��Ŀ����
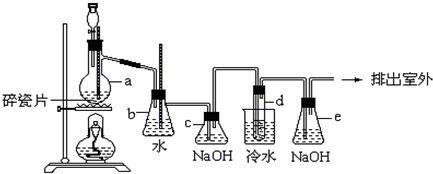
10����ѧС���������������������װ�ã�����ͼ�����û������Ʊ�����ϩ��
��֪��

| �ܶȣ�g/cm3�� | �۵㣨�棩 | �е㣨�棩 | �ܽ��� | |
| ������ | 0.96 | 25 | 161 | ������ˮ |
| ����ϩ | 0.81 | -103 | 83 | ������ˮ |
��12.5mL�����������Թ�A�У��ټ���1mLŨ���ᣬҡ�Ⱥ�������Ƭ����ֹ���У���������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
�ٵ���B���˵�������е�������������
���Թ�C���ڱ�ˮԡ�е�Ŀ���Ƿ�ֹ����ϩ�Ļӷ���
��2���Ʊ���Ʒ
�ٻ���ϩ��Ʒ�к��л������������������ʵȣ����뱥��ʳ��ˮ�������á��ֲ㣬����ϩ���ϲ㣨��ϡ����¡�������Һ����C�������ţ�ϴ�ӣ�
A��KMnO4��Һ B��ϡH2SO4C��Na2CO3��Һ
���ٽ�����ϩ����ͼװ��������ȴˮ��g�ڽ��루�g����f����������ʱҪ������ʯ�ң���Ŀ���dz�ȥˮ�֣�
���ռ���Ʒʱ�����Ƶ��¶�Ӧ��83�����ң�ʵ���ƵõĻ���ϩ��Ʒ�����������۲��������ܵ�ԭ����C��
A������ʱ��70�濪ʼ�ռ���Ʒ
B��������ʵ����������
C���Ʊ���Ʒʱ���������Ʒһ������
��3���������ֻ���ϩ��Ʒ�ʹ�Ʒ�ķ�������������BC��
A���ֱ�������Ը��������Һ
B���ֱ�����ý�����
C���ֱ�ⶨ�е㣮
���� ��1�����������ɵĻ���ϩ�ķе�Ϊ83�棬Ҫ�õ�Һ̬����ϩ������B���˵���������������ã����ڻ���ϩ������
�ڱ�ˮԡ��Ŀ���ǽ��ͻ���ϩ�������¶ȣ�ʹ��Һ����
��2���ٻ���ϩ�������Ȼ�����Һ�����ܶȱ�ˮС���ֲ��ϩ���ϲ㣬���ڷ�Һ��ϩ��Ʒ�л�������������ͻ��������ᴿ����ʱ��c��Na2CO3��Һ��ϴ�ӿɳ�ȥ�
��Ϊ����������Ч������ȴˮ���¿ڣ�g�����룻��ʯ������ˮ��Ӧ�����������ƣ����ڻ���ϩ��������
�۸��ݱ������ݿ�֪����ֻ���ϩ�ķе�Ϊ83�棻�ֲ�Ʒ�л��л����������²ⶨ���ĵĻ������������ƵõĻ���ϩ��Ʒ�����������۲�����
��3�����ݻ����û�й̶��ķе㣬���������й̶��ķе㣬ͨ���ⶨ����ϩ��Ʒ�ͻ���ϩ��Ʒ�ķе㣬���жϲ�Ʒ�Ĵ��ȣ�
��� �⣺��1�����������ɵĻ���ϩ�ķе�Ϊ83�棬Ҫ�õ�Һ̬����ϩ������B���˵���������������ã����ڻ���ϩ������
�ʴ�Ϊ��������
�ڱ�ˮԡ��Ŀ���ǽ��ͻ���ϩ�������¶ȣ�ʹ��Һ�������ٻӷ�
�ʴ�Ϊ����ֹ����ϩ�Ļӷ���
��2���ٻ���ϩ�����࣬�������Ȼ�����Һ�����ܶȱ�ˮС�������á��ֲ��ϩ���ϲ㣬���ڷ�Һ��ϩ��Ʒ�л�������������ͻ����������룺�Ʊ����������ᴿ����ʱ��c��Na2CO3��Һ��ϴ�ӿɳ�ȥ�
�ʴ�Ϊ���ϣ�c��
��Ϊ����������Ч��������װ��Ҫ�������ܣ���ȴˮ���¿ڣ�g�����룬��ʯ������ˮ��Ӧ�����������ƣ���ȥ�˲�����ˮ���õ������Ļ���ϩ��
�ʴ�Ϊ��g����ȥˮ�֣�
�۸��ݱ������ݿ�֪����ֻ���ϩ�ķе�Ϊ83�棬���ռ���ƷӦ�����¶���83�����ң����ֲ�Ʒ�л��л����������²ⶨ���ĵĻ������������ƵõĻ���ϩ��Ʒ�����������۲�����
�ʴ�Ϊ��83�棻C��
��3�������Ʒ�뾫Ʒ�ɼ�������ƣ��۲��Ƿ�������������������壬���Ǿ�Ʒ�������û�й̶��ķе㣬���������й̶��ķе㣬ͨ���ⶨ����ϩ��Ʒ�ͻ���ϩ��Ʒ�ķе㣬���жϲ�Ʒ�Ĵ��ȣ�
�ʴ�Ϊ��BC��
���� �������л��ϳ�Ϊ�����ۺϿ�����ʵ�����ƻ���ϩ��֪ʶ�ʹ����������ʣ�������ͬѧ���������ʵ�����������Ŀ�Ѷ��еȣ�ע�����ʵ��ԭ���ͷ������ر���ʵ��Ļ���������ѧϰ��ע����ۣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | HCl��Na2CO3��HNO3��KHCO3 | B�� | BaCl2��Na2SO4��Ba��OH��2��CuSO4 | ||
| C�� | HCl��KOH��H2SO4��Ba��OH��2 | D�� | H2SO4��NaHCO3��NaHSO4��KHCO3 |
��1������ͼ1�����ڻ�����Ũ��ˮ����ʯ����ȡ������װ�ü�ͼ��
��2���Ʊ���������淋�װ����ͼ2��ʾ�����ɵİ��������С�������������Ȼ�̼�У�
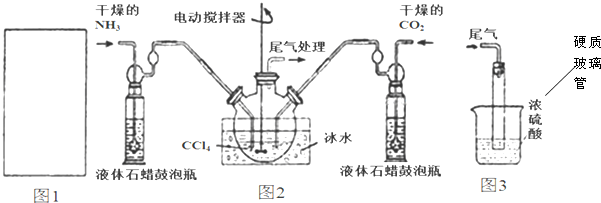
�ٴӷ�Ӧ��Ļ�����з������Ʒ��ʵ�鷽���ǹ��ˣ���������ƣ���
��ͼ3��Ũ���������������δ��Ӧ�İ�������ֹ�����е�ˮ�������뷴Ӧ��ʹ���������ˮ�⣮
��3����һ���������İ�������粒����������Ƶ��ܱ���������У���������������䣬��������������Բ��ƣ����ں㶨�¶���ʹ��ﵽ�ֽ�ƽ�⣮ʵ���ò�ͬ�¶��µ�ƽ�����������±���
| �¶ȣ��棩 | 15.0 | 20.0 | 25.0 | 30.0 | 35.0 |
| ƽ����ѹǿ��kPa�� | 5.7 | 8.3 | 12.0 | 17.1 | 24.0 |
| ƽ��������Ũ�ȣ���10-3mol/L�� | 2.4 | 3.4 | 4.8 | 6.8 | 9.4 |
A���ܱ������ڻ�������ѹǿ����
B���ܱ���������������������
C���ܱ������л��������ܶȲ���
D���ܱ������а����������������
�ڸ÷ֽⷴӦ���ʱ��H��0�����������=����������25.0��ʱ�ֽ�ƽ�ⳣ����ֵ=1.6384��10-8��
��ȡһ�����İ�������粒������һ�����������ܱ���������У���25.0���´ﵽ�ֽ�ƽ�⣮���ں�����ѹ�������������������粒�������������ӣ�����ӡ��������١����䡱����
�ٵ��������������ڴ����еĺ������������ʱ�������������ȣ��漰���·�Ӧ��
2NO2��g��+NaCl��s��?NaNO3��s��+ClNO��g����H1 K1
4NO2��g��+2NaCl��s��?2NaNO3��s��+2NO��g��+Cl2��g����H2 K2
2NO��g��+Cl2��g��?2ClNO��g����H3 K3
���H3=2��H1-��H2���á�H1�͡�H2��ʾ����K3=$\frac{K_1^2}{K_2}$����K1��K2��ʾ����
��NaOH��Һ�������յ��������NO2+NO+2NaOH=2NaNO2+H2O�������£���һ������NaOH
��Һ����NO��NO2������壬�õ�0.1mol/L��NaNO2��Һ����֪HNO2�ĵ��볣��Ka=7.1��10-4��
��ô������NO2-��ˮ�ⳣ��Kh=1.4��10-11��0.1mol/L NaNO2��Һ�д�c��Na+ ����c��OH- ����c��NO2- ����c��HNO2���Ĵ�С˳��Ϊc��Na+ ����c��NO2-����c��OH-����c��HNO2����
��2����3.0L�ܱ������У�ͨ��0.10mol CH4��0.20mol NO2����һ���¶��½��з�Ӧ��CH4��g��+2NO2��g��?CO2��g��+N2��g��+2H2O��g����H��0����Ӧʱ�䣨t����������������ѹǿ��p�������ݼ��±���
| ʱ��t/min | 0 | 2 | 4 | 6 | 8 | 10 |
| ��ѹǿp/100kPa | 4.80 | 5.44 | 5.76 | 5.92 | 6.00 | 6.00 |
����һ����װ���У�ͨ��һ����CH4��NO2���������ͬʱ���ڣ��ڲ�ͬ�¶��£�NO2��ת������ͼ��������Ϊ��Ӧ�¶ȣ�������ΪNO2ת����/%����
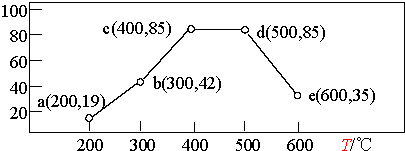
������������ȷ����AD��
A�����¶�ά����200�����ʱ�䣬NO2��ת���ʽ�����19%
B����Ӧ���ʣ�b���v ���棩��e��� v ���棩
C��ƽ�ⳣ����c��=d��
D�����b��ʱNO2��ת���ʺͷ�Ӧ���ʣ����ʵ����»�����c��CH4��