��Ŀ����
����Ŀ��������������NOx���Ǵ�����Ⱦ��֮һ����ҵ����һ���¶Ⱥʹ�����������NH3��NOx��ԭ����N2��ijͬѧ��ʵ�����ж�NH3��NOx��Ӧ������̽�����ش��������⣺
��1���������Ʊ�

�������ķ���װ�ÿ���ѡ����ͼ�е�_________����Ӧ�Ļ�ѧ����ʽΪ_______________��
��Ԥ�ռ�һƿ����İ�����ѡ����ͼ�е�װ�ã�������˳��Ϊ������װ����______��������������Сд��ĸ��ʾ����
��2����������������ķ�Ӧ
�������ռ�����NH3����ע����X�У�Ӳ�ʲ�����Y�м�����������������NO2�������ü���K1��K2�к�������һ���¶��°�ͼʾװ�ý���ʵ�顣

�������� | ʵ������ | ����ԭ�� |
��K1���ƶ�ע����������ʹX�е����建��ͨ��Y���� | ��Y����_____________ | ����Ӧ�Ļ�ѧ����ʽ ____________ |
��ע���������˻�ԭ�����̶�����װ�ûָ������� | Y����������ˮ�� | ���ɵ���̬ˮ���� |
��K2 | ��_______________ | ��______________ |
���𰸡���1���� A�� 2NH4Cl+Ca��OH��2![]() CaCl2+2NH3��+2H2O����B��NH3��H2O
CaCl2+2NH3��+2H2O����B��NH3��H2O![]() NH3��+H2O����
NH3��+H2O����
��d��c��f��e��i��
��2��������ɫ������ɫ������dz����8NH3+6NO2 ![]() 7N2 +12H2O��
7N2 +12H2O��
��Z��NaOH��Һ����������������Ӧ��������������٣�Y����ѹǿС��������ѹ��
��������
�����������1������ʵ�����У���������������NH4Cl��ϼ��ȷ������ֽⷴӦ��ȡ����������������NH4Cl���ǹ��壬�������������ȡ���壬��Ӧѡ��Aװ�ý��У���ȡ������������Ӧ�Ļ�ѧ����ʽΪ2NH4Cl+Ca��OH��2![]() CaCl2+2NH3��+2H2O��Ҳ��������NH3��H2O�IJ��ȶ��ԣ����������ȷֽ����������ˮ��ȡ��������ʱӦ��ѡ���װ����װ��B����Ӧ�Ļ�ѧ����ʽ�ǣ�NH3��H2O
CaCl2+2NH3��+2H2O��Ҳ��������NH3��H2O�IJ��ȶ��ԣ����������ȷֽ����������ˮ��ȡ��������ʱӦ��ѡ���װ����װ��B����Ӧ�Ļ�ѧ����ʽ�ǣ�NH3��H2O![]() NH3��+H2O��������A����B��װ���Ƶõİ��������ڰ����Ǽ������壬������Ҫ���ü��Ը������ʯ�ҽ��и��Ȼ���ٸ��ݰ������ܶȱȿ���С�����ʣ��������ſ������ռ��������Ǵ�����Ⱦ�Ҫ����β������������������ˮ�м������ܽ�����ʣ���ˮ�����ռ�����β����������װ�õ�����˳��Ϊd��c��f��e��i����2����NO2����ǿ�����ԣ�NH3��ǿ��ԭ�ԣ������������ᷢ��������ԭ��Ӧ����������ˮ�����ݵ����غ㡢ԭ���غ㣬�ɵ÷�Ӧ�ķ���ʽ��8NH3+6NO2
NH3��+H2O��������A����B��װ���Ƶõİ��������ڰ����Ǽ������壬������Ҫ���ü��Ը������ʯ�ҽ��и��Ȼ���ٸ��ݰ������ܶȱȿ���С�����ʣ��������ſ������ռ��������Ǵ�����Ⱦ�Ҫ����β������������������ˮ�м������ܽ�����ʣ���ˮ�����ռ�����β����������װ�õ�����˳��Ϊd��c��f��e��i����2����NO2����ǿ�����ԣ�NH3��ǿ��ԭ�ԣ������������ᷢ��������ԭ��Ӧ����������ˮ�����ݵ����غ㡢ԭ���غ㣬�ɵ÷�Ӧ�ķ���ʽ��8NH3+6NO2![]() 7N2 +12H2O�����ݷ�Ӧ����ʽ��֪��Ӧ�����ǣ�Y���ڿ�������ɫ������ɫ��dz����Ϊ��ɫ��ͬʱ��ע�������ڱ���ˮ���������������������֪�����÷�Ӧ�ķ���ʽ��8NH3+6NO2
7N2 +12H2O�����ݷ�Ӧ����ʽ��֪��Ӧ�����ǣ�Y���ڿ�������ɫ������ɫ��dz����Ϊ��ɫ��ͬʱ��ע�������ڱ���ˮ���������������������֪�����÷�Ӧ�ķ���ʽ��8NH3+6NO2 ![]() 7N2 +12H2O������Ӧ��������������������ʵ������٣����Ի�ʹ����������ѹǿ��С������K2���ձ���NaOH��Һ�ڴ���ѹǿ�������»ᵹ������Y���ڣ�����Һ����Y���ڵ�ԭ���������÷�Ӧ�����������С�ķ�Ӧ����Ӧ��������װ����ѹǿ���ͣ���������ѹ�������¶�������������
7N2 +12H2O������Ӧ��������������������ʵ������٣����Ի�ʹ����������ѹǿ��С������K2���ձ���NaOH��Һ�ڴ���ѹǿ�������»ᵹ������Y���ڣ�����Һ����Y���ڵ�ԭ���������÷�Ӧ�����������С�ķ�Ӧ����Ӧ��������װ����ѹǿ���ͣ���������ѹ�������¶�������������

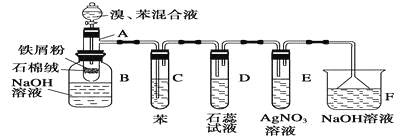
����Ŀ����ͼ����Ϊ�����Ƶá����Ӳ����������ʵ�װ�ã����ȼ��г�����ʡ�ԣ�������������ܴﵽĿ�ĵ����� ��

ѡ�� | ���� | װ����ҩƷ | ||
a | b | c | ||
A | C2H2 | ����ʳ��ˮ+��ʯ | CuS04��Һ | ��ˮ |
B | CO2 | ϡ����+ʯ��ʯ | ����NaHC03��Һ | �������Һ |
C | C2H4 | ������+ NaOH�Ҵ���Һ | ˮ | K2z Cr207,������Һ |
D | SO2 | ̼+Ũ���� | ������ˮ | Ʒ����Һ |
����Ŀ����1���״�����Ҫ�Ļ���ԭ�ϣ��ֿ���ȼ�ϡ����úϳ�������Ҫ�ɷ�ΪCO��CO2��H2���ڴ����������ºϳɼ״�����Ҫ��Ӧ���£�
��CO��g��+2H2��g��![]() CH3OH��g�� ��H1
CH3OH��g�� ��H1
��CO2��g��+3H2��g��![]() CH3OH��g��+H2O��g�� ��H2= ��58 kJ/mol
CH3OH��g��+H2O��g�� ��H2= ��58 kJ/mol
��CO2��g��+H2��g��![]() CO��g��+H2O��g�� ��H3
CO��g��+H2O��g�� ��H3
��֪��Ӧ���е���صĻ�ѧ�������������£�
��ѧ�� | H��H | C��O | C ��CO�еĻ�ѧ���� | H��O | C��H |
E/��kJ/mol�� | 436 | 343 | 1076 | 465 | 413 |
�ش��������⣺
����H3=_____kJ/mol
��25����101 kPa�����£����16g�״���ȫȼ���ͷų�Q kJ����������д����ʾ�״�ȼ���ȵ��Ȼ�ѧ����ʽ_______________
��2��25������a mol��L��1��ˮ��b mol��L��1����������Ϻ���Һ�����ԣ����ʱ��Һ��c��NH4+��__________c��Cl����������������������������ú�a��b�Ĵ���ʽ��ʾ���¶���NH3��H2O�ĵ���ƽ�ⳣ��Kb = _________
��3��800��ʱ����2L�ܱ������з�����Ӧ2NO��g����O2��g��![]() 2NO2��g�����ڷ�Ӧ��ϵ�У�n��NO����ʱ��ı仯���±���ʾ��
2NO2��g�����ڷ�Ӧ��ϵ�У�n��NO����ʱ��ı仯���±���ʾ��
ʱ����s�� | 0 | 1 | 2 | 3 | 4 | 5 |
n��NO����mol�� | 0.020 | 0.010 | 0.008 | 0.007 | 0.007 | 0.007 |
����ͼ�б�ʾNO2�仯��������___________��O2��ʾ��0��2s�ڸ÷�Ӧ��ƽ������v��____

����˵���÷�Ӧ�Ѿ��ﵽƽ��״̬����_______
a��v��NO2��=2v��O2��
b��������ѹǿ���ֲ���
c��v����NO����2v����O2��
d�������ڵ��ܶȱ��ֲ���