��Ŀ����
����Ŀ������˵����ȷ����( )
A.NA���������ӵ�������1mol C4H10�к����ۼ���ĿΪ14NA
B.ij��Ӧ����H = ��88kJmol-1��������Ӧ���һ��С��88kJmol-1
C.��֪ij�¶��£�Kw = 1��10-13������pH =8��NaOH��Һ��pH =5��H2SO4��Һ��ϱ����¶Ȳ��䣬��ʹ�����ҺpH =7����NaOH��Һ��H2SO4��Һ�������Ϊ11��9
D.��Ũ��Ϊ0.1molL-1 HF��Һ��ˮ����ϡ�����У�����Ⱥ�Ka(HF)���ֲ��䣬![]() ʼ�ձ�������
ʼ�ձ�������
���𰸡�C
��������
A. 1mol C4H10�к�3��C��C��10��C��H���ۼ����ۼ���ĿΪ13NA����A����
B. �ʱ�Ĵ�С���������淴Ӧ�Ļ�ܵIJ�ֵ��ij��Ӧ����H = ��88kJmol-1��������Ӧ��ܲ�һ��С��88kJmol-1����B����
C. ˮ�����ӻ�����Ϊ1��10-13��![]() ��pH =6.5ʱ����Һ�����ԣ�pH =5��H2SO4��Һ��
��pH =6.5ʱ����Һ�����ԣ�pH =5��H2SO4��Һ��![]() ��pH =8��NaOH��Һ������
��pH =8��NaOH��Һ������![]() ��
��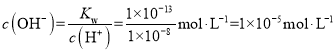 ��Ҫʹ���Һ��pH =7����Һ�ʼ��ԣ������������ӹ����������Һ�����������ӵ����ʵ���Ũ��
��Ҫʹ���Һ��pH =7����Һ�ʼ��ԣ������������ӹ����������Һ�����������ӵ����ʵ���Ũ��![]() ����������Һ��NaOH��Һ���������ΪX��Y����������Һ��Ϻ����OH-���ʵ���Ũ��Ϊ
����������Һ��NaOH��Һ���������ΪX��Y����������Һ��Ϻ����OH-���ʵ���Ũ��Ϊ![]() ������Y��X = 11:9����C��ȷ��
������Y��X = 11:9����C��ȷ��
D. ��Ũ��Ϊ0.1molL-1 HF��Һ��ˮ����ϡ�����У����������Ka(HF)���ֲ��䣬c��H+����c��F-����С��c��OH-��������Һ�еĵ���غ�c(H+)=c(F-)+c(OH-)��![]() =
=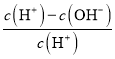 =1-
=1-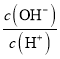 ������D����
������D����
������������ΪC��

 ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д�