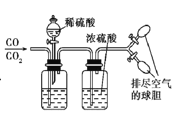
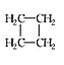��Ŀ����
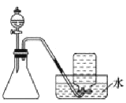
����Ŀ��Ϊ�ⶨijƷ��ϴ������(����)������ijѧ��������֪���ʵ���Ũ�ȵ�NaOH��Һ���ⶨ��ϴ��(����)�����ʵ���Ũ��ʱ��ѡ���̪��ָʾ��������д���пհף�
��1����ʽ�ζ��ܵ�ʹ�÷�������ȷ������Ⱥ�˳��Ϊ____(����ĸ)��
A����NaOH��Һ��ϴ����������NaOH��Һ
B���ų�NaOH��Һ���еζ�
C��������ʼ����
D����©����ˮϴ2��3��
��2���ñ���NaOH��Һ�ζ����������ʱ�����ֿ��Ƽ�ʽ�ζ��ܵIJ���������ҡ����ƿ���۾�Ӧע��___��
��3�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼ��ʾ��������NaOH��Һ�����Ϊ___mL��
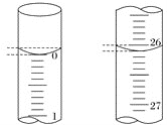
��4��ijѧ����������ʵ��ֱ��¼�й��������±���
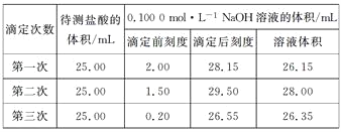
��ѡ�����к��������ݼ����ϴ������(����)�����ʵ���Ũ�ȣ�c(HCl)��____��
��5���������в���������ϴ������(����)��Ũ�ȵ�Ӱ�죺
A����ȡ����Һʱ����ʼ���Ӷ��������Ӷ�������____(����ƫ��������ƫС��������Ӱ��������ͬ)��
B������ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ����____��
���𰸡�DACB ��ƿ����Һ��ɫ�ı仯 26.10 0.105 mol��L-1 ƫ�� ƫ��
��������
(1)�ζ������е���Ҫ�������裺���ζ����Ƿ�©ˮ����ϴ�ζ��ܡ�װҺ��������ʼ�������ζ���
��2�����ݵζ������ľ���Ҫ��ش�
��3������NaOH��Һ�����Ϊ���ζ����IJ
(4)���εζ��еڶ��������������������ϴ���ȥ��![]() (��Һ)��26.25mL������n(HCl)=n(NaOH)���㣻
(��Һ)��26.25mL������n(HCl)=n(NaOH)���㣻
(5)����c(����)��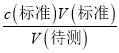 ������
������
(1)�ζ������е���Ҫ�������裺���ζ����Ƿ�©ˮ����ϴ�ζ��ܡ�װҺ��������ʼ�������ζ����յ��жϡ����㡢���ݴ�������ȷ��˳����DACB��
(2)�ڵζ����������ֿ��Ƽ�ʽ�ζ��ܵIJ���������ҡ����ƿ���۾�Ӧע����ƿ����Һ��ɫ�ı仯���ж��յ㡣
(3)��ʽ�ζ�������ʼ����Ϊ0.00 mL���յ����Ϊ26.10 mL������NaOH��Һ���Ϊ26.10 mL��
(4)���εζ��еڶ��������������������ϴ���ȥ��![]() (��Һ)��26.25mL�����ݷ�Ӧ����ʽ��n(HCl)=n(NaOH)�� c(HCl)��25.00mL=0.1000 mol��L��1��26.25 mL������c(HCl)��0.105 mol��L��1��
(��Һ)��26.25mL�����ݷ�Ӧ����ʽ��n(HCl)=n(NaOH)�� c(HCl)��25.00mL=0.1000 mol��L��1��26.25 mL������c(HCl)��0.105 mol��L��1��
(5) A.����c(����)��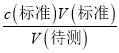 ��ȡ����Һʱ�����Ӻ��ӣ�����Һ�����ʵ���ƫ�����V(��)ƫ��c(����)ƫ��
��ȡ����Һʱ�����Ӻ��ӣ�����Һ�����ʵ���ƫ�����V(��)ƫ��c(����)ƫ��
B.����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ�����V(��)ƫ��֪c(����)ƫ��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
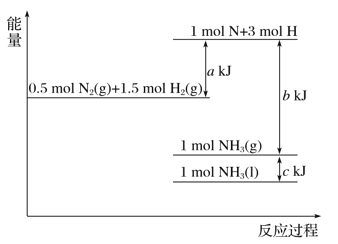
Сѧ��10����Ӧ����ϵ�д�����Ŀ���ס������������ܱ������о�������Ӧ��C(s)��2H2O(g)=CO2(g)��2H2(g)��H��0���й�ʵ���������±���ʾ��
���� | �ݻ�/L | �¶�/�� | ��ʼ��/mol | ƽ����/mol | ƽ�ⳣ�� | |
C(s) | H2O(g) | H2(g) | ||||
�� | 2 | T1 | 2.0 | 4.0 | 3.2 | K1 |
�� | 1 | T2 | 1.0 | 2.0 | 1.2 | K2 |
����˵����ȷ����(����)
A.K1��12.8
B.T1��T2
C.T1 ��ʱ����������ٳ���0.1 mol H2O(g)����ƽ�������ƶ���CO2(g)�������������
D.��T2�¶��£���2 L�����ܱ������г���1.0 mol CO2��2.0 mol H2����ƽ��ʱ��CO2��ת���ʴ���40%
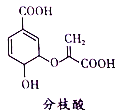
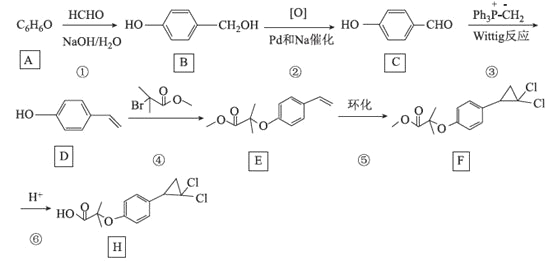
����Ŀ�����������Ϊȫ��������������������Ҫ���л��ܼ����±��Dz��ֻ�����������������Ľṹ��ʽ������ʽ��ijЩ�л�������ķ�Ӧʽ(����Pt��Ni�Ǵ���)��
�ṹ��ʽ |
|
|
|
����ʽ |
|
|
|
��![]() +H2
+H2![]()
![]()
��![]() +H2
+H2![]()
![]()
��![]() +H2
+H2![]()
![]()
��![]() +KMnO4
+KMnO4![]()
![]() +
+![]() +Mn2+
+Mn2+
�ش��������⣺
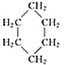
(1)��������___________��ͬ���칹�塣
(2)�ӷ�Ӧ�١��ۿ��Կ������������������ӳɷ�Ӧ�Ļ�������______(������)���ж�����Ϊ_________��
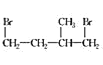
(3)������������±�ص��ʡ�±���ⷢ�����ƵĿ����ӳɷ�Ӧ���绷������HBr��һ��������Ӧ���仯ѧ����ʽΪ(����ע����Ӧ����)____________��
(4)д�����������ϩ��һ�ַ������Լ�___________����������� __________��