��Ŀ����
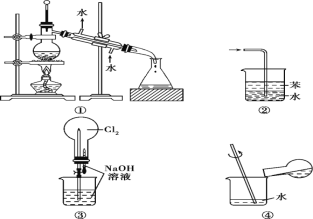
����Ŀ��ij��ѧ��ȤС������ͭ��Ũ���ᷴӦ��̽��SO2�IJ������ʣ�ʵ��װ����ͼ��ʾ��
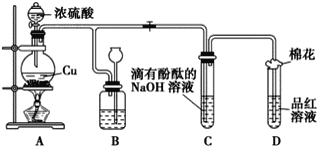
��1��װ��A�з����Ļ�ѧ��Ӧ�Ļ�ѧ����ʽΪ________________________________
��2��װ��B�������������������壬B�й��ƿ��Ӧʢ�ŵ�Һ����________(�����)��
��ˮ �ڱ��͵�NaHSO3��Һ ��NaOH��Һ ������KMnO4
��3��װ��C��D�в�����������ͬ����ԭ��ȴ��ͬ��C��������SO2����Һ�е�NaOH��Ӧʹ���Լ�������D����������SO2����____________��
��4��װ��D���Թܿ���һ����������Ӧ����һ��Һ�壬��������____________
��5����ʵ������һ��������ͭƬ��25.0 mL 18.0 mol��L��1��Ũ�����ڼ���������ַ�Ӧ������ͭƬ���������ʣ�ࡣ�ٶ���Ӧǰ����Һ��������䣬��ʣ�������Ũ��Ϊd mol��L��1������Ӧ����Һ�л�����������ع��壬��ֽ���ֱ��ʣ��ͭƬǡ����ȫ��Ӧ����Ӧ���������____________mol(�ú�d�Ĵ���ʽ��ʾ)��
���𰸡�Cu+2H2SO4(Ũ) ![]() CuSO4+SO2��+2H2O �� Ư��(��Ư����) ���ն����SO2��ֹ��Ⱦ����
CuSO4+SO2��+2H2O �� Ư��(��Ư����) ���ն����SO2��ֹ��Ⱦ���� ![]() (��0.0125d ��1.25��10-2d�� )
(��0.0125d ��1.25��10-2d�� )
��������
��1��װ��A��ͭ��Ũ���������������ͭ����������ˮ��
��2����������������ˮ���������������ڱ��͵�NaHSO3��Һ���۶���������NaOH��Һ��Ӧ�����������ƺ�ˮ�� �ܶ�����������KMnO4��Һ����Ϊ��������ӡ�
��3��SO2����Ư���ԣ���ʹƷ����Һ��ɫ��
��4�����������ж�������������Һ�����ն�������
��5����Ӧ����Һ�л�����������ع��壬������Ӧ3Cu+8H++2NO3-=3Cu2++2NO+4H2O��
��1��װ��A��ͭ��Ũ���������������ͭ����������ˮ����Ӧ�Ļ�ѧ����ʽΪCu+2H2SO4(Ũ) ![]() CuSO4+SO2��+2H2O
CuSO4+SO2��+2H2O
��2����������������ˮ���������������ڱ��͵�NaHSO3��Һ���۶���������NaOH��Һ��Ӧ�����������ƺ�ˮ�� �ܶ�����������KMnO4��Һ����Ϊ��������ӡ�װ��B�������������������壬B�й��ƿ��Ӧʢ�ŵ�Һ���DZ��͵�NaHSO3��Һ��ѡ�ڣ�
��3��SO2����Ư���ԣ���ʹƷ����Һ��ɫ��Ʒ����ɫ������SO2��Ư���ԣ�
��4��װ��D���Թܿ���һ����������Ӧ��������������Һ�������������ն����SO2��ֹ��Ⱦ������
��5���跴Ӧ���������xmol��
3Cu + 8H+ + 2NO3- = 3Cu2++2NO+4H2O��
8 2
d mol��L��1![]() 2
2![]() 0.025L x
0.025L x
![]()
x=0.0125dmol

 �п������п��Ծ����ϵ�д�
�п������п��Ծ����ϵ�д� ��������״Ԫ��ϵ�д�
��������״Ԫ��ϵ�д� �ƸԿ�����ҵ��ϵ�д�
�ƸԿ�����ҵ��ϵ�д� ��Ԫ����ĩ��ϰ�ȷ��ϵ�д�
��Ԫ����ĩ��ϰ�ȷ��ϵ�д�����Ŀ������г���A��R9��Ԫ�������ڱ��е�λ�ã��û�ѧ����ش�
���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A |
|
2 | E | F | ||||||
3 | A | C | D | G | R | |||
4 | B | H |
��1�������Ԫ���л�ѧ��������õ���___��
��2��DԪ�ص�����������Ӧ��ˮ�������������Ʒ�Ӧ�����ӷ���ʽ��__��
��3��A��B��C����Ԫ�ذ�ԭ�Ӱ뾶�ɴ�С��˳������Ϊ__��
��4��F��G��H����Ԫ�ؼ��⻯��е��ɸߵ��͵�˳��__��/p>
��5��EԪ��������ĵ���ʽ��__��F���⻯���ڳ����¸�B��Ӧ�Ļ�ѧ����ʽ��__��
��6��������ʵ����Ԫ�������ɽ��͵��ǣ�����ĸ��ţ�__��
A��D������������Ӧˮ����ļ�������Mg(OH)2
B��G����̬�⻯����ȶ�������HF
C��FeG3����Һ�����ڿ�ʴͭ�Ƶ�ӡˢ��·��
��7��G�ĵ��ʿ��Դ�NaBr��Һ���û���Br2���ʣ���ԭ�ӽṹ�ĽǶȽ�����ԭ��_____��