��Ŀ����
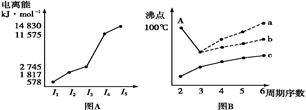
(9��)Ԫ����������ָ������ѧϰԪ�ؼ��仯����֪ʶ����Ҫ���ߡ���֪����Ԫ�أ�����Po���IJ���֪ʶ���±���ʾ��
| Ԫ�� | 8O | 16S | 34Se | 52Te |
| �����۵㣨�棩 | -218.4 | 113 | | 450 |
| ���ʷе㣨�棩 | -183 | 444.6 | 685 | 1390 |
| Ԫ����Ҫ���ϼ� | -2 | -2,+4,+6 | -2,+4,+6 | |
| ԭ�Ӱ뾶 | ������ | |||
| ������H2��Ӧ��� | ��ȼʱ���� | ���Ȼ��� | �����ѻ��� | ����ֱ�ӻ��� |
��1�����������۵㷶Χ������________________��
��2��Ԫ���ڵ���Ҫ���ϼۿ�����________________��
��3���������ڵ��⻯��ˮ��Һ��������ǿ������˳����________________(�û�ѧʽ��ʾ)��
��4���������н�ǿ��__________��������ԡ���ԭ�ԡ��������¶���ڿ����г��ڱ����ױ��ʣ�����ܷ�����Ӧ�Ļ�ѧ����ʽΪ_________________________________��
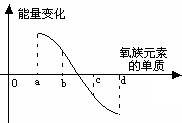
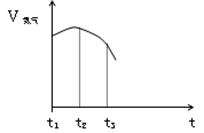
��5����ͼ��ʾΪ����Ԫ�ص�����H2��Ӧ�����е������仯ʾ��ͼ������a��b��c��d�ֱ��ʾ����Ԫ����ijһԪ�صĵ��ʣ�������Ϊ��ͬ���ʵ����ĵ�����H2��Ӧ�����е������仯�������仯��0��ʾ���ȣ������仯��0��ʾ���ȣ�����b����___________ _____�� d���� (��д��������)��
��1������113�棬С��450��(1��) ��2����2��+4��+6 (1��)
��3��H2Te��H2Se��H2S (2��) ��4����ԭ��(1��)��2H2Se+O2==2Se+2H2O(2��)
��5����(1��)����(1��)��
���������������1������������Ԫ���е����۵�ı仯��8O��52Te�����ߵĹ����еõ���
��2����ͬһ������Ԫ�ص���Ҫ���ϼ���ͬ���ڵ���Ҫ���ϼۿ����У�2��+4��+6��
��3����ͬһ�����У�Ԫ��ԭ������Խ��Ԫ�صķǽ�����Խ�����γɵ��⻯����ȶ���Խ��⻯��ˮ��Һ������Խǿ�����������ڵ��⻯��ˮ��Һ��������ǿ������˳����H2Te��H2Se��H2S��
��4����ͬһ�����У��γɵļ������ӵĻ�ԭ����ԭ�������������ǿ�����������н�ǿ�Ļ�ԭ�ԡ�
��5���ڴ˷�Ӧ�����зų�������Խ���⻯����ȶ���Խǿ���õ�a���⻯�����ȶ���d���⻯���ȶ������ɵá�
���㣺����������Ԫ��Ϊ���壬������Ԫ�������ɵ�Ӧ�á�

 ͬ��������ϰϵ�д�
ͬ��������ϰϵ�д� �ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д�
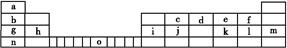
�ο�ͨ�γ̱�˼ά����������ѵ��ϵ�д���10�֣�Ԫ�����ڱ���Ԫ����������ѧϰ���о�������ʵ�����к���Ҫ�����á��±��г��ˢ١������Ԫ�������ڱ��е�λ�á�
| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | | | | �� | | �� | | |
| 3 | �� | �� | �� | | | | �� | �� |
| 4 | �� | | | | | | �� | |
(2)�ڢ١��ڡ�������Ԫ�ص��������Ӧ��ˮ�����У�������ǿ���� (�ѧʽ)��
(3)�١��ڡ�������Ԫ�ذ����Ӱ뾶�ɴ�С��˳������Ϊ (�����ӷ���)��
(4)��Ԫ���γɵľ���ǿ�����Ե��⻯�����ʽ�� �� ��Ԫ����һ���⻯���ڳ�������ڷ�����Ӧ�Ļ�ѧ����ʽΪ ��
����ѧ����ѡ��3�����ʽṹ�����ʡ�(15��)
±��Ԫ�صĵ��ʺͻ�����ܶ࣬���ǿ���������ѧ���ʽṹ�����ʵ����֪ʶȥ��ʶ���������ǡ�
��1��±��Ԫ��λ��Ԫ�����ڱ���_________������ļ۵����Ų�ʽΪ____________________��
��2����һ��Ũ�ȵ���Һ�У���������Զ����ӵ�(HF)2��ʽ���ڵġ�ʹ�������ӵϵ���������________��
��3��������±��ṩ�ĵ�һ�����������жϣ����п������ɽ��ȶ��ĵ��������ӵ�±��ԭ����_________��
| | �� | �� | �� | �� |
| ��һ������ (kJ/mol) | 1681 | 1251 | 1140 | 1008 |
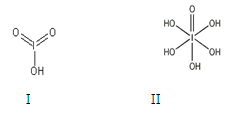
��Ƚ϶�������ǿ����HIO3_____ H5IO6(������� ����������)��
��5����֪ClO2�� Ϊ���ͣ�������ԭ����Χ���ĶԼ۲���ӡ�ClO2�� ������ԭ�ӵ��ӻ��������Ϊ___________��д��һ��ClO2�� �ĵȵ�����__________��
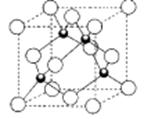
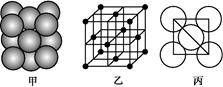
��6����ͼΪ�⾧�徧���ṹ���й�˵������ȷ����_____________��
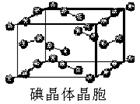
A������ӵ�������2�ֲ�ͬ��ȡ��2��ȡ��ͬ�ĵ������4��λ��������λ�γɲ�ṹ
B���þ�̯����֪ƽ��ÿ����������4����ԭ��
C���⾧��Ϊ��������Ŀռ�ṹ����ԭ�Ӿ���
D���⾧���д��ڵ�������зǼ��Լ��ͷ��»���
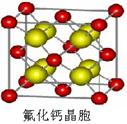
��7����֪CaF2����(��ͼ)���ܶ�Ϊ��g/cm3��NAΪ�����ӵ��������������ڵ�����Ca2���ĺ˼��Ϊa cm����CaF2����Է����������Ա�ʾΪ___________��
[��ѧ��ѡ��3���ʽṹ������](15��)
±��Ԫ�صĵ��ʺͻ�����ܶ࣬���ǿ���������ѧ���ʽṹ�����ʵ����֪ʶȥ��ʶ���������ǡ�
��1��±��Ԫ��λ�����ڱ���_________������ļ۵����Ų�ʽΪ____________________��
��2���ڲ�̫ϡ����Һ�У���������Զ����ӵ�(HF)2��ʽ���ڵġ�ʹ�������ӵϵ���������________��
��3��������±��ṩ�ĵ�һ�����������жϣ����п������ɽ��ȶ��ĵ��������ӵ�±��ԭ����_________��
| | �� | �� | �� | �� | �� |
| ��һ������ ��kJ/mol�� | 1681 | 1251 | 1140 | 1008 | 900 |
 ����HIO4��ǰ��Ϊ��Ԫ�ᣬ����ΪһԪ�ᡣ��Ƚ϶�������ǿ��:H5IO6_____HIO4����������� ������������
����HIO4��ǰ��Ϊ��Ԫ�ᣬ����ΪһԪ�ᡣ��Ƚ϶�������ǿ��:H5IO6_____HIO4����������� ��������������5������ˮ�е��ܽ����ȻС�����ڵ⻯����Һ���ܽ��ȴ������������������Һ�з������з�ӦI-+I2=I3-��I3-���ӵ�����ԭ����Χ�Ҽ����ӶԶ���Ϊ_____���µ��ӶԶ���Ϊ______�� I3-���ӵĿռ乹��Ϊ___________��
��KI3���Ƶģ�����CsICl2������֪CsICl2���ȶ��������ֽ⣬���������ɾ����ܸ�������ʣ�����������_____ʽ������ A��CsICl2=CsCl+ICl B��CsICl2=CsI+Cl2
��6����֪ClO2-Ϊ���ͣ�������ԭ����Χ���ĶԼ۲���ӡ�ClO2-������ԭ�ӵ��ӻ��������Ϊ___________��д��һ��ClO2-�ĵȵ�����__________��
��7����ͼΪ�⾧�徧���ṹ���й�˵������ȷ����_____________��

A������ӵ�������2�ֲ�ͬ��ȡ��2��ȡ��ͬ
�ĵ������4��λ��������λ�γɲ�ṹ
B���þ�̯����֪ƽ��ÿ����������4����ԭ��
C���⾧��Ϊ��������Ŀռ�ṹ����ԭ�Ӿ���
D���⾧���еĵ�ԭ�Ӽ���ڷǼ��Լ��ͷ��»���
��8����֪CaF2���壨����ͼ�����ܶ�Ϊ��g/cm3��NAΪ�����ӵ����������ڵ�����Ca2+�ĺ˼��Ϊa cm����CaF2����Է����������Ա�ʾΪ___________��

 [Cu(NH3)3CO]Ac
[Cu(NH3)3CO]Ac ������ĿΪ ��
������ĿΪ ��