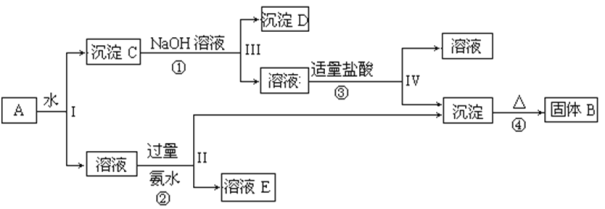
��Ŀ����
����Ŀ��ij�¶��£���һ��2 L���ܱ������У�����4 mol A��2 mol B�������·�Ӧ��3A(g)��2B(g)![]() 4C(s)��D(g)����Ӧ2 min��ﵽƽ�⣬�������1.6 mol C������˵����ȷ����
4C(s)��D(g)����Ӧ2 min��ﵽƽ�⣬�������1.6 mol C������˵����ȷ����
A.ǰ2 min��D��ƽ����Ӧ����Ϊ0.2 mol��L��1��min��1
B.��ʱ��B��ƽ��ת������40%
C.�������ϵ��ѹǿ��ƽ�ⲻ�ƶ�
D.����B��ƽ�������ƶ���B��ƽ��ת��������
���𰸡�B
��������
A�����ݷ���ʽ��֪����Ӧ2 min��ﵽƽ�⣬�������1.6 mol C�������ɵ�D�����ʵ���Ϊ0.4 mol��ǰ2 min��D��ƽ����Ӧ����Ϊ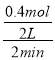 ��0.1 mol��L��1��min��1��ѡ��A����
��0.1 mol��L��1��min��1��ѡ��A����
B�����ݷ���ʽ��֪���μӷ�Ӧ��B�����ʵ���Ϊ0.8mol����B��ת����=![]() ��100%=40%��ѡ��B��ȷ��
��100%=40%��ѡ��B��ȷ��
C���÷�Ӧ������Ӧ���������С������ѹǿƽ�������ƶ���ѡ��C����
D������B��������B��Ũ������ƽ��������Ӧ�����ƶ���A��ת��������B��ת���ʽ��ͣ�ѡ��D����
��ѡB��

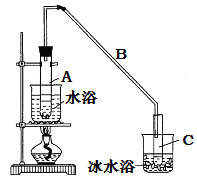
����Ŀ��ij��ѧС���������������������װ��(��ͼ)���Ի������Ʊ�����ϩ
��֪��![]()
![]()
![]() +H2O
+H2O
�ܶ�(g/cm3) | �۵�(��) | �е�(��) | �ܽ��� | |
������ | 0.96 | 25 | 161 | ������ˮ |
����ϩ | 0.81 | -103 | 83 | ������ˮ |
(1)�Ʊ���Ʒ����12.5 mL�����������Թ�A�У��ټ���l mLŨ���ᣬҡ�Ⱥ�������Ƭ��������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
��A�����Ƭ��������_________________________��
�ڵ���B���˵�������е�������_____________��
(2)�Ʊ���Ʒ��
�ٻ���ϩ��Ʒ�к��л������������������ʵȡ����뱥��ʳ��ˮ�������á��ֲ㣬����ϩ��_________��(���ϻ���)����Һ����_________ (������)ϴ�ӡ�
a.KMnO4��Һ b.ϡH2SO4 c.Na2CO3��Һ
���ٽ�����ϩ����ͼװ��������ȴˮӦ��_________�ڽ��룬Ŀ����ʹ��ȴˮ�������γ���������
���ռ���Ʒʱ�����Ƶ��¶�Ӧ��_________���ң�ʵ���ƵõĻ���ϩ��Ʒ�����������۲��������ܵ�ԭ����__________��
a������ʱ��70 �濪ʼ�ռ���Ʒ
b��������ʵ����������
c���Ʊ���Ʒʱ���������Ʒһ������