��Ŀ����
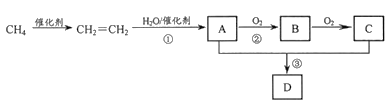
����Ŀ�����������仯�����ںϽ���ϼ������ȷ���Ӧ�ù㷺��ij��������Ҫ�ɷ���NiFe2O4(������)��NiO��FeO��CaO��SiO2�ȣ������ǴӸÿ����л���NiSO4�Ĺ������̣�

��֪��(NH4)2SO4��350�����ϻ�ֽ�����NH3��H2SO4��NiFe2O4�ڱ��չ���������NiSO4��Fe2(SO4)3��
(1)����ǰ��������(NH4)2SO4�����ĥ�������ĥ��Ŀ����______________________��
(2)�����ݡ�������Fe2(SO4)3����FeO(OH)�����ӷ���ʽΪ___________________�����������ijɷֳ�Fe2O3��FeO(OH)�����______________(�ѧʽ)��
(3)Ϊ��֤��Ʒ���ȣ�Ҫ��⡰����Һ������������ȡһ������Ľ���Һ���������ữ����SnC12��Fe3+��ԭΪFe2+������SnC12�����ʵ���������Fe3+���ʵ�����_____������ȥ������SnC12����������K2Cr2O7����Һ�ζ���Һ�е�Fe2+���ζ�ʱ��Ӧ�����ӷ���ʽΪ____________��
(4)������Һ����c(Ca2+)=1.0��10-3mol���������ʴﵽ99��ʱ�����ƺ����Һ��c(F-)=____mol��L-1[��֪Ksp(CaF2)=4.0��10-11]��
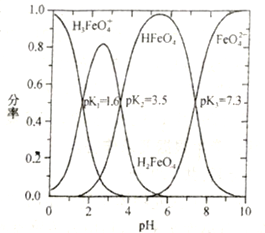
(5)�������У���ȡ������Һ�������(V0��VA)����Һ��Ni2+��Fe2+����ȡ��Ӱ����ͼ��ʾ��V0��VA�����ȡֵ��______________��

���𰸡� ����Ӵ�������ӿ췴Ӧ���ʣ�ʹ��Ӧ����� Fe3++2H2O![]() FeO(OH)��+3H+ SiO2��CaSO4 0.5 Cr2O72��+6Fe2++14H+
FeO(OH)��+3H+ SiO2��CaSO4 0.5 Cr2O72��+6Fe2++14H+![]() 2Cr3++6Fe3++7H2O 2.0��10��3 0.25
2Cr3++6Fe3++7H2O 2.0��10��3 0.25
�����������黯ѧ�������̣���1����ĥ����������泥���Ŀ��������Ӵ�������ӿ췴Ӧ���ʣ�ʹ��Ӧ����֣���2�����ݹ����У�������ˮ��������ˮ�ⷴӦ������Ӧ����ʽΪFe3��+2H2O ![]() FeO(OH)��+3H�������ݿ����еijɷ֣�SiO2����ˮ��Ӧ���������к���SiO2���������350�����Ϸֽ��NH3��H2SO4��CaO�����ᷴӦ����CaSO4��CaSO4����ˮ���������л�����CaSO4����3�����ݵ�ʧ������Ŀ�غ㣬n(SnCl2)��2=n(Fe3��)��1���ó�SnCl2�����ʵ���������Fe3�����ʵ�����0.5��������Fe2���Ļ�ԭ�ԣ���K2Cr2O7����������ԭ��Ӧ�������ӷ�Ӧ����ʽΪCr2O72��+6Fe2��+14H��=2Cr3��+6Fe3��+7H2O ����4�������ʴﵽ99%�����ƺ���Һ��c(Ca2��)=1.0��10��3��1%mol��L��1=1.0��10��5mol��L��1����c(F��)=
FeO(OH)��+3H�������ݿ����еijɷ֣�SiO2����ˮ��Ӧ���������к���SiO2���������350�����Ϸֽ��NH3��H2SO4��CaO�����ᷴӦ����CaSO4��CaSO4����ˮ���������л�����CaSO4����3�����ݵ�ʧ������Ŀ�غ㣬n(SnCl2)��2=n(Fe3��)��1���ó�SnCl2�����ʵ���������Fe3�����ʵ�����0.5��������Fe2���Ļ�ԭ�ԣ���K2Cr2O7����������ԭ��Ӧ�������ӷ�Ӧ����ʽΪCr2O72��+6Fe2��+14H��=2Cr3��+6Fe3��+7H2O ����4�������ʴﵽ99%�����ƺ���Һ��c(Ca2��)=1.0��10��3��1%mol��L��1=1.0��10��5mol��L��1����c(F��)=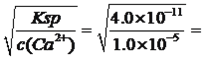 2.0��10��3mol��L��1����5����������ͼ��Fe2������ȡ��Ni2��������ȡ����˸���ͼ��V0/VA�����ȡֵΪ0.25��
2.0��10��3mol��L��1����5����������ͼ��Fe2������ȡ��Ni2��������ȡ����˸���ͼ��V0/VA�����ȡֵΪ0.25��