��Ŀ����
��13�֣�
�������ڹ���Ԫ��Fe��Ti����C��H��N��O�γɶ��ֻ����
��1����H��C��N��O����Ԫ�صĵ縺����С�����˳��Ϊ ��
��������������ȷ���� ��������ĸ��
A����ΪHCHO��ˮ���Ӽ����γ����������CH2O������ˮ
B��HCHO ��CO2�����е�����ԭ�Ӿ�����sp2�ӻ�
C��C6H6�������6��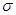 ����1����
����1����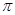 ����C2H2�ǷǼ��Է���
����C2H2�ǷǼ��Է���
D��CO2������۵㡢�е㶼�ȶ������辧��ĵ�
�����ᣨHOCN����һ����״���ӣ����������ᣨHNCO����Ϊͬ���칹�壬������ڸ�ԭ���������Ѵﵽ�ȶ��ṹ����д������Ľṹʽ ��
(2) Feԭ�ӻ�������Χ�н϶���������Ŀչ������һЩ���ӻ������γ�����
����Feԭ�ӻ������γ������ķ��ӻ�����Ӧ�߱��Ľṹ������
���������������[Fe(CN)6]4-�����__________��
a�����ۼ� b���Ǽ��Լ� c����λ�� d���Ҽ� e���м�
��д��һ���� CN_ ��Ϊ�ȵ�����ĵ��ʷ���ʽ______________________��
�����Ȼ���������Ϊ���壬�۵� ���е�
���е� ����
����
 ������������������ˮ��Ҳ���������ѡ���ͪ���л��ܼ����ݴ��ж����Ȼ����ľ�������Ϊ___________��
������������������ˮ��Ҳ���������ѡ���ͪ���л��ܼ����ݴ��ж����Ȼ����ľ�������Ϊ___________��
��3����Ti�������ڱ���λ��______________ ��
����Ԫ��ԭ�ӵ�������Ų����������ɽ�Ԫ�����ڱ��ֳ������������Ti���� ����
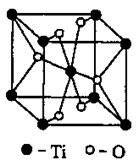
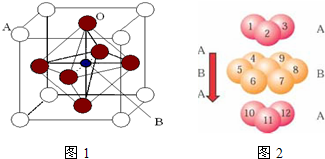
��Ti��һ��������X���侧���ṹ����ͼ��ʾ����X�Ļ�ѧʽΪ ����ҵ������X��̼�ᱵ������״̬����ȡ������M��M�ɿ���һ�ֺ������Σ�,�Ʊ�M�Ļ�ѧ��Ӧ����ʽ��________________________________________��
��13�֣�
��1����H<C<N<O��1�֣� ��B��1�֣� ��N��C��O��H��1�֣�
��2���پ��й¶Ե��ӣ�1�֣� ��b ��1�֣� N2 ��1�֣� �۷��Ӿ��壨1�֣�
��3���ٵ������� �ڢ�B�壨1�֣� d����1�֣�
��TiO2��2�֣� TiO2+BaCO3==BaTiO3+CO2����2�֣�
��������

 ��У����ϵ�д�
��У����ϵ�д�