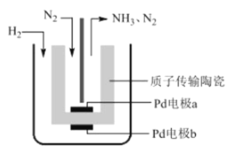
ЬтФПФкШн
ЁОЬтФПЁПФГЭЌбЇгУ10 mol/LЕФХЈбЮЫсХфжЦ250mL 1 mol/LЕФЯЁбЮЫсЃЌВЂНјаагаЙиЪЕбщЁЃЧыЛиД№ЯТСаЮЪЬтЃК
(1)ашвЊСПШЁХЈбЮЫс_________mLЁЃ
(2)ХфжЦИУЯЁбЮЫсЪБЪЙгУЕФвЧЦїГ§СПЭВЁЂЩеБЁЂВЃСЇАєЭтЃЌЛЙБиаыгУЕНЕФвЧЦїга_______ЁЂ______ЕШЁЃ
(3)ШЁЫљХфжЦЕФЯЁбЮЫс100mLЃЌгывЛЖЈжЪСПЕФаПГфЗжЗДгІЃЌаПШЋВПШмНтКѓЃЌЩњГЩЕФЦјЬхдкБъзМзДПіЯТЕФЬхЛ§ЮЊ0.896LЃЌдђВЮМгЗДгІЕФаПЕФжЪСПЮЊ______g ЃЛЩшЗДгІКѓШмвКЕФЬхЛ§ШдЮЊ100 mL ЃЌдђЗДгІКѓШмвКжаHЃЋЕФЮяжЪЕФСПХЈЖШЮЊ________ЁЃ
ЁОД№АИЁП25.0 НКЭЗЕЮЙм 250mLШнСПЦП 2.6 0.2mol/L
ЁОНтЮіЁП
(1)ЩшХЈбЮЫсЕФЬхЛ§ЮЊVЃЌдђ10molЁСV=1mol/LЁС0.25LЃЌV=0.025L=25.0mLЃЛ
(2)гУНКЭЗЕЮЙмЖЈШнЃЌгУ250mLШнСПЦПХфжЦШмвКЃЌЙЪД№АИЮЊНКЭЗЕЮЙмЃЛ250mLШнСПЦПЃЛ
(3)бЮЫсКЭаПЗДгІЕФЗНГЬЪНЮЊЃКZn+2HCl=ZnCl2+H2ЁќЃЌЩшВЮМгЗДгІЕФаПЕФжЪСПЮЊxЃЌВЮМгЗДгІЕФбЮЫсЕФЮяжЪЕФСПЮЊyЁЃ
Zn+2HCl=ZnCl2+H2Ёќ
65g 2mol 22.4L
x y 0.896L
x=![]() =2.6gЃЌy=
=2.6gЃЌy=![]() =0.08molЃЌЪЃгрбЮЫсЕФЮяжЪЕФСП=1mol/LЁС0.1l-0.08mol=0.02molЃЌдђЧтРызгХЈЖШ=
=0.08molЃЌЪЃгрбЮЫсЕФЮяжЪЕФСП=1mol/LЁС0.1l-0.08mol=0.02molЃЌдђЧтРызгХЈЖШ=![]() =0.2mol/LЃЌЙЪД№АИЮЊ2.6ЃЛ0.2mol/LЁЃ
=0.2mol/LЃЌЙЪД№АИЮЊ2.6ЃЛ0.2mol/LЁЃ