��Ŀ����
7��������Ⱦ�Ѿ���Ϊ�ҹ�������Ե���Ҫ������⣮�������л�����Ⱦ��ԭ�������������ȷ���ǣ�������| A�� | �������ɴ���ȼ��ú����Ȼ����ʯ�͵Ȳ�����CO2ƫ������ | |
| B�� | ��ɫ��ѧ�ĺ����ǿ���Ӧ���¼������Ӵ�Ի�����Ⱦ���������� | |
| C�� | �ô���������������β���е�CO��NO��CO+NO$\frac{\underline{\;����\;}}{\;}$C+NO2 | |
| D�� | ��ø����ϴ�·۵�ʹ�ã����Խ���ˮ����Ⱦ���纣��ijೱ�� |
���� A��PM2.5��������������ҪԪ�ף�
B����ɫ��ѧ�ĺ����Ǵ�Դͷ�ž�������Ⱦ��
C������������������β���е�CO��NO���ɶ�����̼�͵����������ʣ�����C+NO2��
D������ֲ���Ӫ��Ԫ�أ�
��� �⣺A���������ɴ���ȼ��ú����Ȼ����ʯ�͵Ȳ����ĸ��ֿ�����к��������£�������Ϊ����������̼����A����
B����ɫ��ѧ�ĺ����Ǵ�Դͷ�ž�������Ⱦ��������������Ⱦ����B����
C������������������β���е�CO��NO���ɶ�����̼�͵����������ʣ�����ʽΪ��2CO+2NO $\frac{\underline{\;����\;}}{\;}$2CO2+N2����C����
D��ˮ��ĸ�Ӫ�����ǵ��³ೱ����Ҫԭ��D��ȷ��
��ѡD��
���� ���⿼�黷����Ⱦ�������������ڻ�ѧ�����彡���Ŀ��飬Ϊ�߿��������ͺ�Ƶ���㣬����������ѧ�������ÿ�ѧ�������ѶȲ���ע����ػ���֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 ��ʦ�㲦��ϵ�д�
��ʦ�㲦��ϵ�д�
�����Ŀ
17������ȷ��ʾ���з�Ӧ�����ӷ���ʽ�ǣ�������
| A�� | ��AlCl3��Һ�м������ϡ��ˮ��Al3++4NH3•H2O=AlO2-+4NH4++2H2O | |
| B�� | ��CuSO4��Һ�м���Na2O2��2Na2O2+2Cu2++2H2O=4Na++2Cu��OH��2��+O2�� | |
| C�� | ��Ca��ClO��2��Һ��ͨ�����������̼���壺Ca2++2ClO-+CO2+H2O=CaCO3��+2HClO | |
| D�� | ��HF��ʴ������SiO2+4H++4F-=SiF4��+2H2O |
18�����е��뷽��ʽ����д����ȷ���ǣ�������
| A�� | NH3•H2O�TNH$\stackrel{+}{4}$+OH- | B�� | BaSO4�TBa2++SO$\stackrel{2-}{4}$ | ||
| C�� | HNO3�TH++NO$\overline{3}$ | D�� | NaOH�TNa++OH- |
19�������ǿ�ѧ�о�����Ҫ�������������ʷ����ȷ���ǣ�������
| A�� | ������ɱ�����ˮ�����ռС�մ� | |
| B�� | ����ʣ����ᣬ���飬�ռʳ�� | |
| C�� | ͬ�������壺ʯī��C60��C80�����ʯ | |
| D�� | ����������������Ȫˮ�����������ᣬʯ��ʯ |
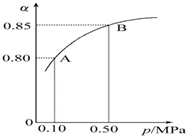 ���ȫ����ڵ������쳣�����������ٴγ�Ϊ���㣮�������Լ�����β���ŷŵĶ�������һ����̼���������NOx���������ѳ�Ϊ������Ⱦ����Ҫ���أ�
���ȫ����ڵ������쳣�����������ٴγ�Ϊ���㣮�������Լ�����β���ŷŵĶ�������һ����̼���������NOx���������ѳ�Ϊ������Ⱦ����Ҫ���أ�