��Ŀ����
5���Ϻ�ijС���ϣ������õĵ�ˮ��Դ�൱�ѷ�����ž�սʿΪ��Ѱ�Һ��ʵ�����ˮԴ���Ե���ɽȪˮ���з������飬�����ʾɽȪˮ����Ӳˮ����1��Ӳˮ��ָ���н϶���Ρ�þ�ε�ˮ��Ӳˮ���Ⱥ�������������ӷ���ʽΪ��Ca2++2HCO3-$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+CO2��+H2O�� д������һ�ֳ�����ļ��ɣ���
��2��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mg CaO����֮�൱�����ʣ���7.1mg MgO������֪ˮ��Ӳ����8�����µ���ˮ����8�����ϵij�ΪӲˮ����֪����ɽȪˮ��c�� Ca2+��=1.2��10-3mol/L��c��Mg2+��=6��l0-4 mol/L����ô��ˮ�ǣ���ǡ�������Ӳˮ��
��3�����ӽ�����������ˮ�ij��÷������۱�ϩ������һ�����ӽ�����֬��д���۱�ϩ���Ƶ���Ľṹ��ʽCH2=CHCOONa��
��4����ž�սʿͨ����ˮ�м���������ˮ���������ӷ���ʽ�����侻ˮԭ����Al3++3H2O?Al��OH��3�����壩+3H+��
��5�����ϻ������ú�ˮ��������õ�ˮ�������Ǻ�ˮ���õ���������õ�ˮ��ԭ��ͼ����֪��ˮ�к�Na+��Cl-��Ca2+��Mg2+��SO42-�����ӣ��缫Ϊ���Ե缫��������������⣺
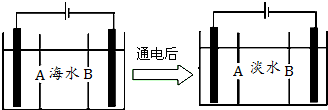
�������ӽ���Ĥ��ָB����A��B����
��д��ͨ����������ĵ缫��Ӧʽ2Cl--2e-�TCl2�����������������ǣ��缫�ϲ������ݣ���Һ�г���������ɫ������
���� ��1��Ӳˮ�Ǻ��н϶�����Ը��Ρ�þ�ε�ˮ��Ӳˮ�е�̼��������ȷֽ�����̼��Ƴ�����
��2������Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mgCaO����֮�൱�����ʣ���7.1mgMgO������ˮ�е�Ca2+��Mg2+���������CaO����������õ���
��3���۱�ϩ���Ƶĵ���Ϊ��ϩ�ƣ�
��4��������ˮ���������������������ӣ��������������������ܹ��������ʿ���������������ˮ��
��5���������ӽ���Ĥֻ��������������ͨ���������ӽ���Ĥֻ��������������ͨ����
�ڸ��������������ӷŵ磬�������������ӵõ�������������������������Ũ������
��� �⣺��1��Ӳˮ�Ǻ��н϶�����Ը��Ρ�þ�ε�ˮ��Ӳˮ�е�̼��������ȷֽ�����̼��Ƴ���������ʽΪ��Ca2++2HCO3-$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+CO2��+H2O��
�ʴ�Ϊ�����Ρ�þ�Σ�Ca2++2HCO3-$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+CO2��+H2O��
��2��ij��Ȼˮ��c��Ca2+��=1.2��10-3mol•L-1��c��Mg2+��=6��10-4mol•L-1��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mgCaO����֮�൱�����ʣ���7.1mgMgO����1Lˮ�и��������ʵ���=1.2��10-3mol���൱��CaO����=1.2��10-3mol��56g/mol=67.2mg��1Lˮ��þ�������ʵ���=6��10-4mol���൱������þ����6��10-4mol��40g/mol=24mg��ˮ��Ӳ��=$\frac{67.2mg}{10mg}$+$\frac{24mg}{7.1mg}$=10�㣬����Ӳˮ��
�ʴ�Ϊ���ǣ�
��3���۱�ϩ���Ƶĵ���Ϊ��ϩ�ƣ��ṹ��ʽ��CH2=CHCOONa���ʴ�Ϊ��CH2=CHCOONa��
��4��������ˮ���������������������ӣ����ӷ���ʽ��Al3++3H2O?Al��OH��3�����壩+3H+��
�ʴ�Ϊ��Al3++3H2O?Al��OH��3�����壩+3H+��
��5���������ӽ���Ĥֻ��������������ͨ���������ӽ���Ĥֻ��������������ͨ������ĤB�����������������������ӷŵ磬���Ը�ĤB�������ӽ���Ĥ��
�ʴ�Ϊ��B��
�ڸ��������������ӷŵ磺2Cl--2e-�TCl2�����������������ӵõ�������������������������Ũ���������ӣ�þ�����γɳ�����
�ʴ�Ϊ��2Cl--2e-�TCl2�����缫�ϲ������ݣ���Һ�г���������ɫ������
���� ���⿼���˻�ѧ������漰Ӳˮ����������������ˮ�ȣ���ȷ��ظ�����ԭ���ǽ���ؼ�����Ŀ�ѶȲ���

 ��ͼ����ʾ��Ӧ��mA��g��+nB��g��?pC��g��+qD��g������H���ڲ�ͬ�¶��¾���һ��ʱ����������ϵ��C�İٷֺ������¶�T�Ĺ�ϵ��ͼ����ʾ��һ�������´ﵽƽ���tʱ�̸ı�ѹǿ�����½���ƽ��ķ�Ӧ���̣��ɴ˿��жϸ÷�Ӧ�У�������
��ͼ����ʾ��Ӧ��mA��g��+nB��g��?pC��g��+qD��g������H���ڲ�ͬ�¶��¾���һ��ʱ����������ϵ��C�İٷֺ������¶�T�Ĺ�ϵ��ͼ����ʾ��һ�������´ﵽƽ���tʱ�̸ı�ѹǿ�����½���ƽ��ķ�Ӧ���̣��ɴ˿��жϸ÷�Ӧ�У�������| A�� | m+n��p+q����H��0 | B�� | m+n��p+q����H��0 | C�� | m+n��p+q����H��0 | D�� | m+n��p+q����H��0 |
| A�� |  ��0.01molKOH ��0.01mol Ca��OH��2 �Ļ����Һ�л���ͨ��CO2 | |
| B�� | 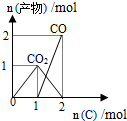 n��O2��=1molʱ��������C��O2���ܱ������еķ�Ӧ���� | |
| C�� | 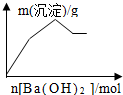 KAl��SO4��2��Һ����μ���Ba��OH��2 ��Һ | |
| D�� | 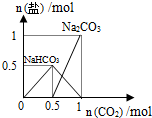 n��NaOH��=1molʱ��CO2��NaOH��Һ��Ӧ���ɵ��� |
| A�� | NO | B�� | CO | C�� | ��ȩ | D�� | CO2 |
| A�� | ������ | B�� | ̼������ | C�� | ������� | D�� | �Ȼ��� |
 ��һ���������ܱ������г���A2��B2��������ӦA2��g��+B2��g��?2C��g����H��0�����ﵽƽ���t0ʱ�����ֻ�����������ʵ���������ı�ijһ��Ӧ������ʹ������ѹǿ��������˵����ȷ���ǣ�������
��һ���������ܱ������г���A2��B2��������ӦA2��g��+B2��g��?2C��g����H��0�����ﵽƽ���t0ʱ�����ֻ�����������ʵ���������ı�ijһ��Ӧ������ʹ������ѹǿ��������˵����ȷ���ǣ�������| A�� | ƽ�����淴Ӧ�����ƶ���ƽ����Է����������� | |
| B�� | ƽ��������Ӧ�����ƶ�����������ܶ����� | |
| C�� | A2ת��������Cƽ��Ũ�ȱ�� | |
| D�� | t0ʱ������ϵ�¶� |
| A�� | ԭ��������ͬ | B�� | ���ʵ�����ͬ | C�� | ����������ͬ | D�� | ������ͬ |
