��Ŀ����
��15�֣��л��������ұ���GB2760��2011���涨���Ѿ���SO2���ʹ����Ϊ0.25g/L��ij��ȤС������9ͼ1װ�ã��г�װ���ԣ��ռ�ij���Ѿ���SO2�����Ժ������Բⶨ��
��1������A��������_______��ˮͨ��A�Ľ���Ϊ_________��
��2��B�м���300.00ml���Ѿƺ��������ᣬ����ʹSO2ȫ���ݳ�����C��H2O2��ȫ��Ӧ���仯ѧ����ʽΪ______________��
��3����ȥC�й�����H2O2��Ȼ����0.0900mol/LNaOH����Һ���еζ����ζ�ǰ������ʱ��Ӧѡ����9��ͼ2�е�_____�����ζ��յ�ʱ��Һ��pH��8.8����ѡ���ָʾ��Ϊ__________������50ml�ζ��ܽ���ʵ�飬���ζ����е�Һ���ڡ�10�����������Һ������������ţ�____���٣�10ml���ڣ�40ml���ۣ�10ml���ܣ�40ml��
��4���ζ����յ�ʱ������NaOH��Һ25.00ml�������Ѿ���SO2����Ϊ��__g/L��
��5���òⶨ�����ʵ��ֵƫ�ߣ�����ԭ����������װ������Ľ���ʩ_______��
��1�������ܻ���������b ��2��SO2��H2O2��H2SO4 ��3���ۣ���̪���� ��4��0.24
��5��ԭ������Ļӷ����Ľ���ʩ���ò��ӷ���ǿ����������������ᣬ��������ˮ�������Ѿƽ��жԱ�ʵ�飬�۳�����ӷ���Ӱ�졣
���������������1�����������Ĺ����ص��֪������A�������ܻ�������������ʱ��ȴˮ������Ӧ�����¿ڽ����Ͽڳ�����ˮͨ��A�Ľ�����b��
��2��SO2���л�ԭ�ԣ�˫��ˮ���������ԣ�����Ϸ���������ԭ��Ӧ�������ᣬ��Ӧ�Ļ�ѧ����ʽΪSO2��H2O2��H2SO4��
��3�����������ǼӦ���ü�ʽ�ζ���ʢ�ţ���Һʱ�IJ���ѡ��ۡ��ζ��յ�ʱ��pH��8.8��˵����Һ�Լ��ԣ���ָʾ���ı�ɫ��Χ�ڼ��������£����ȵı�ɫ��Χ��3.1��4.4����̪�ı�ɫ��Χ��8.2��10.0������ѡ���ָʾ���Ƿ�̪�����ڵζ��ܵĿ̶����϶�����������������һ��û�п̶��ߣ���˸õζ�����ʵ����Һ������ǣ�50ml��10ml��40mol��
��4�����ݷ���ʽ��֪2NaOH��H2SO4��SO2����SO2��������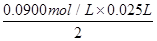 ��64g/mol��0.072g��������Ѿ���SO2�ĺ���Ϊ
��64g/mol��0.072g��������Ѿ���SO2�ĺ���Ϊ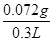 ��0.24g/L��
��0.24g/L��
��5�����������ǻӷ����ᣬ����Cװ�õ��������SO2����Ȼ��⣬�Ȼ���Ҳ���������Ʒ�Ӧ���Ӷ�ʹ����������������Һ��������ӣ����²ⶨ���ƫ�ߡ���˸Ľ��Ĵ�ʩΪ�ò��ӷ���ǿ�ᣬ��������������ᣬ��������ˮ�������Ѿƽ��жԱ�ʵ�飬�۳�����ӷ���Ӱ�졣
���㣺���黯ѧʵ�鷽�����������

 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�����ʵ������������ܹ��ﵽ��Ӧʵ��Ŀ�ĵ���
| | ʵ������ | ʵ��Ŀ�� |
| A | ��SO2ͨ��Ʒ����Һ�У�Ʒ����ɫ��������ɫ��Ʒ����Һ����Һ�ָ���ɫ | ֤��������IJ��ȶ��� |
| B | �����£���Na2CO3��Һ�м�����BaSO4��ĩ�����ˣ���ϴ���ij����м�ϡ���ᣬ���������ݲ��� | ֤��������KSP(BaSO4)��KSP(BaCO3) |
| C | �����²ⶨ���ʵ���Ũ����ͬ������ʹ�����Һ��pH������pHС�ڴ���pH | ֤����ͬ�����£���ˮ��HCl����̶ȴ���CH3COOH |
| D | ��Ũ�����̼��ϼ��ȣ�ֱ�ӽ����ɵ�����ͨ�������ij���ʯ��ˮ��ʯ��ˮ����� | �������������CO2�Ĵ��� |
������������(�ṹ��ʽ��ͼ)��һ�ּ�ǿ�����������Ȼ�����
��1������������������ˮ������е�����������X�����������X�ķ���ʽΪ ��
��2������Ч�ȡ�����ָ��KI����������ͬ����I2����Cl2��������ָ�������������֮�ȣ����ٷ�����ʾ��Ϊ�ⶨ������������ġ���Ч�ȡ��������ֳ�ȡij��������������Ʒ0.5680 g����ˮ������KI�����ᣬ���Ƴ�100 mL����Һ��ȷ��ȡ25.00 mL����Һ�ڵ���ƿ�У���0.1500 mol��L��1 Na2S2O3����Һ�ζ�����Һ����ɫʱ���������ָʾ���������ζ����յ�(������Ӧ�ķ���ʽΪ��2Na2S2O3��I2=Na2S4O6��2NaI)���ظ��ⶨ2�Σ����õ�����������±���
| �ζ���� | ����Һ���/mL | ��Һ�ζ���������/mL | ��Һ�ζ����յ����/mL |
| 1 | 25.00 | 0.06 | 24.04 |
| 2 | 25.00 | 0.02 | 24.02 |
| 3 | 25.00 | 0.12 | 24.14 |
�ٵζ��յ�۲쵽������Ϊ ��
������0.1500 mol��L��1 Na2S2O3��Һ100 mL������Na2S2O3��5H2O������Ϊ ��
�ۼ������Ʒ�ġ���Ч�ȡ�����(д���������)��
��16�֣�ij��ѧ��ȤС�����ʵ�飬��Ũ������ͭ��Ӧ�Ƶ�SO2���������ʵ��̽����
��1��װ��A�з�����Ӧ�Ļ�ѧ����ʽ
��_______��װ��B��������_______��
���װ��C��Ŀ������֤SO2��
_______________�ԣ�D��NaOHȫ��ת��Ϊ
NaHSO3�ı�־��____________��
��3����Ӧ����ʱ��ƿ��Cu��ʣ�࣬ij
ͬѧ��ΪH2SO4Ҳ��ʣ�࣬�����������ʵ
�鷽�����ⶨʣ��H2SO4����������ȴ����
��ϡ�ͺ��������ʵ�飬�ܴﵽĿ����________������ţ���
a�� ������к͵ζ����ⶨ b�� ������Zn��Ӧ����������H2�����
c�� ��PH�Ʋ���ҺPHֵ d�� ������BaCl2��Һ��Ӧ���������ɳ���������
��4����Dƿ����NaHSO3��Һ�м���Ư����Һ����Ӧ�����ֿ��������
I�� HSO3-��ClO-�պ÷�Ӧ�ꣻ II�� Ư�۲��㣻 III�� Ư�۹���
ͬѧ�Ƿֱ�ȡ���������Һ���Թ��У�ͨ������ʵ��ȷ���÷�Ӧ������һ���������������±���
| ʵ����� | ʵ����� | ���� | ��Ӧ�Ŀ������ |
| �� | �μ��������۵⻯����Һ���� | | III |
| �� | �μ������غ�ɫ��KI3��Һ���� | | II |
| �� | �������������KMNO4��Һ���� | ��Һ����ɫ | |
| �� | ���뼸С��CaCO3���� | �����ݲ��� | |
Ŀǰ���еĹ���������Դ�����������Ϊ,������Դ��Լ40����ǰ�Ĺ������Һ���������ֻ���ϵͳ���ձ��������ؽṹ,��Fe2S2��Fe4S4��Fe8S7��,��Щ����ؽṹ������������Դ����ط�Ӧ��ij��ѧ��ȤС�����о�ij����ؽṹ�����ʱ,���������ʵ�顣
��ʵ��� �������ȷ��: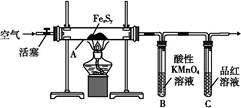
��ͼ����װ��,����װ�õ������Ժ�,��Ӳ�ʲ�����A�з���1.0 g����ؽṹ(���в��ֲ���Ӧ������),���Թ�B�м���50 mL 0.1 mol��L-1������KMnO4��Һ,���Թ�C�м���Ʒ����Һ��ͨ�����������,���ֹ�����ת��Ϊ����ɫ����������ȫת����B����Һת����250 mL����ƿ,ϴ���Թ�B���ݡ�ȡ25.00 mL����Һ��0.01 mol��L-1�IJ���(H2C2O4)���вⶨʣ��KMnO4��ҺŨ�ȵĵζ�����¼��������:
| �ζ����� | ������Һ ���/mL | ������Һ���/mL | |
| �ζ�ǰ�̶� | �ζ���̶� | ||
| 1 | 25.00 | 1.50 | 23.70 |
| 2 | 25.00 | 1.02 | 26.03 |
| 3 | 25.00 | 0.00 | 24.99 |
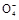 +2H2O+5SO2
+2H2O+5SO2 2Mn2++5S
2Mn2++5S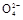 +4H+
+4H+��2Mn
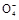 +6H++5H2C2O4
+6H++5H2C2O4 2Mn2++10CO2��+8H2O
2Mn2++10CO2��+8H2O��ʵ��� ��������ȷ��:
��ʵ���Ӳ�ʲ�����A�еIJ����������ϡ������,��ֽ�������,����Һ�м���������NaOH��Һ,���˺�ȡ����,��������յ�0.6 g���塣
�Իش���������:
(1)��顰ʵ�����װ�������Եķ����� ��
(2)�ζ��յ���жϷ����� ��
(3)�Թ�C��Ʒ����Һ�������� ��
��ͬѧ���,��ȥCװ��,��ʵ��û��Ӱ��,��Ŀ������������������������� (ѡ�ͬ�⡱��ͬ�⡱),������ ��
(4)����ʵ����ʵ����е����ݿ�ȷ��������ؽṹ�Ļ�ѧʽΪ ��
������̽���� �ζ�������,ϸ�ĵ�С�����ָ�KMnO4��ɫ��ȥ�����ʽ�ƽ���ζ�ʱҪ��öࡣΪ�о����ԭ��,��ͬѧ��������������ʵ��,ʵ���������±�:
| ��� | �¶�/�� | �ữ��H2C2O4 ��Һ/mL | KMnO4 ��Һ/mL | ��Һ�� ɫʱ��/s |
| 1 | 25 | 5.0 | 2.0 | 40 |
| 2 | 25 | 5.0(������������ ��ˮ��MnSO4��ĩ) | 2.0 | 4 |
| 3 | 60 | 5.0 | 2.0 | 25 |
(5)������������,�ζ������з�Ӧ���ʽϿ��һ�ֿ���ԭ���� ��
ij��ȤС�����þ���ˮ�ķ�Ӧ�Ʋ�þҲ���뱥��̼��������Һ��Ӧ��������ʾ��þ�뱥��̼��������Һ��Ӧ������������Ͱ�ɫ���������ȤС�����������ʵ�鷽������֤���̽����Ӧԭ����
ʵ��1����ɰֽ��ȥþ����������Ĥ���������ʢ���������з�̪�ı���̼��������Һ���Թ��У�Ѹ�ٷ�Ӧ�������������ݺͰ�ɫ�������Һ��dz���졣
��1���������
��ͬѧ�Է�Ӧ�в����İ�ɫ�������������¼��裺
����1������Ϊ ��
����2������ΪMgCO3��
����3�������Ǽ�ʽ̼��þ[xMgCO3��yMg(OH)2]
��2����ƶ���ʵ��ȷ�����ﲢ��֤�²⣺
| ʵ����� | ʵ�� | Ԥ������ͽ��� |
| ʵ��II | ��ʵ��I���ռ����������ȼ | |
| ʵ��III | ȡʵ��I�еİ�ɫ�����ϴ�ӣ��������� | �� ��ɫ��������ܺ���MgCO3 |
| ʵ��IV | ȡʵ��I�еij���Һ�������м�������CaCl2ϡ��Һ | ������ɫ��������Һ�д��� ���� |
��3����ƶ���ʵ��ȷ��ʵ��I�IJ����ȡʵ��I�����ø�������İ�ɫ������31.0 g����ּ��������ٲ�������Ϊֹ����ʹ�ֽ����������ȫ������ʢ������Ũ�����ϴ��ƿA��ʢ��������ʯ�ҵĸ����B��ʢ��������ʯ�ҵĸ����C�С�ʵ��ǰ��װ��A����1.8 g��װ��B����13.2 g����ȷ����ɫ������Ļ�ѧʽ ��
��4�����ϻ�ѧ����ͻ�ѧƽ���ƶ�ԭ������Mg��NaHCO3��Һ��Ӧ�����������ݵ�ԭ�� ��
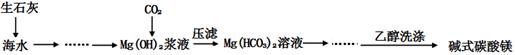
 ��x+y��MgO+xCO2��+��y+z��H2O
��x+y��MgO+xCO2��+��y+z��H2O
 Na2S2O3��aq�� ��III��
Na2S2O3��aq�� ��III��

 I2+5SO42��+H2O
I2+5SO42��+H2O