��Ŀ����
�ྦྷ����̫���ܹ����ҵ����Ҫԭ���ϡ�
(1)��ʯӢɰ����ȡ�ֹ裬����ط�Ӧ���Ȼ�ѧ����ʽ���£�
SiO2(s)��C(s)=SiO(g)��CO(g)����H��a kJ��mol��1
2SiO(g)=Si(s)��SiO2(s)����H��b kJ��mol��1
�ٷ�ӦSiO2(s)��2C(s)=Si(s)��2CO(g)�Ħ�H��________ kJ��mol��1(�ú�a��b�Ĵ���ʽ��ʾ)��
��SiO�Ƿ�Ӧ�����е��м�����������ʱ��SiO��NaOH��Һ��Ӧ(����֮һ�ǹ�����)�Ļ�ѧ����ʽΪ________________________________��
(2)�ֹ��ᴿ��������֮һ���Ƚ��ֹ���HCl�Ƶ�SiHCl3�����ᴿ������H2��ԭ��
SiHCl3(g)��H2(g) Si(s)��3HCl(g)��
Si(s)��3HCl(g)��
��ͬ�¶ȼ���ͬ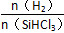 ʱ����Ӧ��X��ƽ��ת���ʹ�ϵ��ͼ��ʾ��
ʱ����Ӧ��X��ƽ��ת���ʹ�ϵ��ͼ��ʾ��

��X��________(�H2����SiHCl3��)��
��������Ӧ��ƽ�ⳣ��K(1 150 ��)________K(950 ��)(�>������<������)��
(3)SiH4(����)�������ߴ��ྦྷ���Ƿdz�����ķ�����
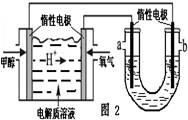
���ôֹ���ԭ�ϣ������ε�ⷨ��ȡ����ԭ����ͼ��ʾ�����ʱ�����ĵ缫��ӦʽΪ_________________________________________��
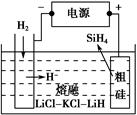
�ڹ��̫���������N��Si����Ԫ����ɵĻ�����Y���ۻ����ϣ�������SiH4��NH3������������������õ�����֪Y��Si����������Ϊ60%��Y�Ļ�ѧʽΪ________��
(1)��ʯӢɰ����ȡ�ֹ裬����ط�Ӧ���Ȼ�ѧ����ʽ���£�
SiO2(s)��C(s)=SiO(g)��CO(g)����H��a kJ��mol��1
2SiO(g)=Si(s)��SiO2(s)����H��b kJ��mol��1
�ٷ�ӦSiO2(s)��2C(s)=Si(s)��2CO(g)�Ħ�H��________ kJ��mol��1(�ú�a��b�Ĵ���ʽ��ʾ)��
��SiO�Ƿ�Ӧ�����е��м�����������ʱ��SiO��NaOH��Һ��Ӧ(����֮һ�ǹ�����)�Ļ�ѧ����ʽΪ________________________________��
(2)�ֹ��ᴿ��������֮һ���Ƚ��ֹ���HCl�Ƶ�SiHCl3�����ᴿ������H2��ԭ��
SiHCl3(g)��H2(g)
 Si(s)��3HCl(g)��
Si(s)��3HCl(g)����ͬ�¶ȼ���ͬ
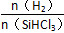 ʱ����Ӧ��X��ƽ��ת���ʹ�ϵ��ͼ��ʾ��
ʱ����Ӧ��X��ƽ��ת���ʹ�ϵ��ͼ��ʾ��
��X��________(�H2����SiHCl3��)��
��������Ӧ��ƽ�ⳣ��K(1 150 ��)________K(950 ��)(�>������<������)��
(3)SiH4(����)�������ߴ��ྦྷ���Ƿdz�����ķ�����
���ôֹ���ԭ�ϣ������ε�ⷨ��ȡ����ԭ����ͼ��ʾ�����ʱ�����ĵ缫��ӦʽΪ_________________________________________��

�ڹ��̫���������N��Si����Ԫ����ɵĻ�����Y���ۻ����ϣ�������SiH4��NH3������������������õ�����֪Y��Si����������Ϊ60%��Y�Ļ�ѧʽΪ________��
��(1)��2a��b����SiO��2NaOH=Na2SiO3��H2��
(2)��SiHCl3����>
(3)��Si��4H����4e��=SiH4������Si3N4
(2)��SiHCl3����>
(3)��Si��4H����4e��=SiH4������Si3N4
��(1)�ٷ�ӦSiO2(s)��2C(s)=Si(s)��2CO(g)���ɵ�һ����Ӧ��2���ڶ�����Ӧ�õ�����H��(2a��b)kJ��mol��1���ڸ��ݲ����й����Ƽ������غ�õ���Ӧ�Ļ�ѧ����ʽΪSiO��2NaOH=Na2SiO3��H2����(2)�ٸ���ͼ�����¶Ȳ��������£�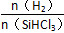 ����X��ƽ��ת����������X��SiHCl3���ڵ�
����X��ƽ��ת����������X��SiHCl3���ڵ�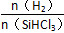 һ��ʱ�������¶ȣ�SiHCl3��ƽ��ת���������������ѧƽ�ⳣ��K(1 150 ��)>K(950 ��)��(3)�ٵ��ʱ��������������Ӧ���ֹ�ʧȥ���ӣ����H������SiH4���壬��缫��ӦʽΪSi��4H����4e��=SiH4������Si��NԪ����ɵĻ�������Si����������Ϊ60%����
һ��ʱ�������¶ȣ�SiHCl3��ƽ��ת���������������ѧƽ�ⳣ��K(1 150 ��)>K(950 ��)��(3)�ٵ��ʱ��������������Ӧ���ֹ�ʧȥ���ӣ����H������SiH4���壬��缫��ӦʽΪSi��4H����4e��=SiH4������Si��NԪ����ɵĻ�������Si����������Ϊ60%����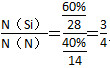 ������ѧʽΪSi3N4��
������ѧʽΪSi3N4��
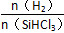 ����X��ƽ��ת����������X��SiHCl3���ڵ�
����X��ƽ��ת����������X��SiHCl3���ڵ�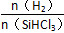 һ��ʱ�������¶ȣ�SiHCl3��ƽ��ת���������������ѧƽ�ⳣ��K(1 150 ��)>K(950 ��)��(3)�ٵ��ʱ��������������Ӧ���ֹ�ʧȥ���ӣ����H������SiH4���壬��缫��ӦʽΪSi��4H����4e��=SiH4������Si��NԪ����ɵĻ�������Si����������Ϊ60%����
һ��ʱ�������¶ȣ�SiHCl3��ƽ��ת���������������ѧƽ�ⳣ��K(1 150 ��)>K(950 ��)��(3)�ٵ��ʱ��������������Ӧ���ֹ�ʧȥ���ӣ����H������SiH4���壬��缫��ӦʽΪSi��4H����4e��=SiH4������Si��NԪ����ɵĻ�������Si����������Ϊ60%����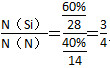 ������ѧʽΪSi3N4��
������ѧʽΪSi3N4��
��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ


 2NH3��g����H1��0��ʵ�����c��N2����ʱ�䣨t���ı仯��ͼ��ʾ��T��ʾ�¶ȣ���
2NH3��g����H1��0��ʵ�����c��N2����ʱ�䣨t���ı仯��ͼ��ʾ��T��ʾ�¶ȣ��� NH3?H2O��aq��+H����aq���Ļ�ѧƽ�ⳣ��Ϊ5.55��10��10 mol��L-1����NH3?H2O�ĵ���ƽ�ⳣ��K= ��������λ��Ч���֣���
NH3?H2O��aq��+H����aq���Ļ�ѧƽ�ⳣ��Ϊ5.55��10��10 mol��L-1����NH3?H2O�ĵ���ƽ�ⳣ��K= ��������λ��Ч���֣��� CH3OH(g)��H2O(g�� ��H3
CH3OH(g)��H2O(g�� ��H3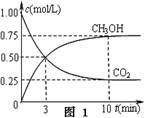
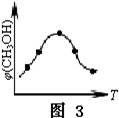
 2NH3(g)��������������ʱ����N2������Ӧ���������淴Ӧ���ʼ�С
2NH3(g)��������������ʱ����N2������Ӧ���������淴Ӧ���ʼ�С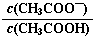 ��ֵ��С
��ֵ��С H2��I2
H2��I2