��Ŀ����
����пΪ��ɫ��ĩ��������ʪ�Ѣ��Ƥ���������ơ�������ҵ������п(����Fe(��), Mn(��), Ni(��)������)����������:
��ҵZnO ����Һ
����Һ
 ��Һ
��Һ
 ��Һ
��Һ
 �˱�
�˱� ZnO
ZnO
��ʾ���ڱ�ʵ�������£�Ni(��)���ܱ�������������صĻ�ԭ������MnO2
�ش���������:
��1����Ӧ���г��������������� ��������Ӧ�����ӷ���ʽΪ ��
�Ӹ��������Һǰ����pH�ϵͣ��Գ��ӵ�Ӱ���� ��
��2����Ӧ�۵ķ�Ӧ����Ϊ �����˵õ��������У����˹�����п��� ��
��3����Ӧ���γɵij���Ҫ��ˮϴ����������Ƿ�ϴ�Ӹɾ��ķ����� ��
��4����Ӧ���в���ijɷֿ�����ZnCO3��xZn(OH)2 .ȡ�ɲٺ���˱�11.2g�����պ�ɵõ���Ʒ8.1 g. ��x���� ��
��ҵZnO
 ����Һ
����Һ
 ��Һ
��Һ
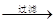 ��Һ
��Һ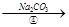
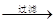 �˱�
�˱�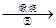 ZnO
ZnO��ʾ���ڱ�ʵ�������£�Ni(��)���ܱ�������������صĻ�ԭ������MnO2
�ش���������:
��1����Ӧ���г��������������� ��������Ӧ�����ӷ���ʽΪ ��
�Ӹ��������Һǰ����pH�ϵͣ��Գ��ӵ�Ӱ���� ��
��2����Ӧ�۵ķ�Ӧ����Ϊ �����˵õ��������У����˹�����п��� ��
��3����Ӧ���γɵij���Ҫ��ˮϴ����������Ƿ�ϴ�Ӹɾ��ķ����� ��
��4����Ӧ���в���ijɷֿ�����ZnCO3��xZn(OH)2 .ȡ�ɲٺ���˱�11.2g�����պ�ɵõ���Ʒ8.1 g. ��x���� ��
��1��Fe2+��Mn2+ �����Ӻ������Ӳ������ɳ������Ӷ�����ȥ����������
��2���û���Ӧ ��
��3��ȡ����ˮϴҺ���Թ��У�����1-2��ϡ���ᣬ�ٵ������ᱵ��Һ�����ް�ɫ�������ɣ���˵�������Ѿ�ϴ�Ӹɾ�
��4��1
��1���������⣬Ni(��)���ܱ���������Ӧ���г���������������Fe2+��Mn2+�����������ӷ���ʽΪMnO4-+3 Fe2++7H2O="3" Fe(OH)3��+ MnO2+5H+��2MnO4-+3Mn2++2H2O=5MnO2��+4H+���Ӹ��������Һǰ����pH�ϵͣ������Ӻ������Ӳ������ɳ������Ӷ�����ȥ���������ʡ�
��2����Ӧ��Ϊп�������ӵķ�Ӧ����Ӧ����Ϊ�û���Ӧ���õ��������У����˹�����п��н�������
��3����������Ƿ�ϴ�Ӹɾ��ķ�����ȡ����ˮϴҺ���Թ��У�����1-2��ϡ�����ữ���ٵ������ᱵ��Һ�����ް�ɫ�������ɣ�˵��û����������ӣ���˵�������Ѿ�ϴ�Ӹɾ�
��4�����ݹ�ϵʽZnCO3��xZn(OH)2 -------------- ��x+1��ZnO
125+99x 81��x+1��
11.2g 8.1g
��ã�x=1
�����㶨λ���������̡���ѧʵ�顢��ѧ����

��ϰ��ϵ�д�
�����Ŀ
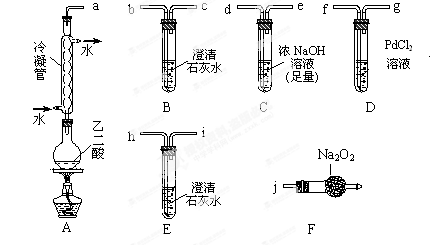
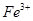 ����ܺ���
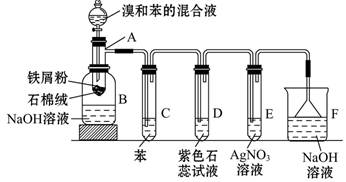
����ܺ���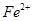 ����Ҫȷ�����е�
����Ҫȷ�����е�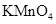 ��Һ
��Һ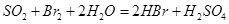 ��Ȼ���������
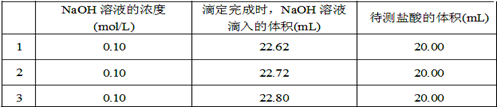
��Ȼ��������� ��Һ�����ʵ�������ø������2��33g�����ڴ���֪����Y��
��Һ�����ʵ�������ø������2��33g�����ڴ���֪����Y�� ���������Ϊ ��
���������Ϊ �� ��Q���塣Ϊ�����������̽��ʵ��״�ã�ͼ�мг�����ʡ�ԣ���
��Q���塣Ϊ�����������̽��ʵ��״�ã�ͼ�мг�����ʡ�ԣ���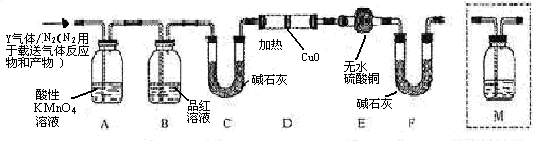
 O2(g) �� H2O(g) ��H����241.8 kJ��mol��1
O2(g) �� H2O(g) ��H����241.8 kJ��mol��1