��Ŀ����
1��2010���Ϸ����籭�����һ�컷�������籭����ʱ��������������������������������û��տ�Ȫˮƿ�����Ƴɵ�һ�ָ��ᡢ�����ķ�װ����֪һ�ֿ������������ϣ����Ľṹ��ʽ���£�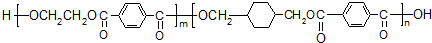
��֪��
��1��
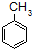 $\stackrel{KMnO_{4}/H+}{��}$
$\stackrel{KMnO_{4}/H+}{��}$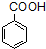
��2��RCOOR1+R2OH��RCOOR2+R1OH��R��R1��R2�������������ֻ������Ͽɲ������кϳ�·�߽��кϳɣ�
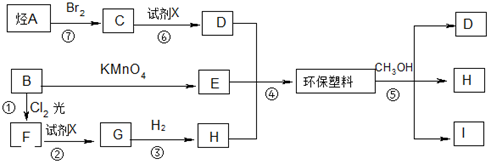
��ش��������⣺
��1��д����A�Ľṹʽ
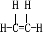 ��
����2����D��E��H�ϳɻ������ϵķ�Ӧ���������۷�Ӧ����Ӧ�ݵķ�Ӧ������ȡ����Ӧ��
��3��д��E�Ľṹ��ʽ
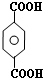 ��B�����ƣ��Զ��ױ����Լ�XΪNaOHˮ��Һ��
��B�����ƣ��Զ��ױ����Լ�XΪNaOHˮ��Һ����4��д����Ӧ�۵Ļ�ѧ����ʽ
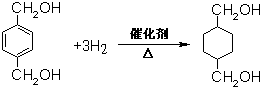 ��
����5��д����Ӧ�Ļ�ѧ����ʽCH2BrCH2Br+2NaOH$\stackrel{��}{��}$CH2OHCH2OH+2NaBr��
���� �ɻ������ϵĽṹ��ʽ��֪��Ӧ�ĵ��ʷֱ���CH2OHCH2OH�� �Լ�
�Լ�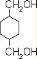 �����ݷ�Ӧ������֪DӦΪCH2OHCH2OH��EΪ
�����ݷ�Ӧ������֪DӦΪCH2OHCH2OH��EΪ ��HΪ
��HΪ ����AΪCH2=CH2��CΪCH2BrCH2Br��BΪ�Զ��ױ���FӦΪ
����AΪCH2=CH2��CΪCH2BrCH2Br��BΪ�Զ��ױ���FӦΪ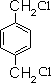 ��GΪ
��GΪ ��IӦΪ�Ա����������������л���Ľṹ�����ʿɽ����⣮
��IӦΪ�Ա����������������л���Ľṹ�����ʿɽ����⣮
��� �⣺�ɻ������ϵĽṹ��ʽ��֪��Ӧ�ĵ��ʷֱ���CH2OHCH2OH�� �Լ�
�Լ� �����ݷ�Ӧ������֪DӦΪCH2OHCH2OH��EΪ
�����ݷ�Ӧ������֪DӦΪCH2OHCH2OH��EΪ ��HΪ
��HΪ ����AΪCH2=CH2��CΪCH2BrCH2Br��BΪ�Զ��ױ���FӦΪ
����AΪCH2=CH2��CΪCH2BrCH2Br��BΪ�Զ��ױ���FӦΪ ��GΪ
��GΪ ��IӦΪ�Ա������������
��IӦΪ�Ա������������
��1�������Ϸ�����֪AΪCH2=CH2���ṹʽΪ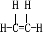 ��
��
�ʴ�Ϊ��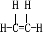 ��
��
��2����D��E��H�ϳɻ������ϵķ�Ӧ���������۷�Ӧ����Ӧ��Ϊȡ����Ӧ��
�ʴ�Ϊ�����۷�Ӧ��ȡ����Ӧ��
��3�������Ϸ�����֪EΪ ��BΪ�Զ��ױ���CΪCH2BrCH2Br����NaOHˮ��Һ��ˮ�������CH2OHCH2OH��
��BΪ�Զ��ױ���CΪCH2BrCH2Br����NaOHˮ��Һ��ˮ�������CH2OHCH2OH��
�ʴ�Ϊ�� �� �Զ��ױ���NaOHˮ��Һ��
�� �Զ��ױ���NaOHˮ��Һ��
��4��GΪ �����б������������������ӳɷ�Ӧ������ʽΪ
�����б������������������ӳɷ�Ӧ������ʽΪ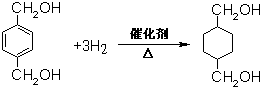 ��
��
�ʴ�Ϊ��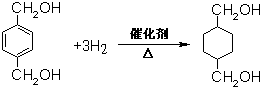 ��
��
��5����Ӧ��Ϊˮ�ⷴӦ����Ӧ�Ļ�ѧ����ʽΪCH2BrCH2Br+2NaOH$\stackrel{��}{��}$CH2OHCH2OH+2NaBr��
�ʴ�Ϊ��CH2BrCH2Br+2NaOH$\stackrel{��}{��}$CH2OHCH2OH+2NaBr��
���� ���⿼���л���ĺϳ����ƶϣ���Ŀ�Ѷ��еȣ�����ע����ݸ߾����ж϶�Ӧ�ĵ���Ϊ������Ĺؼ�������л�������ʺͷ�Ӧ�������ƶ��������ʣ�

 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�| A�� | B��C��D�γɵļ����ӣ��뾶��С����B | |
| B�� | E�γ�����⻯���ȶ��Ա�Bǿ | |
| C�� | A��C��D�γɵ�����������ˮ��������������Ӧ | |
| D�� | A��B�γɵĻ�������B��E�γɵĻ����ﻯѧ��������ͬ |
| A�� | ����50% | B�� | ����50% | C�� | С��50% | D�� | ��ȷ�� |
| A�� | ��Ȳ��ȼ����Ϊ1256 kJ/mol | |
| B�� | ��ת��10 mol���ӣ�������2.5 mol O2 | |
| C�� | ������2 mol Һ̬ˮ�����H=-1256 kJ/mol | |
| D�� | ���γ�4 mol̼�����õ��Ӷԣ���ų�������Ϊ1256 kJ |
| A | B |
| ����ʹ��������Ȼ�̼��Һ��ɫ�� �ڱ���ģ��Ϊ�� 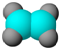 ������ˮ��һ�������·�Ӧ | ����C��H����Ԫ����ɣ� �����ģ��Ϊ�� 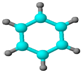 |
��1��A�뺬������Ȼ�̼��Һ��Ӧ������������ƽ���1��2-�������飻д����һ�������£�A���ɸ߷��ӻ�����Ļ�ѧ��Ӧ����ʽnCH2�TCH2$\stackrel{����}{��}$
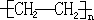 ��
����2��A�����������ӳɷ�Ӧ������C��C�ڷ�����ɺͽṹ�����Ƶ��л�����һ���ࣨ�׳ơ�ͬϵ��������Ǿ�����ͨʽCnH2n+2����n=4ʱ�������л��↑ʼ����ͬ���칹�壮
��3��B���е������Ǣ٢ڢۣ�����ţ���
����ɫ��ζҺ�塢���ж����۲�����ˮ�����ܶȱ�ˮ��������KMnO4��Һ����ˮ��Ӧ��ɫ�����κ������²���������Ӧ��
��4��д����Ũ���������£�B��Ũ���ᷴӦ�Ļ�ѧ����ʽ��
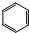 +HO-NO2$��_{50-60��}^{Ũ����}$
+HO-NO2$��_{50-60��}^{Ũ����}$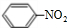 +H2O��
+H2O�� | A�� | ��ͭпԭ�����CuΪ���� | |
| B�� | ԭ����У���ѧ��ת��Ϊ���� | |
| C�� | ��ԭ����У������Ϸ���������Ӧ�������Ϸ�����ԭ��Ӧ | |
| D�� | ��ԭ����У����ӴӸ��������������Һ�������� |
| A�� | ���ӻ�������һ�����н������� | B�� | ���ۻ����ﲻ���ܺ����Ӽ� | ||
| C�� | ���ӻ�������ܺ����ۼ� | D�� | CaCl2�������Ӽ����������ۼ� |
| A�� | ���·������������ۻ�Ϊͬ���칹�� | |
| B�� | �þƾ����������������ò��������ʱ������� | |
| C�� | ��ըʳ��Ļ����ͺ�ţ�Ͷ��ǿ������ı������� | |
| D�� | ĥ�����Ĵ��������ʣ�������к��ʱ���˰����� |