ΦΘ®Υ°Θ©ΘΚΦ”»κ–¬÷ΤΒΡ…ζ ·Μ“Θ§»ΜΚσ’τΝσ | |
| BΘ° | ““Υα““θΞΘ®““ΥαΘ©ΘΚΦ”»κ±ΞΚΆΧΦΥαΡΤ»ή“ΚΘ§»ΜΚσΖ÷“Κ | |
| CΘ° | ““ΆιΘ®““œ©Θ©ΘΚΆ®»κΥα–‘ΗΏΟΧΥαΦΊ»ή“Κ÷–œ¥Τχ | |
| DΘ° | δε±ΫΘ®δεΘ©ΘΚΦ”»κ«β―θΜ·ΡΤ»ή“ΚΘ§»ΜΚσΖ÷“Κ |
| AΘ° | Ρ≥‘ΣΥΊΜυΧ§‘≠ΉνΆβ≤ψ”–3ΗωΈ¥≥…Ε‘ΒγΉ”Θ§ΉνΡΎ≤ψ”–2ΗωΒγΉ”Θ§Τδ‘ΣΥΊΖϊΚ≈ΈΣN | |
| BΘ° | Ρ≥‘ΣΥΊΒΡΗΚ“ΜΦέάκΉ”ΒΡΒγΉ”≤ψΫαΙΙ”κκ≤œύΆ§Θ§ΤδΜυΧ§‘≠Ή”ΦέΒγΉ”≈≈≤Φ ΫΈΣ[Ne]3s23p5 | |
| CΘ° | Ρ≥‘ΣΥΊΒΡ’ΐ»ΐΦέάκΉ”ΒΡ3dΡήΦΕΈΣΑκ≥δ¬ζΘ§ΤδΜυΧ§‘≠Ή”≈≈≤Φ ΫΈΣ[Ar]3d64s2 | |
| DΘ° | Ρ≥‘ΣΥΊΜυΧ§‘≠Ή”ΒΡM≤ψ»Ϊ≥δ¬ζΘ§N≤ψΟΜ”–≥…Ε‘ΒγΉ”Θ§÷Μ”–“ΜΗωΈ¥≥…Ε‘ΒγΉ”Θ§ΤδΜυΧ§‘≠Ή”ΒΡΒγΉ”≈≈≤Φ ΫΈΣ[Ar]3d54s1 |