��Ŀ����
15������˵������ȷ���м�������������ڿ�����ȼ�����ɵ���ɫ��Na2O2
������480mL0.5mol/L��NaOH��Һ����Ҫ��������ƽ�����������ƹ���9.6g
������������������л�ԭ��
��Fe��OH��3��FeCl2��H2SiO3����ֱ���û��Ϸ�Ӧ�Ʊ�
��SO2��ʹ���Ը��������Һ��ɫ������������Ư����
����������Ϊ5%��25%�İ�ˮ�������Ϻ�������Һ�����ʵ�������������15%
�߸����Cl2����ˮ����ʹ�ʻ���ɫ
���к��Ȳⶨʵ������Ҫ�õ��IJ�������ֻ���ձ����¶ȼƺͻ��β������������
�ᴿ�����ڿ����о��ñ������Ϊ�����˻�ѧ��ʴ��
| A�� | 3�� | B�� | 4�� | C�� | 5�� | D�� | 6�� |
���� �����ڿ�����ȼ�����ɹ������ƣ�
��û��480mL����ƿ��ѡ�������Դ�����ӽ�������ƿ����Ӧѡ��500mL����ƿ�������������Ƶ�����Ϊ0.5L��0.5mol•L-1��40g/mol=10.0g��
�������Ȼ�������Ϊ+1�ۣ���Ϊ-1�۽��
��������������������ˮ�������������������Ȼ����������������Ȼ�������
�ݶ������������Եĸ�����ط���������ԭ��Ӧʹ����ɫ��
����������Ϊ5%��25%�İ�ˮ��������Ϻ�������Һ�����ʵ�������������15%�����ڵ�����ʱ25%�İ�ˮ��������ټ���һЩ5%�İ�ˮ�����ǵ���������Խ���������Ϊ5%��25%�İ�ˮ�������ϣ����ʵ���������С��15%��
�ߴ��������Ư���ԣ�����������Ư���ԣ�
�������к����������װ�ý��
�ᴿ������Ҫ������ѧ��ʴ��
��� �⣺�����ڿ�����ȼ�����ɹ������ƣ���������Ϊ����ɫ������ȷ��
��û��480mL����ƿ��ѡ�������Դ�����ӽ�������ƿ����Ӧѡ��500mL����ƿ�������������Ƶ�����Ϊ0.5L��0.5mol•L-1��40g/mol=10.0g����ȡ9.6g���ʣ�������ҺŨ��ƫ�ͣ��ʴ���
���Ȼ�������Ϊ+1�ۣ���Ϊ-1�����ϼۼȿ��������ֿ��Խ��ͣ����Լ������������л�ԭ�ԣ�����ȷ��
��������������������ˮ�������������������Ȼ����������������Ȼ�����������Fe��OH��3��FeCl2����ֱ���û��Ϸ�Ӧ�Ʊ����ʴ���
��SO2��ʹ���Ը��������Һ��ɫ�����������л�ԭ�ԣ��ʴ���
����������Ϊ5%��25%�İ�ˮ�������Ϻ�������Һ�����ʵ���������С��15%���ʴ���
��HClO����ǿ��������Ư�ף������Cl2�����ʻ�������HClO����ˮ����HClO�����Ը����Cl2����ˮ����ʹ�ʻ���ɫ������ȷ��
���к��Ȳⶨʵ������Ҫ�õ��IJ����������ձ����¶ȼƺͻ��β���������Ͳ����������֣��ʴ���
�ᴿ������Ҫ������ѧ��ʴ������ȷ��
��ѡ��B��
���� ����Ϊ�ۺ��⣬�漰Ԫ�ػ�����֪ʶ���к��ȵIJⶨ�������ĸ�ʴ���������Һ�����ƣ���ȷ���ʵ����ʼ����ʵ���ԭ���Ͳ����ǽ���ؼ�����Ŀ�ѶȲ���

��֪������������������������ʽ����ʱ��Һ��pH������
| ������ | Fe��OH��3 | Fe��OH��2 | Al��OH��3 |
| ��ʼ���� | 2.3 | 7.5 | 3.4 |
| ��ȫ���� | 3.2 | 9.7 | 4.4 |
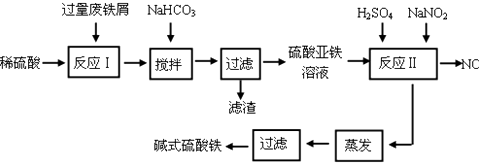
��1����μӿ����м���ܽ⣬д�����ְ취���衢���ȣ�
��2����������NaHCO3��Ŀ���ǵ���pH��4.4-7.5��Χ�ڣ�
��3����Ӧ���м���NaNO2��Ŀ���ǽ�������������Ϊ�����ӣ�
��4����ʽ����������ˮ�����ɵ�Fe��OH��2+���ӿɲ���ˮ������Fe2��OH��42+�ۺ����ӣ���ˮ�ⷴӦ�����ӷ���ʽΪ2Fe��OH��2++2H2O?Fe2��OH��42++2H+��
��5����ҽҩ�ϳ����������������ᡢ����Ļ��Һ��Ӧ�Ʊ���ʽ�������������ҹ�����������Ʒ�в��ú���Fe2+��NO3-��Ϊ�������ò�Ʒ���Ƿ���Fe2+��Ӧʹ�õ��Լ�ΪC��
A����ˮ B��KSCN��Һ C������KMnO4��Һ D��NaOH��Һ
��6��Ϊ�ⶨ��Fe2+��Fe3+��Һ����Ԫ�ص��ܺ�����ʵ��������£�ȷ��ȡ20.00mL��Һ�ڴ�����ƿ�У���������H2O2������pH��2�����ȳ�ȥ����H2O2���������KI��ַ�Ӧ������ 0.1000mol/L Na2S2O3����Һ�ζ����յ㣬���ı���Һ20.00mL��
��֪��2Fe3++2I-�T2Fe2++I2 I2+2S2O32-�T2I-+S4O62-
����Һ����Ԫ�ص��ܺ���Ϊg/L�����ζ�ǰ��Һ��H2O2û�г��������ⶨ����Ԫ�صĺ�������ƫ�� ���ƫ�ߡ���ƫ�͡������䡱����
| A�� | 0.3mol | B�� | ��0.3mol | ||
| C�� | ����0.3mol����0.6mol | D�� | ����0.6mol |
| A�� | 1��3 | B�� | 4��3 | C�� | 2��1 | D�� | 3��1 |
| A�� | ��������CuSO4���� | B�� | ��������CH3COONa���� | ||
| C�� | ��������NH4HSO4���� | D�� | ��������Na2CO3���� |
| A�� | �������ɴ���ȼ��ú����Ȼ����ʯ�͵Ȳ�����CO2ƫ������ | |
| B�� | ��ɫ��ѧ�ĺ����ǿ���Ӧ���¼������Ӵ�Ի�����Ⱦ���������� | |
| C�� | �ô���������������β���е�CO��NO��CO+NO$\frac{\underline{\;����\;}}{\;}$C+NO2 | |
| D�� | ��ø����ϴ�·۵�ʹ�ã����Խ���ˮ����Ⱦ���纣��ijೱ�� |
| A�� | ̼��Ķ������뷽��ʽ��H2O+HCO3-?H3O++CO32- | |
| B�� | ��Ļ�ѧ�ɷֶ�Ϊ��ˮ��þ�����Σ��磺Ca2Mg5Si8O22��OH��2�����������ʾΪ2CaO•5MgO•8SiO2•H2O | |
| C�� | NaH����ˮ��Ӧ�ķ���ʽ��NaH+D2O�TNaOD+HD�� | |
| D�� | ������NA�����ӵ�H2O��CH4������ͬ����� |
| A�� | �ñ���ȡ��ˮ�е���ʱ��Ӧ����ı���Һ�ӷ�Һ©���¿ڷų� | |
| B�� | ������ƿ������Һ������ʱ���ӿ̶��ߣ�������ҺŨ��ƫС | |
| C�� | ����ˮ��pH�����ò�����պȡ��ˮ����pH��ֽ�ϣ������ɫ�������ɫ���Ƚ� | |
| D�� | ��������ʱ��������������ʯ�����ϣ����þƾ��Ƶ�������� |
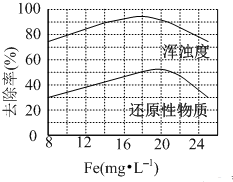 FeCl3 ���о�ˮ���ã�����ʴ�豸�����ۺ��Ȼ�����һ�����͵���������������ˮ��FeCl3 ��Ч���Ҹ�ʴ��С����ش��������⣺
FeCl3 ���о�ˮ���ã�����ʴ�豸�����ۺ��Ȼ�����һ�����͵���������������ˮ��FeCl3 ��Ч���Ҹ�ʴ��С����ش��������⣺