��Ŀ����
(1)������ɴ���̫��վ�й������Ա�����ܱյĹ���������Ҫ��������������CO2���ɴ��ڿ���ʹ��һ�����ʣ���������CO2������������������Ҫ��Ӧ�ķ���ʽΪ_______________��������560�������������ʱ����Ӧ��ת�Ƶ��ӵ���Ŀ��___________��
(2)��NaOH��Һ��ClO2��H2O2��Ӧ���Եõ��������ƣ�NaClO2����ClO2����Ԫ���Ļ��ϼ���__________�����Ʊ��������Ƶķ�Ӧ��ClO2��___________(�����������ԭ������������Ӧ������1molNaClO2��ת�Ƶ������ʵ���Ϊ__________mol��
(3)��Һ�ĵ���������С��Ҫ��������Ũ�ȴ�С�����ģ����á�Һ�嵼���Ե�ʵ��װ�á�����ͼ��ʾ��������ʵ�飬�����ܳ��֣���һ��һϨ��һ�����������__________��

A����BaCl2��Һ�е���ϡH2SO4
B����Ba(OH)2��Һ�е���ϡH2SO4
C����NaOH��Һ�е������ᣬд����ѡ������ӷ�Ӧ����ʽ_______________��
(4)��֪���������ռ���Һ��Ӧ���£�3Cl2+6NaOH=NaClO3+5NaCl+3H2O��
����˫���ŷ��������ת�Ƶķ������Ŀ��
�ڵ���0.1mol����ת��ʱ���μӷ�Ӧ��Cl2�ڱ�״���µ����_______mL��


 ��
�� ����������ͬ��������ͬ��ͬ�ֺ���
����������ͬ��������ͬ��ͬ�ֺ��� 3
3
 ��˵����ȷ����( )
��˵����ȷ����( ) ��Na2O���ʵ�Na2O2����������ͼ��ʵ��װ�òⶨNa2O2�����Ĵ��ȣ�ʵ��ʱ����Ʒ������D�С�(�ɹ�ѡ�õķ�Ӧ��ֻ��CaCO3���塢6 mol/L���ᡢ6 mol/L���������ˮ)
��Na2O���ʵ�Na2O2����������ͼ��ʵ��װ�òⶨNa2O2�����Ĵ��ȣ�ʵ��ʱ����Ʒ������D�С�(�ɹ�ѡ�õķ�Ӧ��ֻ��CaCO3���塢6 mol/L���ᡢ6 mol/L���������ˮ)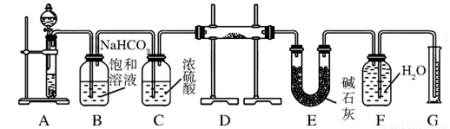
 ��
��