��Ŀ����
��10�֣�ijУ��ѧ�о���ѧϰС����������˽�������ݣ��Ҷ���(HOOC-COOH���ɼ�дΪH2C2O4)�׳Ʋ��ᣬ������ˮ�����ڶ�Ԫ��ǿ��(Ϊ�������)��������ǿ��̼�ᣬ���۵�Ϊ101.5�棬��157��������Ϊ̽������IJ��ֻ�ѧ���ʣ�����������ʵ�飺
(1)��ʢ��1 mL����NaHCO3��Һ���Թ��м��������Ҷ�����Һ���۲쵽����ɫ���ݲ������÷�Ӧ�����ӷ���ʽΪ___________________________________��
(2)��ʢ���Ҷ��ᱥ����Һ���Թ��е��뼸�������ữ��KMnO4��Һ������������Һ���Ϻ�ɫ��ȥ��˵���Ҷ������_____________(������ԡ�������ԭ�ԡ������ԡ�)������ƽ�÷�Ӧ�����ӷ���ʽ��
____ MnO4�C + ____ H2C2O4 + ____ H+ =" ____" Mn2+ + ____ CO2�� + ____ H2O
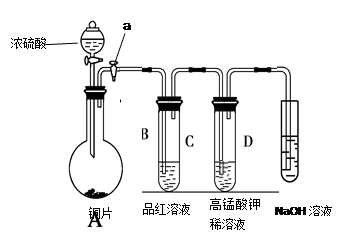
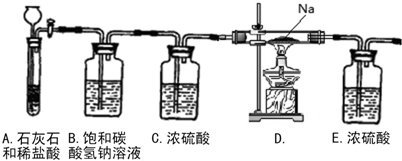
(3)��һ�������Ҷ�������Թ��У�����ͼ��ʾװ�ý���ʵ��(ע�����Բ�����������Ҽг�װ��δ���)��
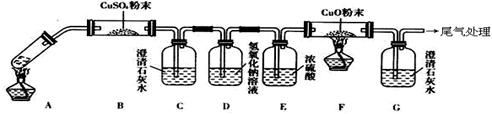
ʵ�鷢�֣�װ��C��G�г���ʯ��ˮ����ǣ�B��CuSO4��ĩ������F��CuO��ĩ�к�ɫ��Ϊ��ɫ���ݴ˻ش�
����װ���У�D��������__________________.
�Ҷ���ֽ�Ļ�ѧ����ʽΪ______________________________________.
(4)��С��ͬѧ��2.52 g���ᾧ��(H2C2O4��2H2O)���뵽100 mL 0.2 mol/L��NaOH��Һ�г�ַ�Ӧ����÷�Ӧ����Һ�����ԣ�������Һ����仯���������й�ϵ��������
A��c(Na+)+c(H+)=c(HC2O4��)+c(OH��)+c(C2O42-)
B��c(HC2O4-)+c(C2O42-)="0.2" mol��L-1
C�� c(HC2O4-)>c(C2O42��)>c(H2C2O4)
D��c(Na+)=c(H2C2O4)+c(HC2O4-)+c(C2O42��)
(1)��ʢ��1 mL����NaHCO3��Һ���Թ��м��������Ҷ�����Һ���۲쵽����ɫ���ݲ������÷�Ӧ�����ӷ���ʽΪ___________________________________��
(2)��ʢ���Ҷ��ᱥ����Һ���Թ��е��뼸�������ữ��KMnO4��Һ������������Һ���Ϻ�ɫ��ȥ��˵���Ҷ������_____________(������ԡ�������ԭ�ԡ������ԡ�)������ƽ�÷�Ӧ�����ӷ���ʽ��
____ MnO4�C + ____ H2C2O4 + ____ H+ =" ____" Mn2+ + ____ CO2�� + ____ H2O
(3)��һ�������Ҷ�������Թ��У�����ͼ��ʾװ�ý���ʵ��(ע�����Բ�����������Ҽг�װ��δ���)��

ʵ�鷢�֣�װ��C��G�г���ʯ��ˮ����ǣ�B��CuSO4��ĩ������F��CuO��ĩ�к�ɫ��Ϊ��ɫ���ݴ˻ش�
����װ���У�D��������__________________.
�Ҷ���ֽ�Ļ�ѧ����ʽΪ______________________________________.
(4)��С��ͬѧ��2.52 g���ᾧ��(H2C2O4��2H2O)���뵽100 mL 0.2 mol/L��NaOH��Һ�г�ַ�Ӧ����÷�Ӧ����Һ�����ԣ�������Һ����仯���������й�ϵ��������
A��c(Na+)+c(H+)=c(HC2O4��)+c(OH��)+c(C2O42-)
B��c(HC2O4-)+c(C2O42-)="0.2" mol��L-1
C�� c(HC2O4-)>c(C2O42��)>c(H2C2O4)
D��c(Na+)=c(H2C2O4)+c(HC2O4-)+c(C2O42��)
(1)HCO3�C + H2C2O4 = HC2O4�C+ CO2��+ H2O(2��)
(2) ��ԭ��(1��) 2 5 6 2 10 8��2��)
(3) ��ȥ��������е�CO2 (1��)�� H2C2O4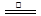 H2O+CO��+CO2�� (2��)
H2O+CO��+CO2�� (2��)
(4) C D(2��)
(2) ��ԭ��(1��) 2 5 6 2 10 8��2��)
(3) ��ȥ��������е�CO2 (1��)�� H2C2O4
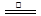 H2O+CO��+CO2�� (2��)
H2O+CO��+CO2�� (2��)(4) C D(2��)
���������(1)��ɫ����Ϊ������̼��HCO3�C + H2C2O4 = HC2O4�C+ CO2��+ H2O
��2������KMnO4Ϊǿ����������ɫ˵���Ҷ�����л�ԭ�ԡ��ɵ�ʧ�����غ��Ԫ���غ���ƽ��
��3���������Ƶ�����Ϊ��ȥ��������е�CO2��װ��C��G�г���ʯ��ˮ����ǣ�˵���ж�����̼��B��CuSO4��ĩ������˵����ˮ������F��CuO��ĩ�к�ɫ��Ϊ��ɫ��˵����һ����̼���ֽⷽ��ʽ��CO2H2C2O4
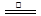 H2O+CO��+CO2�� ��4����Ӧ������ԣ��ɵ� c(HC2O4-)>c(C2O42��)>c(H2C2O4)�������������ӵ���غ�;c(Na+)=c(H2C2O4)+c(HC2O4-)+c(C2O42��).
H2O+CO��+CO2�� ��4����Ӧ������ԣ��ɵ� c(HC2O4-)>c(C2O42��)>c(H2C2O4)�������������ӵ���غ�;c(Na+)=c(H2C2O4)+c(HC2O4-)+c(C2O42��).������������һ��ʵ��̽���⣬����ѧ�������ͽ�������������ע��ƽʱֻ�ǵĻ����ǽ���Ĺؼ����ۺ��Խ�ǿ���Ѷȴ�

��ϰ��ϵ�д�
 ��������������������ϵ�д�
��������������������ϵ�д�
�����Ŀ
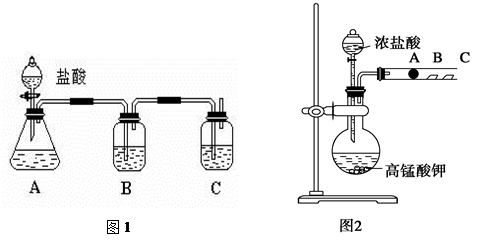
 ��CCl4��S2Cl2���� 2S��Cl2
��CCl4��S2Cl2���� 2S��Cl2 ��S2Cl2��
��S2Cl2�� 2SCl2��
2SCl2��