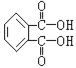
��Ŀ����
����Ŀ����Ԫ�������ˮ�帻Ӫ��������Ҫԭ����ˮ�г�������NO3��ʽ���ڡ�
��1����pHΪ4��6ʱ����H2��Pd-Cu���½�NO3-��ԭΪN2������ˮ��NO3���÷�Ӧ�����ӷ���ʽΪ____������H2��CO2�Ļ���������H2��NO3-ȥ��Ч�����ã���ԭ����____��
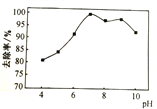
��2��NaClO�����ɳ�ȥ��������Ӧ������ͼ1��ʾ������H2O��NaCl��ȥ����ʵ������ͬ�����£���ͬ��Ӧʱ�䣬pH�백����ȥ���ʹ�ϵ��ͼ2��ʾ���¶��백��ȥ���ʹ�ϵ��ͼ3��ʾ��
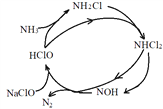

ͼ1 ͼ2 ͼ3
��NaClO����NH3���ܷ�Ӧ�Ļ�ѧ����ʽΪ____��
����ͼ2��ʾ����pH��9ʱ��pHԽ��ȥ����ԽС����ԭ����____��
����ͼ3��ʾ���¶ȵ���15��ʱ���¶�Խ��ȥ����Խ����ԭ����____�����¶ȸ���25��ʱ���¶�Խ��ȥ����ҲԽ�ͣ���ԭ����____��
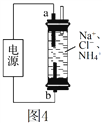
��3���õ绯ѧ����ȥ����ˮ�еİ������ں�NH4+�ķ�ˮ�м����Ȼ��ƣ��ö��Ե缫��⡣��Ӧװ����ͼ4��ʾ������ʱ��a���ĵ缫��ӦʽΪ____��
���𰸡� 2NO3�C + 5H2 + 2H+ ��N2��+ 6H2O CO2������Һ�У���ά��pH��4��6֮�� 2NH3 + 3NaClO��N2��+ 3H2O + 3NaCl ClO��Ũ�ȼ�С��������Ӧ���ʼ��� �¶ȵ�ʱ��Ӧ������ �¶ȸ�ʱ���ӿ���HClO�ķֽ⣨��NaClO�ֽ⣩�������ʽ��� 2H2O + 2NH4+ + 2e�C��2NH3��H2O + H2����2H+ + 2e�C��H2��
����������������1�������ܽ��������ԭΪ��������Һ�����ԣ�˵����H+���뷴Ӧ����������������ˮ����2������ͼʾ�ҳ���Ӧ����������3������ͼ�е��ԭ�����
�������1�������ܽ��������ԭΪ��������Һ�����ԣ�˵����H+���뷴Ӧ����������������ˮ����дʱע�ⷴӦ���������ʴ�Ϊ��2NO3�C + 5H2 + 2H+ ��N2��+ 6H2O����������������Ϊ�����pHΪ4��6������H2��CO2�Ļ���������H2��CO2������Һ�У���ά��pH��4��6֮�䣬ʹNO3-ȥ��Ч�����ã���ˣ������Ϊ��2NO3�C + 5H2 + 2H+ ��N2��+ 6H2O ��CO2������Һ�У���ά��pH��4��6֮�䡣
��2������ͼ֪��NaClO����NH3����N2��H2O��NaCl,���ݵ�ʧ�����غ㡢ԭ���غ�д����Ӧ�Ļ�ѧ����ʽΪ2NH3 + 3NaClO��N2��+ 3H2O + 3NaCl��
����pH��9ʱ��ClO-��Ũ�ȼ�С��������Ӧ���ʼ�����ȥ���ʽ��͡�
����ͼ֪�¶ȵ���15��ʱ����Ӧ��������ȥ���ʽ��͡����¶ȸ���25��ʱ���ӿ���HClO�ķֽ⣨��NaClO�ֽ⣩�������ʽ��͡�
��ˣ�������ȷ��Ϊ��NH3 + 3NaClO��N2��+ 3H2O + 3NaCl ��ClO-��Ũ�ȼ�С��������Ӧ���ʼ��� ���¶ȵ�ʱ��Ӧ������ �� �¶ȸ�ʱ���ӿ���HClO�ķֽ⣨��NaClO�ֽ⣩�������ʽ��͡�
��3����ͼ��֪���ó�Ϊ���أ�aΪ������bΪ������a��������ԭ��Ӧ���缫��ӦʽΪ��2H2O + 2NH4+ + 2e�C��2NH3��H2O + H2����2H+ + 2e�C��H2������ˣ������Ϊ��2H2O + 2NH4+ + 2e�C��2NH3��H2O + H2����2H+ + 2e�C��H2����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�