��Ŀ����
��ҵ�Ͽ���ʳ�κ�ʯ��ʯΪ��Ҫԭ�ϣ�����ͬ�ķ������������ش��������⣺
��1��¬����������ʳ�Ρ�ʯ��ʯ��Ũ���ᡢ��̿Ϊԭ�ϣ��ڸ����½������գ��ٽ�ȡ���ᾧ���Ƶô��
��ʳ�κ�Ũ���ᷴӦ�Ļ�ѧ����ʽΪ___________��
�������ƺͽ�̿��ʯ��ʯ��Ӧ�Ļ�ѧ����ʽΪ_________����֪����֮һΪCaS����
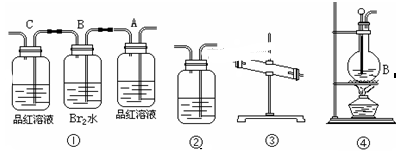
��2������Ĺ�������ͼ��ʾ���õ���̼�����ƾ��������ɴ��
| | |
| |  |
��װ�����з�����Ӧ�Ļ�ѧ����ʽΪ_______��

��3�������Ƽ����ĸĽ������ŵ���______________��
��4��������Ϊ̼�������̼�����ƵĻ�ѧ�������ƣ���Ҳ���ð�����Ȼ��غ�ʯ��ʯΪԭ����̼��ء�������ͼ���ܽ�ȣ�S�����¶ȱ仯���ߣ�����˵���Ƿ���У�__________��
��1����2NaCl+H2SO4(Ũ)  Na2SO4+2HCl��
Na2SO4+2HCl��
��Na2SO4+4C+CaCO3 CaS+Na2CO3+4CO��
CaS+Na2CO3+4CO��
Na2SO4+2C+CaCO3 CaS+Na2CO3+2CO2
CaS+Na2CO3+2CO2
��2����Ca��OH��2 NH3
��NH3+CO2+NaCl+H2O=NaHCO3��+NH4Cl
��3�������ԭ�ϵ������ʣ����ٷ�����CaCl2�����ŷţ������˰�����ŵ㣬����������ȱ�㣬ʹʳ�ε���������ߣ�NH4Cl �������ʣ�����ϳɰ������ϣ�ʹ�ϳɰ���ԭ���� CO ת���� CO2������ CaCO3�� CO2��һ����
��4�������У���ΪKHCO3��NH4Cl���ܽ�������¶ȸ���40��ʱ��KHCO3���ܽ�ȴ���NH4Cl�����½ᾧʱ�������϶��KCl��
����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
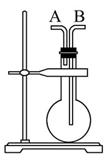
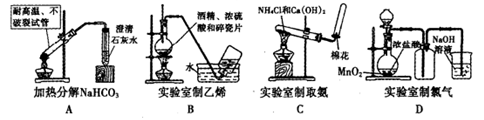
Сѧ��10����Ӧ����ϵ�д�ͬһ���Ʊ�װ�ÿ�������ȡ��ͬ�����壬��ֻ����ͼװ����ȡ���壬��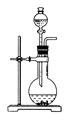
(1)�����±��ո�����������ҩƷ��
| �������� | ҩƷ | |
| ��Һ©���� | ��ƿ�� | |
| O2 | | MnO2 |
| H2 | ϡH2SO4 | |
| NH3 | | CaO |
| NO | ϡHNO3 | Cu |
| CO2 | ϡH2SO4 | ʯ��ʯ |
| C2H4 | C2H5OH | ϡH2SO4 |
| C2H2 | | CaC2 |
(2)д������ҩƷ�Ʊ�O2�Ļ�ѧ����ʽ ��
(3)д������ҩƷ�Ʊ�C2H2�Ļ�ѧ����ʽ ��
(4)�뻭������NH3��װ��ͼ�����������������������
(5)���и�ȫҩƷ����Ŀ�У����������Ʊ���Ӧ�������(�ɲ�����)��
������ ������ ��
������ ������ ��
������ ������ ��
��ѧС���������������������װ��(����ͼ)���û������Ʊ�����ϩ��
��֪��
| | �ܶ�(g/cm3) | �۵�(��) | �е�(��) | �ܽ��� |
| ������ | 0.96 | 25 | 161 | ������ˮ |
| ����ϩ | 0.81 | ��103 | 83 | ������ˮ |

(1)�Ʊ���Ʒ
��12.5 mL�����������Թ�A�У��ټ���1 mLŨ���ᣬҡ�Ⱥ�������Ƭ����ֹ���У���������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
�ٵ���B���˵�������е�������________��
���Թ�C���ڱ�ˮԡ�е�Ŀ����______________________________��
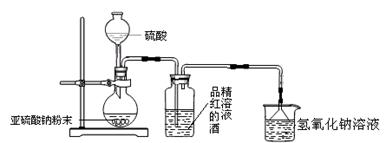
(2)�Ʊ���Ʒ
�ٻ���ϩ��Ʒ�к��л������������������ʵȡ����뱥��ʳ��ˮ�������á��ֲ㣬����ϩ��________��(��ϡ����¡�)����Һ����________(������)ϴ�ӡ�
A��KMnO4��Һ B��ϡH2SO4 C��Na2CO3��Һ

���ٽ�����ϩ����ͼװ��������ȴˮ��________�ڽ���(�g����f��)������ʱҪ������ʯ�ң���Ŀ����_____________________________________��
���ռ���Ʒʱ�����Ƶ��¶�Ӧ��________���ң�ʵ���ƵõĻ���ϩ��Ʒ�����������۲��������ܵ�ԭ����________��
A������ʱ��70 �濪ʼ�ռ���Ʒ
B��������ʵ����������
C���Ʊ���Ʒʱ���������Ʒһ������
(3)�������ֻ���ϩ��Ʒ�ʹ�Ʒ�ķ�������������________��
A���ֱ�������Ը��������Һ
B���ֱ�����ý�����
C���ֱ�ⶨ�е�
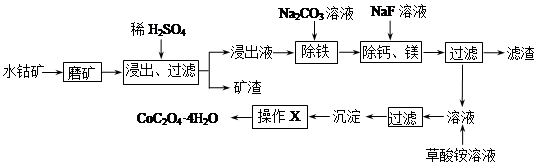

 ��
�� CoO2+LiC6��д���õ�طŵ�ʱ��������Ӧʽ ��
CoO2+LiC6��д���õ�طŵ�ʱ��������Ӧʽ ��


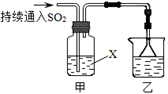
 Ca2��+
Ca2��+