��Ŀ����
2����֪A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E������A��B��C��ͬһ���ڵķǽ���Ԫ�أ�������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��AC2���ĸ����ۼ���B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߣ�E��ԭ������Ϊ24���ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ����1��A��B��C�ĵ�һ��������С�����˳��ΪC��O��N��
��2��B���⻯��Ľṹʽ��
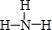 ��
����3��д��������AC2�ĵ���ʽ
 ��һ����B��C��ɵĻ�������AC2����������ȣ��仯ѧʽΪN2O��
��һ����B��C��ɵĻ�������AC2����������ȣ��仯ѧʽΪN2O����4��E�ĺ�������Ų�ʽ��1s22s22p63s23p63d54s1��
��5��B������������Ӧ��ˮ�����ϡ��Һ��D�ĵ��ʷ�Ӧʱ��B����ԭ����ͼۣ��÷�Ӧ�Ļ�ѧ����ʽ��4Mg+10HNO3=4Mg��NO3��2+NH4NO3+3H2O��
���� A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E��������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��C�γ�-2�������ӣ���Dλ��C����һ���ڣ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߣ������д��������C�γ�-2�������ӣ���CΪ��Ԫ�أ���DΪþԪ�أ����ں˵����B��C����BΪ��Ԫ�أ�A��B��C��ͬһ���ڵķǽ���Ԫ�أ������ڵڶ����ڣ���AC2Ϊ�Ǽ��Է��ӣ���AΪ̼Ԫ�أ�E��ԭ������Ϊ24����EΪCrԪ�أ��ݴ˽��
��� �⣺A��B��C��D��E�������ڱ���ǰ�����ڵ�Ԫ�أ����ǵĺ˵����A��B��C��D��E��������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��C�γ�-2�������ӣ���Dλ��C����һ���ڣ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߣ������д��������C�γ�-2�������ӣ���CΪ��Ԫ�أ���DΪþԪ�أ����ں˵����B��C����BΪ��Ԫ�أ�A��B��C��ͬһ���ڵķǽ���Ԫ�أ������ڵڶ����ڣ���AC2Ϊ�Ǽ��Է��ӣ���AΪ̼Ԫ�أ�E��ԭ������Ϊ24����EΪCrԪ�أ�
��1��AΪ̼Ԫ�ء�BΪ��Ԫ�ء�CΪ��Ԫ�أ�ͬ����������ҵ�һ�����ܳ��������ƣ���Ԫ��ԭ��2p�ܼ���3�����ӣ����ڰ����ȶ�״̬�����������ͣ���һ�����ܸ���ͬ�������ڵ�Ԫ�أ����Ե�һ��������С�����˳��ΪC��O��N��
�ʴ�Ϊ��C��O��N��
��2��BΪ��Ԫ�أ����⻯��ΪNH3��������Nԭ����Hԭ���γ�1�Թ��õ��Ӷԣ���ṹʽΪ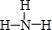 ��
��
�ʴ�Ϊ��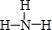 ��
��
��3��������AC2��CO2��������̼ԭ������ԭ��֮���γ�2�Թ��õ��Ӷԣ�����ʽΪ ��һ����NԪ�ء�OԪ�ػ�������CO2��Ϊ�ȵ����壬�仯ѧʽΪN2O��
��һ����NԪ�ء�OԪ�ػ�������CO2��Ϊ�ȵ����壬�仯ѧʽΪN2O��
�ʴ�Ϊ�� ��N2O��
��N2O��
��4��EΪCrԪ�أ�ԭ������Ϊ24��ԭ�Ӻ�����24�����ӣ���������Ų�ʽ�� 1s22s22p63s23p63d54s1��
�ʴ�Ϊ��1s22s22p63s23p63d54s1��
��5��B������������Ӧ��ˮ����ΪHNO3��D�ĵ���ΪMg��HNO3ϡ��Һ��Mg��Ӧʱ��NԪ�ر���ԭ����ͼۣ�������NH4NO3��Mg������ΪMg��NO3��2����NH4NO3��Mg��NO3��2�Ļ�ѧ�������ֱ�Ϊx��y������ݵ���ת���غ���[5-��-3��]��x=2y������x��y=4��1���÷�Ӧ�Ļ�ѧ����ʽ�ǣ�4Mg+10HNO3=4Mg��NO3��2+NH4NO3+3H2O��
�ʴ�Ϊ��4Mg+10HNO3=4Mg��NO3��2+NH4NO3+3H2O��
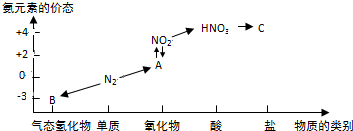
���� �����Ƕ����ʽṹ�Ŀ��飬�漰�ṹ����λ�ù�ϵ�������ܡ�����ʽ���������Ų���������ԭ��Ӧ�ȣ��Ѷ��еȣ��Ƕ�ѧ���ۺ������Ŀ��飬�⻯��ķе������ͬ����������Ԫ���⻯��ķе�����ƶϵ�ͻ�ƿڣ�

| A�� | M��NO3��2 | B�� | M��NO3��3 | C�� | MNO3 | D�� | M2��NO3��2 |
| A�� | �������� | B�� | �������� | C�� | �������� | D�� | �������� |
| A�� | H2O��HD��C3H8 | B�� | P2O5��CO2��H3PO4 | C�� | SO2��SiO2��CS2 | D�� | CCl4����NH4��2S��H2O2 |
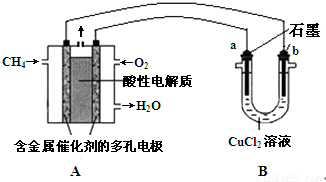
 ����������ԭ��Ӧ��2Ag+��aq��+Cu��s���TCu2+��aq��+2Ag��s����Ƶ�ԭ�����ͼ��ʾ��
����������ԭ��Ӧ��2Ag+��aq��+Cu��s���TCu2+��aq��+2Ag��s����Ƶ�ԭ�����ͼ��ʾ��
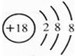 ��
��